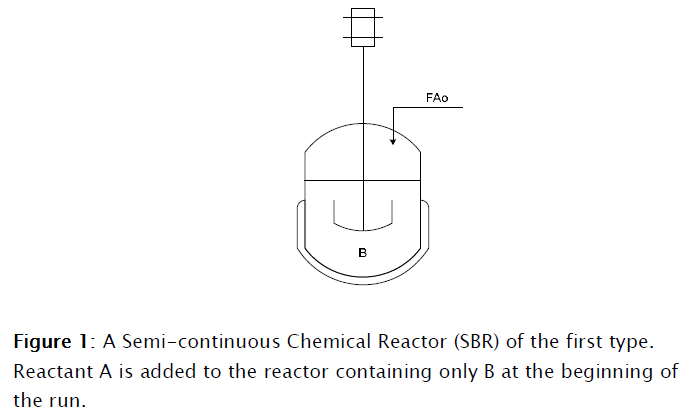
The Semicontinous Reactor: Modeling, Simulation and Control Part I Modeling and Simulation
Rónanth Zavaleta Mercado
Member of the Academy of Sciences of Bolivia
Dean of the School of Engineering, Universidad Católica Boliviana San Pablo. 14 de Septiembre Nº 4807, Obrajes, La Paz, Bolivia.
ronanth.zavaleta@gmail.com
Recibido: 1 de septiembre 2013 ]]> Aceptado: 14 de diciembre 2014
Abstract:
Conservation equations are applied to the Semicontinuous Reactor, also known as the Semibatch Reactor (SBR) for the isothermal, adiabatic and nonisothermal case, to generate the necessary differential equations for modeling and simulation. The resulting equations, expressed in terms of dimensionless normalized variables, are solved numerically for consistency testing by means of computer algebra software (Mathcad®). The model is also expressed in terms of meaningful dimensionless groups of the Damköhler and Biot type. The consistent model is then simulated for typical combinations of operating parameters in preparation for process control and stability simulation and analysis.
The SBR is typically non steady, of relatively high operating costs and low throughputs. However high conversions can be achieved and good temperature control allows for the minimizations of unwanted side reactions by means of maintaining low concentrations of one of the reactants. This reactor is extensively used in the manufacture of high value added chemicals (fine chemicals) and in reactions where a careful control is mandatory, such as in the production of high explosives. It is used in case of multiphase reactions involving gas bubbling such as the production of mono, di and tri chlorobenzene.
Part I of this article is devoted to generating a robust modeling and simulation tool for the SBR, while Part II explores its control and stable behavior.
Key words: Semicontinuous Reactor, Semibatch Reactor, SBR control, stable behavior of reactors.
]]> Introduction
A Semicontinuous Reactor or also called Semibatch Reactor (SBR) is used for small scale operations, for testing new processes in process development engineering, for the manufacture of expensive products, for processes that are difficult to be converted to continuous operation and for those processes that require a tight control of operating parameters and the minimization of unwanted side reactions by means of reactant concentration manipulation. Thus, it is used to carry out reactions involving explosives and the like. It is also used when multiphase reactions are being carried out, such as the continuous bubbling of a gaseous reactant in a liquid medium.
The SBR is inherently unsteady and there are two basic types of SBR operations. In the first one, as shown in Figure 1, a reactant (A) is slowly added to a reactor containing another reactant B, loaded prior to the beginning of the reaction run. Several tactics of addition of A can be applied, as well as different set ups. The reactant A can be added continuously at a constant rate as the reaction proceeds, or fed in a varying rate sequence as a function of time. This approach is used mainly when unwanted side reactions are to be avoided, or when the reactions involved are highly exothermic. In some reactions, the reactant A is a gas that is continuously bubbled through liquid B, such as in the cases of reactions involving ammonolysis, chlorination and hydrolysis. In the second type, reactants A and B are charged simultaneously and one of the products is continuously withdrawn in order to shift equilibrium and attaining this way, higher conversions. Also higher concentrations are achieved this way, with a corresponding reduction of purifying costs, increase rate of reaction and reduced reaction times. When one of the reactants is removed from the reactor by vaporization the reaction operation is called reactive distillation, such as the case of acetylation and esterification reactions. One typical example of the later one is the production of ethyl acetate and other aromatic esters, where the reversible reaction equilibrium is shifted to the right increasing the achievable conversion by removal of a volatile byproduct. It is also the case when glycerin is continuously withdrawn in the production of biodiesel, even though for this reaction the byproduct is not vaporized but rather removed as an insoluble dense phase.

Only the first type of SBR is considered for the present work.
1. Conservation of matter applied to SBR: the mathematics.
1.1 Second order elementary reaction rate: the isothermal case.
Consider the following elementary liquid phase reaction
]]>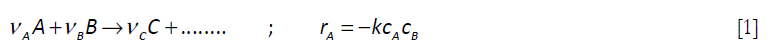
where A is added to a vessel containing pure B, where the chemical reaction takes place. Balances performed on the number of moles of A and B, taking the system as the contents of the reactor, leads to, for A

NA is the number of moles of A, FAo, the molar feed rate, V the volume of reactants and t, time. For B,
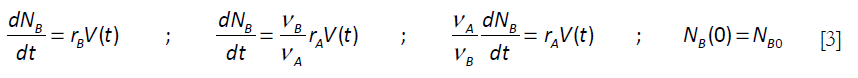
For the elementary chemical reaction considered, rA / VA = rB / VB ; rB = rA (VB / VA) . The following equation is derived by elimination of the chemical reaction rate terms in equations [2] and [3]:
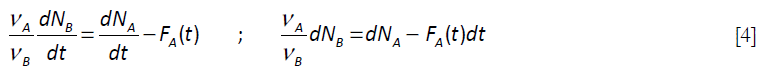
that upon integration renders.
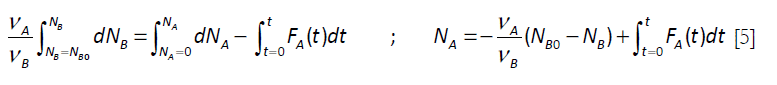
This expression for the number of moles of A can be introduced in Equation [3], with the rate expression written in terms of the number of moles
]]>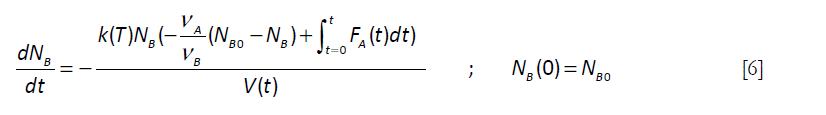
For the simple case for which the feed rate is constant (FA)
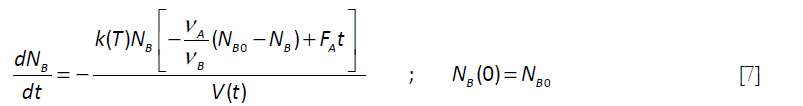
The volume V is a function of time In Equation [7]. Its time dependence can be derived by a global material balance on the reactor contents, ρ being the density of the reactor contents and ρo that of the feed

If the density of the reactors content is the same as the density of the feed

that can be integrated to give
![]()
By substitution in Equation [7]
]]>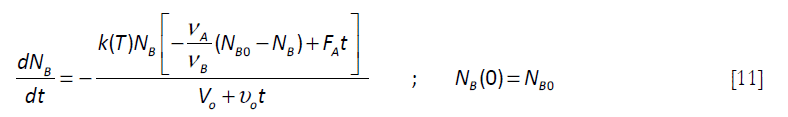
This equation has the restriction that the feed FAf stops when the total number of moles of A, NAf, has been added to the system. Therefore a feeding time tf is defined by the equation

For the case of constant feed rate, the feeding time becomes
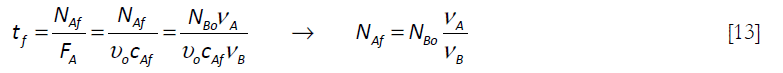
Equation [11] can be converted to a dimensionless form using the following variables, the conversion X of B and the initial spatial time τo = Vo/υo
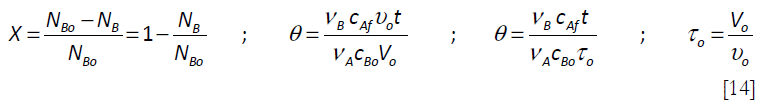
to give the following equation
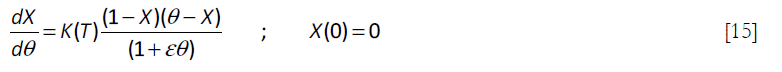
where ![]() the ratio of the stoichiometric initial concentrations of B in the reactor and A in the feed. The K(T) term is
the ratio of the stoichiometric initial concentrations of B in the reactor and A in the feed. The K(T) term is ![]() , that can be expressed in in a more convenient form
, that can be expressed in in a more convenient form
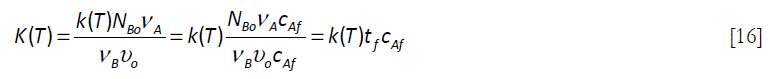
From Equation [14], for a dimensionless final feed time corresponding to θ = 1,

Therefore, Equation [16] becomes
![]()
And the final equation for the SBR up to the final feed time is
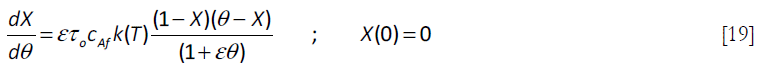
For the cases where a stoichiometric amount of A is added to the reaction mixture, the reaction proceeds as if it were taking place in a batch reactor after the addition of the reactant A is terminated, that occurs when θ = 1. Thus, for θ > 1, the equation reduces to
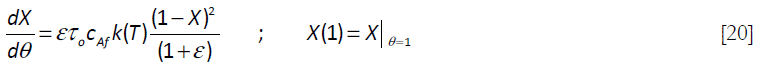
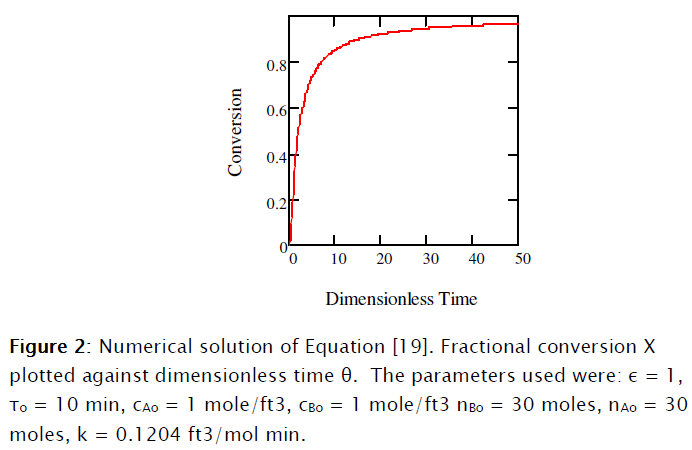
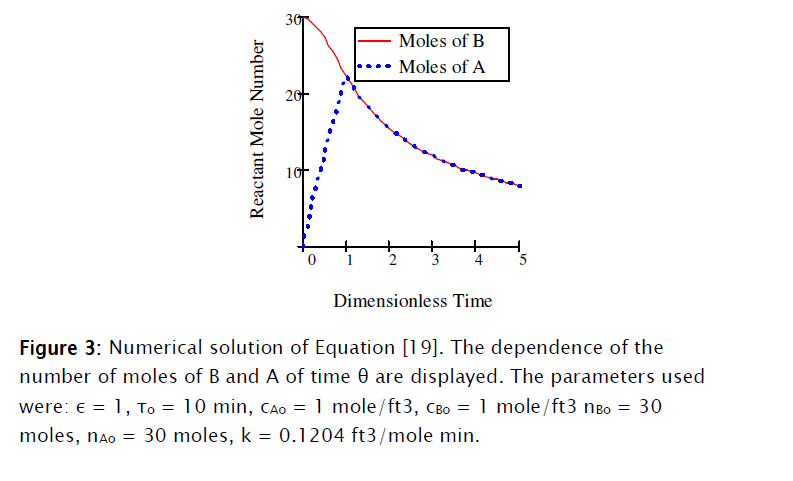
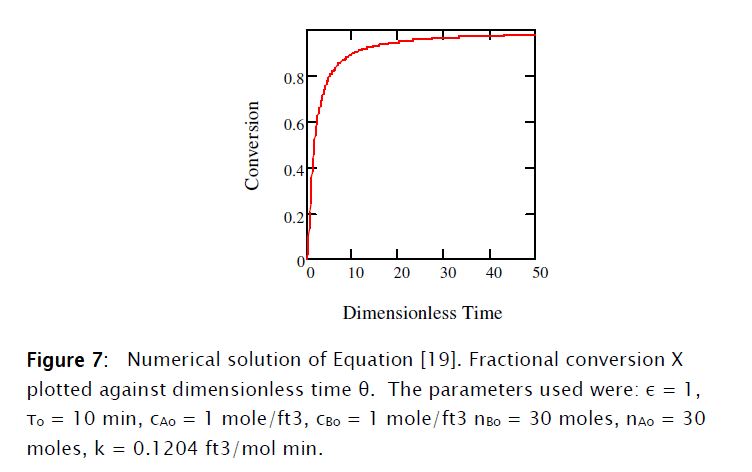
This equation was solved numerically using Mathcad 15® as shown in Subprogram 1.
]]>
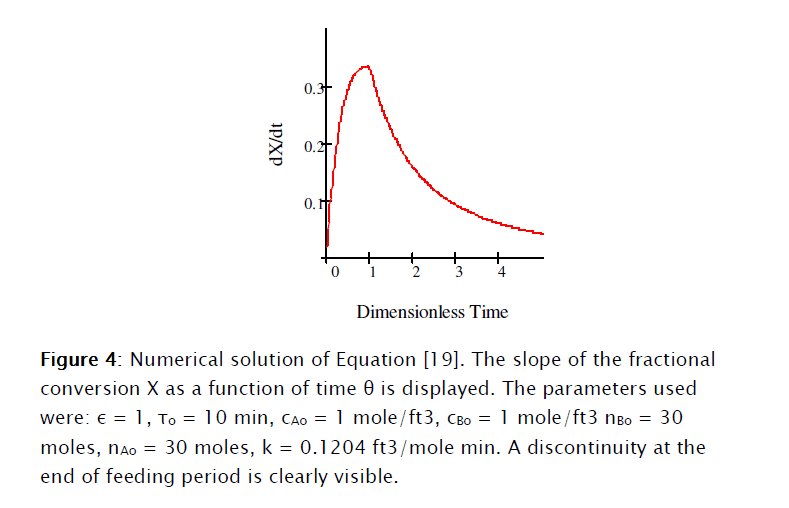


]]> 2. Second order elementary reaction rate: the adiabatic case.
For this case an adiabatic energy balance is required

or

Where nT refers to the total number of moles, Um, the molar internal energy, Hm,f the molar enthalpy of the feed stream and Ff, the molar feed rate. If species A, B, C, D and S (solvent) are the chemical species in the reactor, then
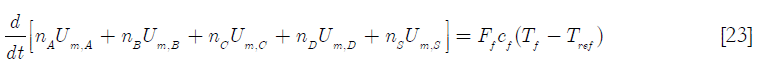
Here cf is the molar specific heat capacity of the feed, Tf the feed temperature and Tref a reference temperature. Expanding Equation [22] and neglecting the internal energy of mixing
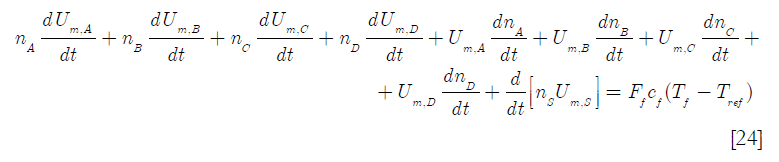
If it is assumed that νA = νB = νC = νD = 1, applying the extent of reaction definition
]]>
that upon substitution and rearranging in Equation [24] becomes
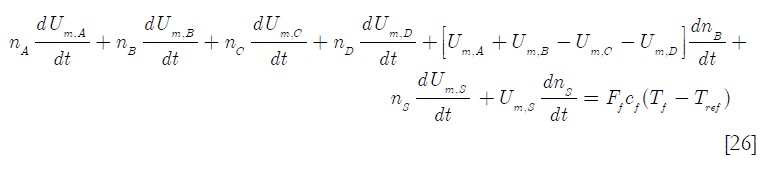
where ns stands for the number of moles of solvent
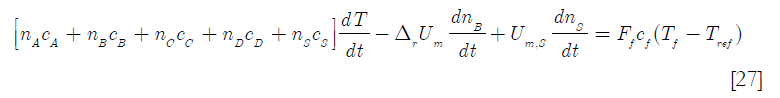
since, by definition

A mass balance on the solvent s gives, csf being the molar concentration of solvent in the feed
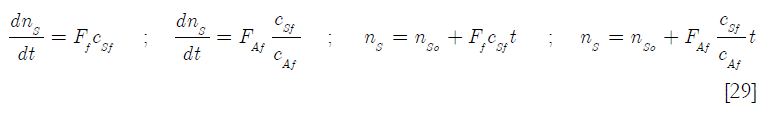
where nso is the initial number of moles of solvent in the reactor. By substitution in Equation [27]
]]>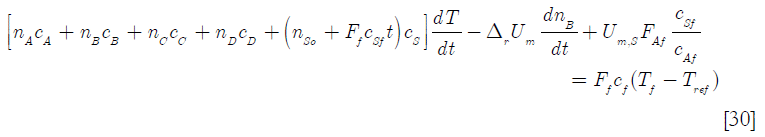
The total number of moles of reactants and solvent present in the reactor at the beginning g of the run, nto, are

Since the density and the heat capacity can be considered constant due to the relative low concentration of reactants, then the initial heat capacity of the reactor contents is
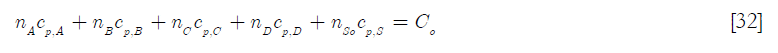
If the feed of reactant A and the solvent associated with it is taken into account, then a heat capacity of the reactors content, C(t) can be derived, taking into account that the number of moles is constant due to the stoichiometry of the reaction, except for the addition of reactant A and the accompanying solvent

Since Fftcf is the heat capacity of the feed. Then Equation [29] becomes, accepting also that for a diluted liquid reaction Um ≈ Hm

Therefore
]]>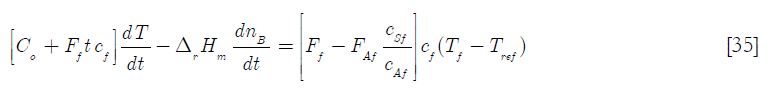
And also, assuming that the average specific heat capacity of the feed is about the same as that of A

Therefore equation [30] becomes

The denominator of this equation is simply the heat capacity of the reactor contents, C = C(t)
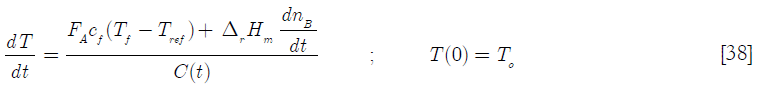
and for the BR

If the reactant feed is fed at the reference temperature, the equations become for the SBR and BR reactors
]]>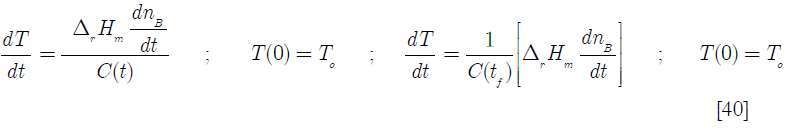
That can be expressed in dimensionless form by means of the following variables, where Ts refers to the set point (the desired temperature for the reaction to be carried out at) and Tref, a reference temperature, say 298 K)
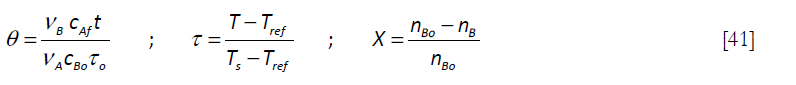
and
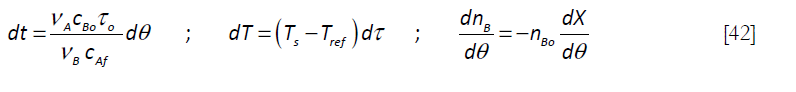
to give
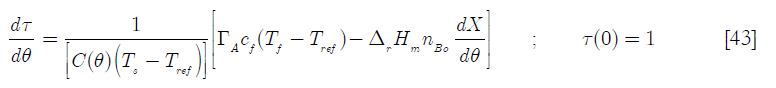
where ΓA is the A reactant feed in terms of the dimensional time defined before. For the BR case, after the feed period elapses
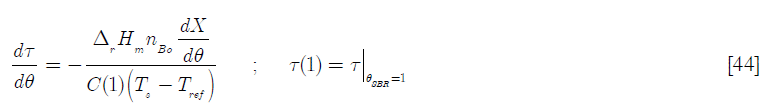
The equations for the SBRBR reactors become
]]>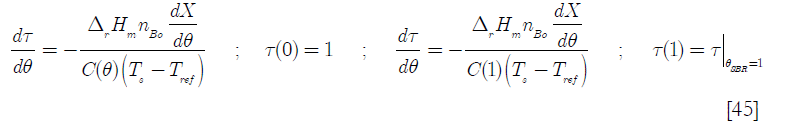
where
![]()
In this equation, the term Γ A is the feed rate of reactant A in moles of A for the dimensionless time (the feed ends at θ = 1. For the BR reactor, the term ![]() is a Damköhler type dimensionless group (Da) that represents the ratio of heat generated by chemical reaction when the total number of moles of B reacts over the heat capacity of the reactor’s content when the feed has been concluded, referred to the set point temperature. Thus
is a Damköhler type dimensionless group (Da) that represents the ratio of heat generated by chemical reaction when the total number of moles of B reacts over the heat capacity of the reactor’s content when the feed has been concluded, referred to the set point temperature. Thus
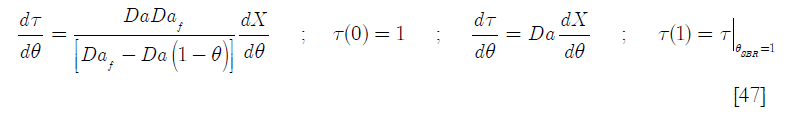
Here the Damköhler type dimensionless groups are, (including the Damköhler group referred to the feed Daf)
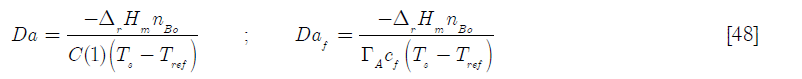
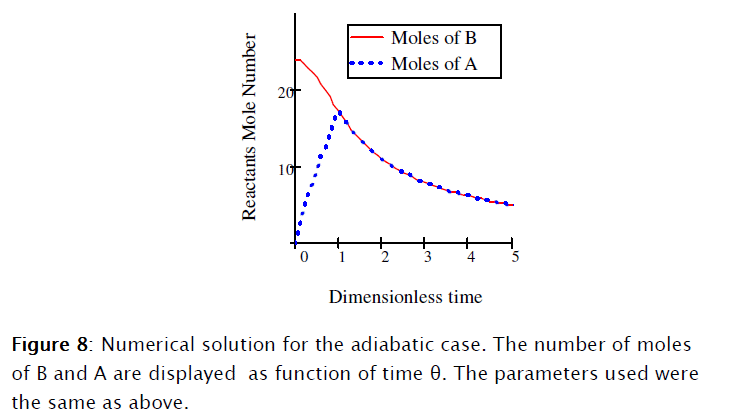
The set of resulting differential equations [19], [20], [37] and [38] I solved using Mathcad 15, as shown in Subprogram 2.
]]>
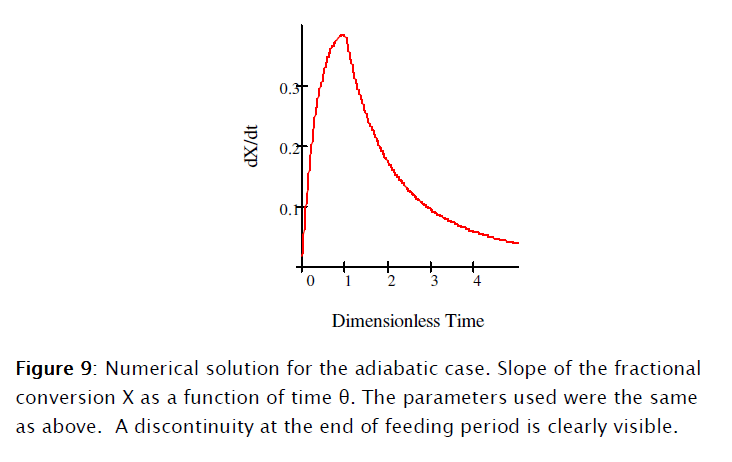
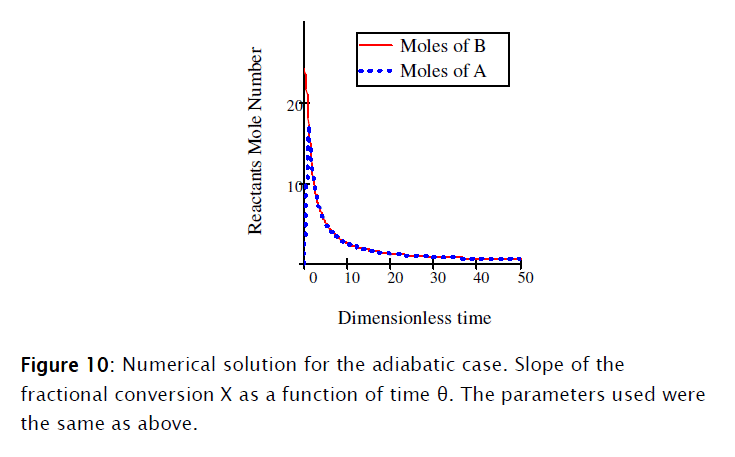
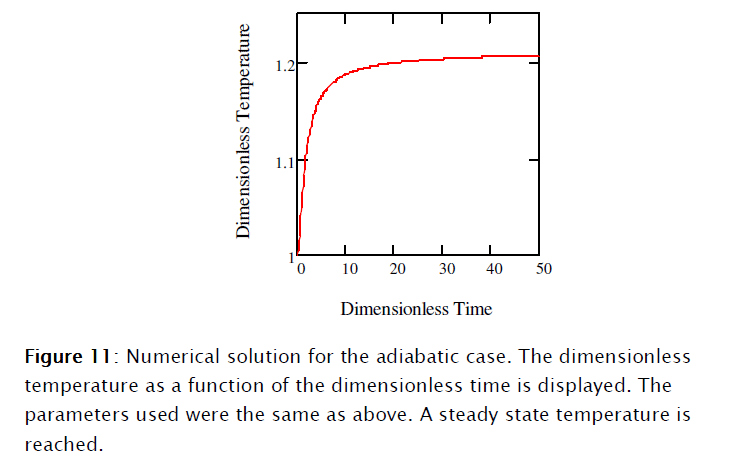
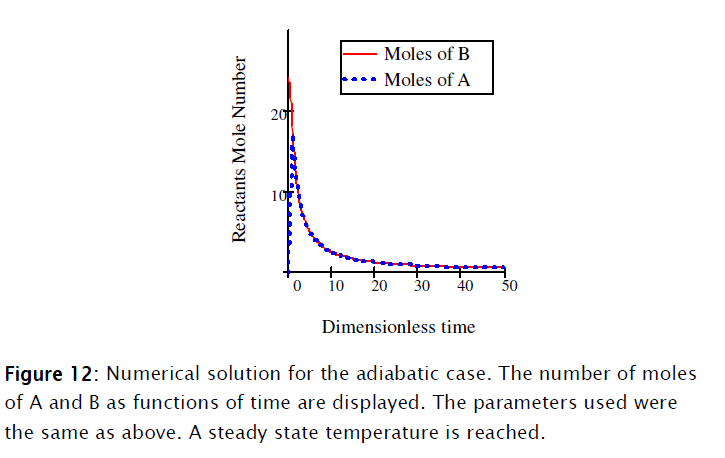
]]>
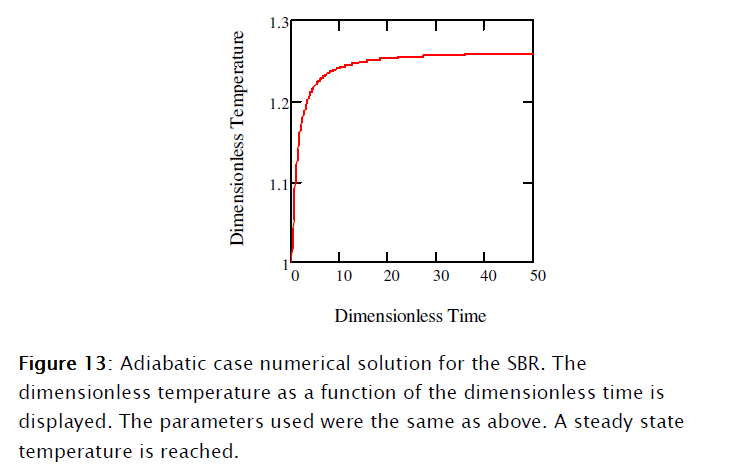

3. Second order elementary reaction rate: the non adiabatic case
For this case the mass balance is, of course, the same as before. The energy balance of Equation [38] becomes
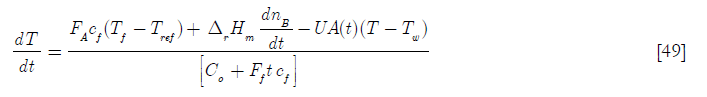
where the cooling effect of the water jacket has been added, the heat transfer area A being a function of time. In terms of the dimensionless variables defined before. Tw is the temperature of the cooling jacket water Thus Equation [49] becomes
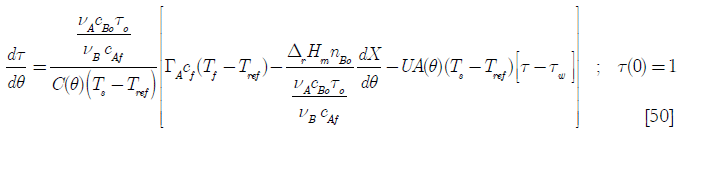
Here the dimensionless temperature τw of the cooling media is
]]>
Setting the feed temperature Tf equal to the reference temperature Tref
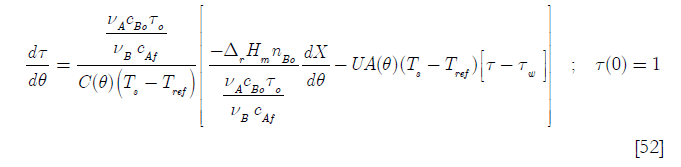
Since the density of the reacting media is supposed constant, as is the specific heat capacity, then the heat transfer area A(t) is proportional to the number of moles of the reactor contents at any time
![]()
and therefore

Rearranging Equation [52]
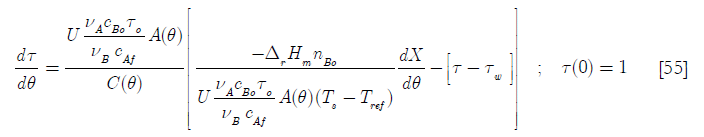
and
]]>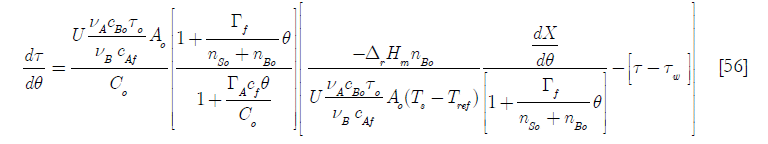
There are some meaningful dimensionless groups in this equation
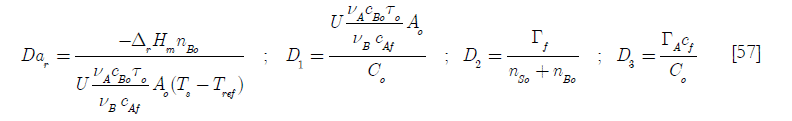
Here the Dar group, another Damköhler type dimensionless group related to the chemical reaction is the ratio of the total heat generated by it when the total number of moles B have been consumed to the initial heat transfer rate; D1, the heat transfer per unit temperature difference taking into account the initial area available for heat transfer over the initial heat capacity; D2 the ratio of the molar feed over the initial number of moles of reactants and D3 the ratio of the heat capacity of the feed of reactant A over the initial contents heat capacity of the reactor.
Upon substitution of Equation [57] on Equation [56] for the SBR up to θ = 1
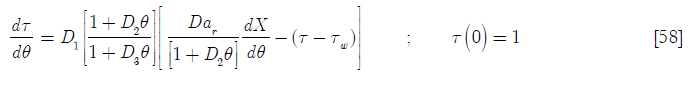
And for the BR behavior (θ > 1)
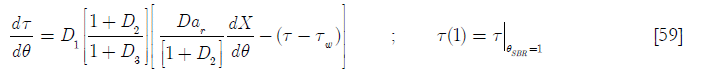
Now it is necessary to derive another equation for Tw from an energy balance on the cooling jacket.
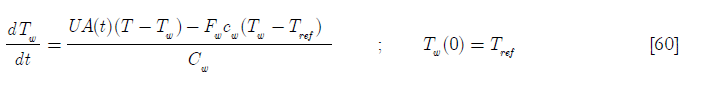
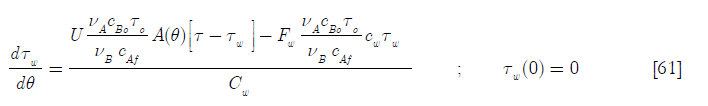
or
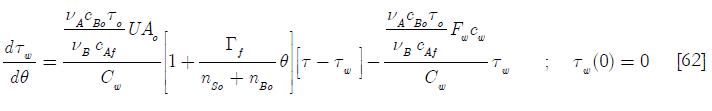
Here again there are some Damköhler type dimensionless groups, namely
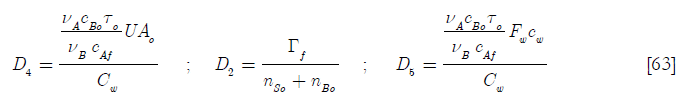
D4 is the heat transfer per unit temperature difference taking into account the initial area available for heat transfer over the heat capacity of the coolant jacket and its contents; D2 the ratio of the molar feed over the initial number of moles of reactants and D5 the ratio of the heat capacity of the feed of reactant A over the heat capacity of the coolant jacket and its contents. Hence
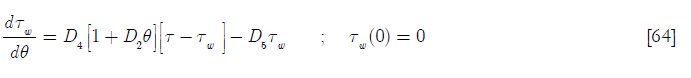
Equations [47], [58] and [64] constitute the nonadiabatic second order type one SBR reactor model. The numeric solution obtained using Mathcad is depicted in Subprogram 3, while the data has been plotted in the figures shown below.
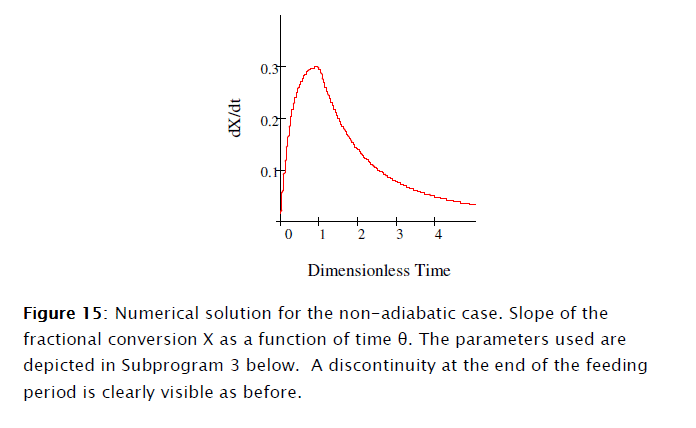
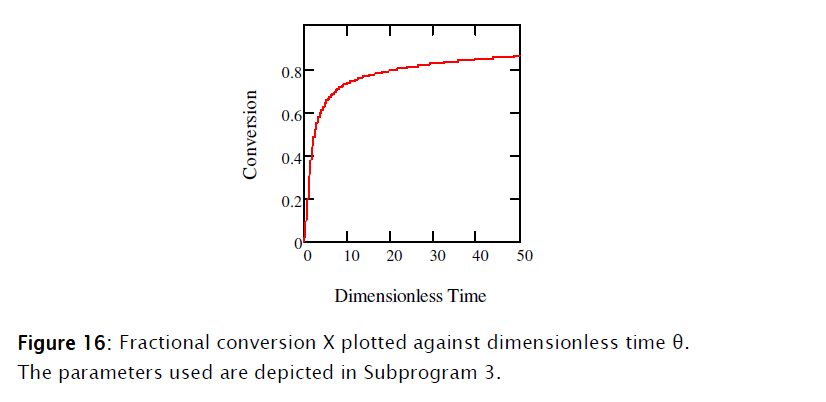
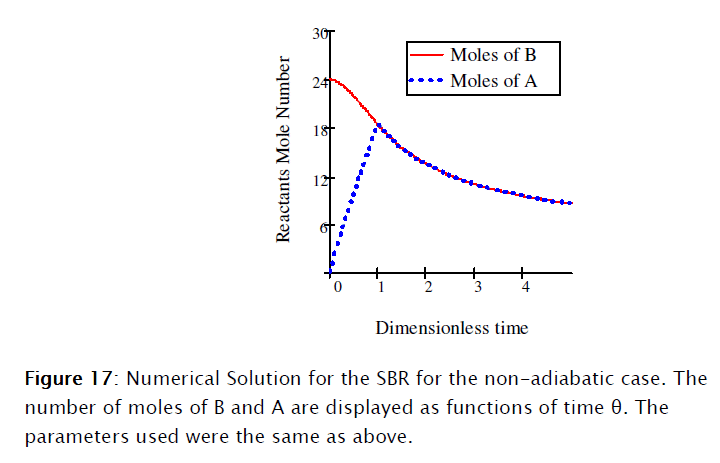
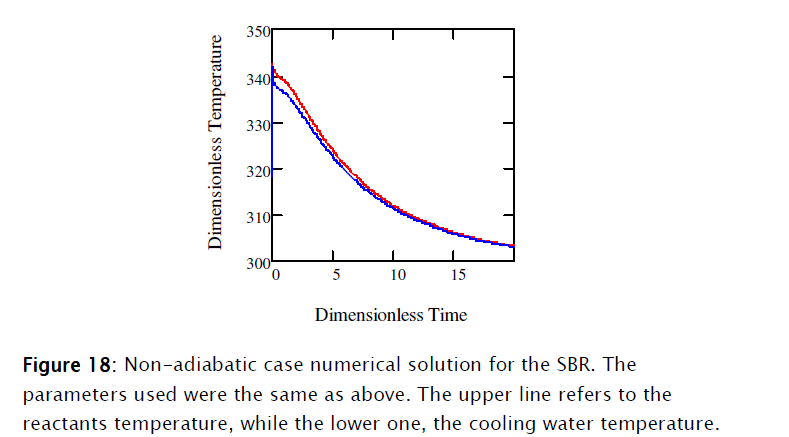
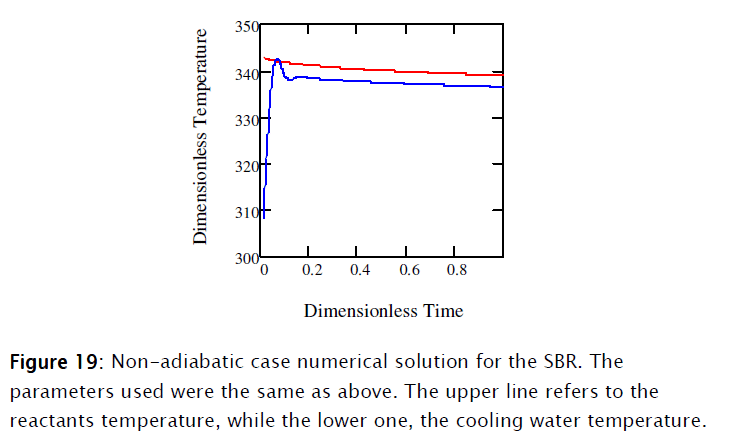

4. Conclusions
A versatile macroscopic model for the SBR reactor has been derived for the isothermal, adiabatic and nonadiabatic cases. A second order elementary reaction was studied. Other kinetics can be studied using the same approach. The model is well suited for its solution utilizing digital computers and modern computer algebra software. The nonadiabatic case is especially interesting since it can be used for modeling nonlinear process control, an area of great interest that is precisely the subject for the next part of this article.
Notation
Variables
]]> ParametersA, B, C, D = chemical species A, B, C, D respectively. A refers also to the area available for heat transfer.
c = specific heat capacity
C = Heat capacity of the reactor contents ]]> F = reactant feed rate
H = Enthalpy
n, N = number of moles
k = chemical reaction rate constant
r = chemical reaction rate
t = time
T = Temperature
U = Internal energy, also the global heat transfer coefficient
V = volume of the chemical reaction media
z = factor de compresión
Greek LettersDa, Daf = dimensionless Damköhler type parameters defined by Equation [48]
Dar = another Damköhler type parameter defined by Equation [57]
D1, D22, D3 = parameters for the energy balance defined by Equation [57]
SubscriptsΔ = incremental operator, when referred to chemical reactions gives the enthalpy or internal energy of reaction
ρ = density of the chemical reaction medium
ε = ratio of the stoichiometric initial concentrations of species B in the reactor and A in the feed
Γ = Feed rate of reactant ]]> ν = stoichiometric coefficient of an elementary chemical reaction.
ϑ = volumetric feed rate
Χ = chemical reaction conversion
Θ = dimensionless time
τ = space time, also a dimensionless temperature
A, B, C, D = species A, B, C, D respectively
o, 0 = refers to a parameter at the beginning of the run
f = refers to a parameter at the end of the run and also to the feed
m = molar, specific property ]]> ref = reference, used with T refers to a reference temperature
S = refers to the solvent
w = refers to the cooling medium
]]>