Patricia Taboada Serrano, Ronanth Zavaleta Mercado
Departamento de Ingeniería Industrial, Universidad Católica Boliviana, La Paz Bolivia
Abstract: Bolivian cocoa beans butter has had problems of sour flavor and smell that inhibited its possibilities of being sold in international markets. An hypothesis following a Strecker type kinetics was postulated for the formation of low molecular weight fatty acids which were deemed responsible for these unwanted properties. Since the formation of these organic acids has probably an origin in the improper handling of the beans by the farmers, and since no alternative source of raw materials was available, a process modification was proposed to deal with these problems. An acid base neutralization reaction using concentrated solutions of potassium carbonate involving several mass transfer steps is proposed as a mechanism for a process developed to remove low molecular fatty acids from cocoa beans butter. The removal of these acids greatly improved the product’s flavor and its physical properties including a narrowing of the band of melting points. The experimental data was well correlated only after the consideration of dead zones in the model proposed. The process modification including a steam jacketed reactor devoted to the neutralization reactor is under way.
Key words: cocoa beans, fatty acids removal, sour flavor
]]> Resumen: La manteca de cacao boliviana ha tenido problemas de sabor amargo y olor que inhibe sus posibilidades de ser vendido en los mercados internacionales. Una hipótesis siguiendo una cinética de tipo Strecker se postuló para la formación de ácidos grasos de bajo peso molecular que se considera responsable de estas propiedades no deseadas. Dado que la formación de estos ácidos orgánicos tiene probablemente un origen en el mal manejo de los granos por parte de los agricultores, y puesto que no hay ninguna fuente alternativa de materia prima disponible, una modificación del proceso fue propuesto para hacer frente a estos problemas. Una base de ácido reacción de neutralización usando soluciones concentradas de carbonato de potasio que implican varios pasos de transferencia de masa se propone como un mecanismo para un proceso desarrollado para eliminar los ácidos moleculares bajos grasos a partir de granos de cacao mantequilla. La eliminación de estos ácidos grasos mejoró en gran medida el sabor del producto y sus propiedades físicas, incluyendo un estrechamiento de la banda de puntos de fusión. Los datos experimentales se correlacionaron bien sólo después de la consideración de zonas muertas en el modelo propuesto. La modificación del proceso que incluye un reactor con camisa de vapor dedicado al reactor de neutralización está en marcha.Palabras clave: granos de cacao, eliminación de los ácidos grasos, el sabor agrio
Certain amounts of large and low molecular weight fatty acids are produced during cacao beans fermentation and drying processes. This quantity depends on conditions such as temperature, drying rate, humidity, oxygen availability and other factors, which have to be controlled carefully during the initial treatment processes [1][2]. Improper practices applied at this stage result in high contents of fatty acids that have strong bitter taste and smell. This mechanism has been studied and involves several steps as postulated by Strecker et al. [2].
Bolivian cacao beans are produced in small quantities by farmers that do not have the technology to control the fermentation and drying processes. Thus, each lot of cacao has its own degree of fermentation, quality and characteristics. Therefore the butter obtained by the chocolate manufacturers is sour and its flavor does not improve during the conventional elaboration process. The influence of the bitter flavor is remarkably harsh on white chocolate, because it acquires a sour flavor that grows worse during the manufacturing process.
The cocoa butter produced from this raw material has an amount of fatty acids that exceeds the allowed levels (1.7 to 1.9 % measured on basis of oleic acid compared to the 0.8 % recommended by Minifie regarding the recommendations European Chocolate Manufacturers [3]). The percentage of free fatty acids present in Bolivian cocoa butter increases if it is calculated on a butyric acid basis, one of the low molecular fatty acids produced during bad practices of fermentation processes. The cocoa butter also presents a lower melting point range (30 to 32 °C compared to 32 to 35 °C of normal cocoa butter [3]), probably due to the presence of low melting point substances (fatty acids).
This paper presents the results of a study about a process modification of fatty acids removal based on the hypothesis that low molecular weight fatty acids intensify the sour flavor.
Some facts had to be taken into account in order to design a process of removal of fatty acids:
· Fatty acids are partially soluble in both, polar and non-polar solvents such as butter or water.
]]> · Although the fatty acids melting point is lower than that of butter, their high solubility in butter and fat disqualifies a phase separation process.· When alkalis (sodium or potassium hydroxide, sodium or potassium carbonate, etc.) are added to fatty acids, organic salts, insoluble in lipids, are produced. These salts are soluble in polar solvents (water).
The food industry made the removal of fatty acids subject of much research and development [4] [5] [6] [6] [7] [8] [35]. Methods such as continuous neutralization with sodium hydroxide, deodorization and others are widespread. However, these methods are suitable for large scale production of fat (plants with productions up to 500 tons per day). The production of the largest chocolate manufacturer in Bolivia does not reach 100 tons per week. Thus, in general, the methods usually applied in industry are not useful for Bolivian chocolate manufacturers because of economies of scale. Therefore, a batch process had to be designed in order to deal with small amounts of cocoa butter production and moderate investment budgets.
The process modification proposed for the removal of fatty acids consists of an heterogeneous reaction of neutralization by means of an alkali.
· The process is carried out in a stirred tank batch reactor where the melted cocoa butter is placed.
· The alkali used is a saturated aqueous solution of potassium carbonate.
· Potassium carbonate is used because of its high solubility in water, that permits the reduction of the amount of solution introduced into the reactor (no more than 1% of the total reaction mass). The use of a solution rather than the pure salt, proved indispensable, suggesting a mechanism involving potassium ions. On the other hand, it is desirable to keep water to a minimum in order to avoid fat or oil hydrolysis [4][6].
· Agitation produces the dispersion of water in cocoa butter.
· Due to the small amount of water in the reactor, fatty acids salts form an insoluble phase.
· During the process, carbon dioxide is produced promoting the fatty acids flocculation and floatation. This phase can be easily removed by skimming. Mounts and Khym [4] reported that fatty acids salts act as coagulants for other particles such as water.
]]> · Water is also removed during the operation.Preliminary laboratory experiments were carried out in order to get acquainted with the behavior of the proposed process and through these initial experiments, some facts presented above were established, leading to a possible reaction mechanism. Detailed experimental work was carried out based on a proposed mechanism.
In order to study the process modification proposed, two sets of experiments were performed. The first one was aimed to studying the physical equilibrium. Potassium carbonate solutions (20 ml) of seven different concentrations were added to 20 g samples of melted butter. The temperature range chosen was 40 °, 50 ° and 60 °C. The samples were placed in a Baxter DX 31 drying oven during 24 hrs. Then, the two phases of each sample were analyzed. The free fatty acids content was determined in the lipoid phase and the free potassium content in the aqueous phase. Only those data from the samples that had zero content of free fatty acids in the lipoid phase were considered in the physical equilibrium study.
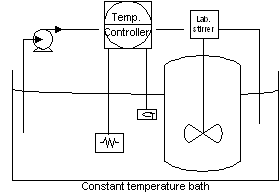
The second set of experiments was aimed to study the reaction kinetics. For this purpose, 400 g of cocoa fat was introduced in a 600 ml Pyrex glass container used as a reactor. This reactor was placed inside a constant temperature water bath 20 minutes before the experiment in order to allow the butter to reach the steady state reaction temperature. Once the reaction temperature was reached, potassium carbonate was introduced into the reactor as a concentrated water solution. The mass of the solution introduced was approximately 1 % of the fat mass. The free fatty acids concentration was determined at different reaction times and at 40 °, 50 ° and 60 °C. Samples of 2 g of fat were taken from the reactor and immediately placed on ice in order to freeze the chemical reaction taking place. An schematic drawing of the equipment used is presented below:

Figura 1: Acid base neutralization reactor.
A Cole-Palmer electric bath temperature controller was used to keep the reactor temperature at the chosen level. The controlled temperature fell in a range of ±1 °C. The controller also included a peristaltic pump to allow the temperature to remain constant. A T-line laboratory stirrer of 1/18 HP was used to keep the reactor content uniform. The variable speed stirrer could be regulated in a range of 75 to 7500 rpm, and had a propeller type stirrer of 5 cm of diameter.
The fatty acids concentration was determined by an analytical technique employed in industrial practice that is based on titration of fatty acids in an alcoholic solution [23]. The alcoholic solution is neutralized with a sodium hydroxide solution. The indicator employed is an alcoholic phenolphthalein solution. It is important to carefully register the strength of the violet color obtained in the alcoholic solution, because it must be reproduced during the titration of the fatty acids. After the alcoholic solution had been neutralized, 5 g of fat are dissolved in 50 ml of the neutralized alcohol at 50 °C. After the fat melts down, a colloidal solution is obtained by strong stirring. A measured volume of a sodium hydroxide solution is added to the fat solution using phenolphthalein as indicator until the desired violet color is obtained. Usually, in industrial practice, sodium hydroxide solutions of 0.1 N are employed, but in this work solutions of 0.01 N were used in order to increase the method’s sensitivity. The method was repeatedly tested and the statistical inference analysis was carried out.
Potassium concentration is indirectly determined by a spectrophotometric analytical technique based on the quantification of the excess of sodium cobalt nitrite salt [21]. The aqueous phase containing potassium is centrifuged during five minutes in order to separate the fatty acids salts formed and determine the amount of free potassium. The sample size depends on the initial concentration of potassium carbonate used for the equilibrium test. A volume of 10 ml, 5 ml, 2 ml or 1 ml of sample is separated for analysis. Each sample is diluted to 20 ml. Then 0.5 g of sodium cobalt nitrite salt is added along with 5 ml of water. The solution is then placed to rest for an hour to enable the insoluble potassium salt to precipitate. A sample of 10 ml is taken from the liquid phase and it is centrifuged during 5 min, in order to separate any fatty acid potassium salt that could remain suspended. From the centrifuged liquid, 1 ml is sampled and it is diluted to 50 ml with distilled water. The absorbance of this solution is finally measured in the spectrophotometer. The lecture of absorbance indicates the amount of sodium cobalt nitrite that did not react with potassium. By means of a material balance, the initial concentration of potassium present in the sample is thus obtained.
Simultaneously, a set of three laboratory experiments were performed in order to establish whether the removal of fatty acids from cocoa butter improved its flavour, diminished its bitterness and improved the overall quality of the white chocolate. Thus, three batches of white chocolate were prepared using butter free of fatty acids. The cocoa butter obtained after the neutralization process did not present the sour flavour and smell previously detected on non-treated products and furthermore, a sweet aromatic flavour with no signs of bitterness was characteristic of the white chocolate prepared. This product was considered of high quality by the costumer, a chocolate manufacturer. This initial experimental work gave strong indication that the removal of fatty acids was essential to the production of high quality white chocolate.
· Ionic potassium transfer from the aqueous phase to the interface.
· Interface chemical reaction.
· Remaining ionic potassium transfer from the interface to the lipoid phase.
· Simultaneous chemical reaction in the lipoid phase.
· Fatty acids transfer from the lipoid phase to the interface.
A model based on the proposed mechanism was set up including mass balances for key components and chemical reaction taking place at the interface and at the lipoid phase. As a first approach a uniform macroscopic model was accepted.
The uniform model was developed on the basis of the following assumptions:
· Each phase is uniform.
· The interface does not accumulate material.
]]> · The process is controlled by mass transfer.· Equilibrium is established at the interface.
The model includes five partial material balances:
· Two balances for ionic potassium in both phases and one balance in the interface.
· One balance for fatty acids in the lipoid phase and another at the interface.
· An empirical mathematical equation for the ionic potassium equilibrium (based on analysis of experimental data).
Five equations are obtained (the equilibrium relation has to be included) in terms of dimensionless variables:
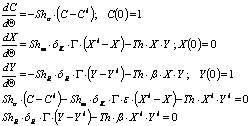
This model has two controlling parameters, the Sherwood number and Th, a number that relates the kinetic constant with some physical properties as described in the nomenclature. All the other parameters are also defined in the nomenclature and relate aqueous and cocoa butter physical properties. Thus, the experimental design includes mainly these two numbers (Sh and Th).
The Sherwood number in stirred tanks is a function of the Reynolds, Froude and Schmidt numbers. Taking into account the size of the laboratory reactor (400 ml), the influence of gravitation forces (represented in Fr) was disregarded in favour of the inertial forces (Re) and the properties of the fluids (Sc). Therefore, the Re and Sc numbers were considered in the experimental design.
]]> The Th number is basically a function of temperature at constant pressure.The model was adjusted to the experimental data by means of a restricted non-linear minimization algorithm (the complex method of Box) driving a set of sub-routines for solving the set of differential equations based on the Runge-Kutta-Fehlberg algorithm. The objective function was the accumulative absolute error.
The figures 1, 2 and 3 show equilibrium experimental data and curve fitting.
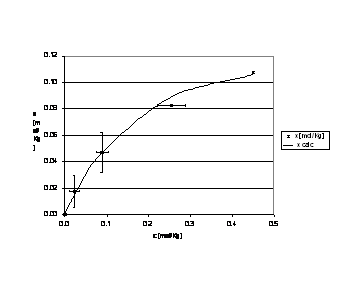
Figura 2: Ionic potassium equilibrium at 40 ° C.
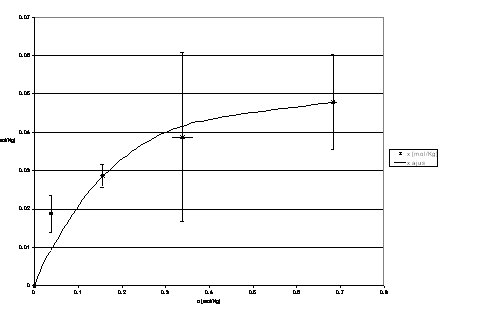
Figura 3: Ionic potassium equilibrium at 50 ° C.

Figura 4: Ionic potassium equilibrium at 60 ° C.
The empirical relation obtained is:
]]>The parameters for each temperature considered in the equilibrium analysis are schematized as follows:
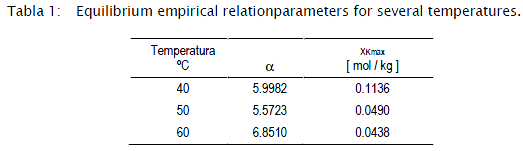
Figures 4, 5, 6, 7 and 8 present the experimental dynamic behaviour of the process together with that of the proposed model. As can be seen the product Reb has been used as an adjusting parameter.
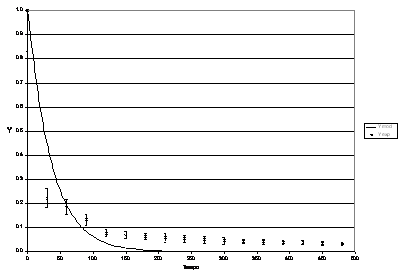
Figura 5: Process dynamic behaviour for Re b = 106.

Figura 6: Process dynamic behaviour for Re b = 152.
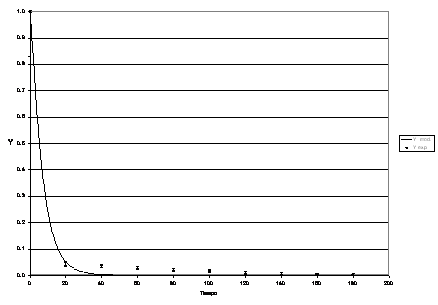
Figura 7: Process dynamic behaviour for Re b = 213.
]]>
Figura 8: Process dynamic behaviour for Re b = 437.
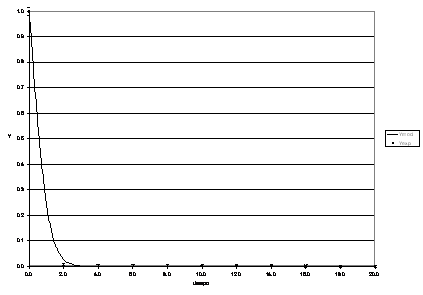
Figura 9: Process dynamic behaviour for Re b = 640.
The analysis of the behaviour of the experimental and the comparison with those predicted by the uniform model results lead to state some facts:
· The model proposed does not correlate the experimental data. A pronounced tailing is present in the experimental data that the model cannot predict. Also, there is a characteristic behaviour that strongly deviates from the data (Figure 4).
· This behaviour is typical of reactors that show segregation, and therefore a model including some degree of deviation of ideal behaviour had to be considered.
In order to consider the above, the following partially segregated model was proposed based on the series of CSTR’s model:
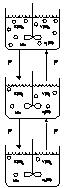
Figura 10: Partially segregated model.
]]> This model is described by the following equations:First stirred tank reactor (1st tank)
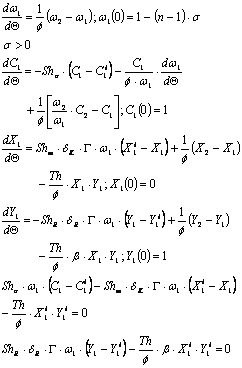
Intermediate stirred tank reactor (j tank)

Last stirred tank reactor (n tank):
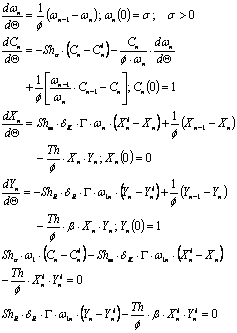
Since a concentration profile is obtained for each reactor, a mean concentration is obtained applying the mean value theorem. The results for a typical run (Re b = 106): are plotted in Figure 10.

Figura 11:Process dynamic behaviour at Re b = 106.
]]> As can be seen this model fails also to correlate the experimental data, even though the absolute error is diminished somewhat. The persistence of the tailing effect suggested that dead zones could be present. Therefore, a model including dead zones was set up as an attempt to correlate the data.This model is described below and is a derivation of the CSTR’s in series model:
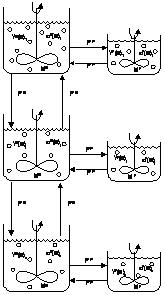
Figura 12: Micro-mixture with segregation and stanched zones model representation.
The corresponding mass balances to the following sets of equations in terms of dimensionless variables:
First stirred tank reactor (1):

For any intermediate stirred tank reactor (j tank):
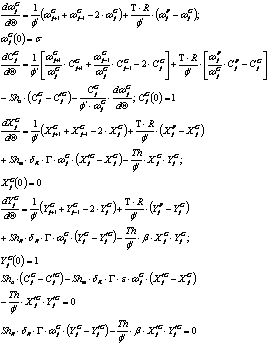
Last stirred reactor tank (n tank):
]]>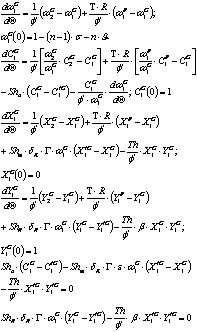
And for any dead zone:
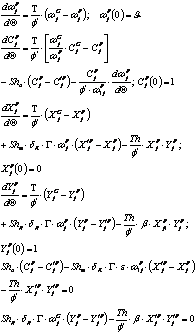
As before a mean concentration was obtained for the whole set by application of the mean value theorem.
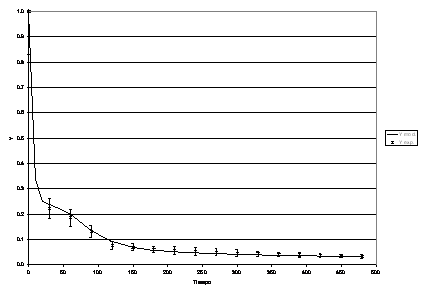
Figura 13: Process dynamic behaviour for Re b = 106.

Figura 14: Process dynamic behavior for Re b = 152.
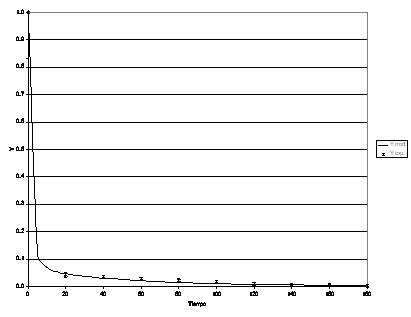
Figura 15: Process dynamic behavior for Re b = 213.
]]>
Figura 16: Process dynamic behaviour for Re b = 437.
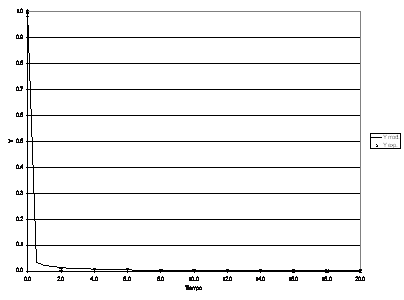
Figura 17: Process dynamic behaviour for Re b = 640.
The model was fitted to the experimental data again by running the restrictive non-linear algorithm driving the Runge-Kutta-Fehlberg subroutines. In this case, since no analytical solution was available for the set of equations, the minimization process included the integration of the mathematical model for each set of parameters tested. In this case, in addition to the Sh in both phases, Th, f, some other parameters were taken into account, such as R, T, the mass ratio of the dead zone reactor to the main reactor and the space time ratio respectively. (See Table 2).
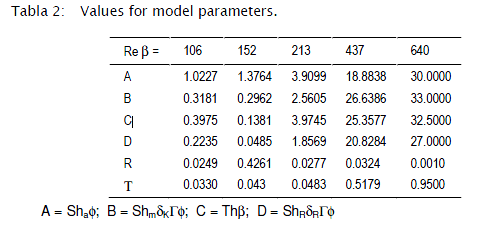
The values of the parameters that best fit the model to the experimental data are presented in Table 2.
The proposed model fits the experimental data and therefore some conclusions can be drawn up:
· The proposed basic mechanism describes adequately the process.
· The process is strongly affected by mixing and the dynamic behaviour could only be explained by the inclusion of dead zones.
]]> · The determinant parameter is Reb suggesting that, in addition to recognizing the effect of stirring in the process as a means of attaining a higher degree of mixing, the amount of ionic potassium available in the lipoid phase is also of importance, implying that the mechanism is dependent on mass transfer of potassium ions from the aqueous phase into the lipoid phase.· The experimental data were well correlated using three CSTR’s in series each provided the corresponding dead zone (side reactor) for selected values of Reb and mass ratios of main and subsidiary reactor (parameters R and T).
· The b parameter is strongly dependent on temperature in the range of interest.
· It appears that the experimental data validates the model and therefore a suitable model has been set up for further work regarding scale up and optimization of process oriented at eliminating the low molecular weight fatty acids. In addition to this, it has been demonstrated that the elimination of these acids strongly enhances the flavour and smell of cocoa butter and in general the quality of white chocolate.
![]() Water concentration [Kg water / Kg butter]
Water concentration [Kg water / Kg butter]
![]() Ionic potassium concentration in aqueous phase [mol K+ / Kg water]
Ionic potassium concentration in aqueous phase [mol K+ / Kg water]
![]() Ionic potassium concentration in lipoid phase [mol K+ / Kg butter]
Ionic potassium concentration in lipoid phase [mol K+ / Kg butter]
![]() Free fatty acids concentration in lipoid phase [mol fat. a. / Kg water]
Free fatty acids concentration in lipoid phase [mol fat. a. / Kg water]
![]() Water concentration introduced into the experimental reactor tank [Kg water/ Kg butter]
Water concentration introduced into the experimental reactor tank [Kg water/ Kg butter]
![]() Ionic potassium concentration of the aqueous solution introduced into the experimental reactor tank [mol K+ / Kg water]
Ionic potassium concentration of the aqueous solution introduced into the experimental reactor tank [mol K+ / Kg water]
![]() Initial concentration of fatty acids inside the laboratory reactor tank [mol fat. a. / Kg butter]
Initial concentration of fatty acids inside the laboratory reactor tank [mol fat. a. / Kg butter]
![]() Time [s]
Time [s]
![]() Dimensionless H2O concentration [0,1]
Dimensionless H2O concentration [0,1]
![]() Dimensionless ionic K concentration in aqueous phase [0,1]
Dimensionless ionic K concentration in aqueous phase [0,1]
![]() Dimensionless ionic K concentration in lipoid phase [0,1]
Dimensionless ionic K concentration in lipoid phase [0,1]
![]() Dimensionless fatty acids concentration in lipoid phase [0,1]
Dimensionless fatty acids concentration in lipoid phase [0,1]
![]() Dimensionless time
Dimensionless time
![]() paddle diameter [m]
paddle diameter [m]
![]() Interfacial area [m2/Kg water]
Interfacial area [m2/Kg water]
![]() Global mass transfer coefficient of ionic potassium in lipoid phase
Global mass transfer coefficient of ionic potassium in lipoid phase ![]()
![]() Global mass transfer coefficient of fatty acids in lipoid phase
Global mass transfer coefficient of fatty acids in lipoid phase ![]()
![]() Kinetic constant
Kinetic constant ![]()
ra º water density [Kg / m3]
rm º butter density [Kg / m3]
DKa º Diffusion coefficient of ionic potassium in water [m2 / s]
DKm º Diffusion coefficient of ionic potassium in cocoa butter [m2 / s]
DR º Diffusion coefficient of fatty acids in cocoa butter [m2 / s]
M º Mass of butter in the reactor tank [Kg]
]]> F º Mass flux between stirred tanks [kg / s]![]() Number of stirred tanks and stanched zones.
Number of stirred tanks and stanched zones.
![]() Residence time [1/s]
Residence time [1/s]
![]() Equilibrium constant.
Equilibrium constant.
R = Ratio of butter mass in a stanched zone and butter mass in a stirred tank.
T = Ratio of the residence time of a stanched zone and the residence time of a stirred tank.
![]()
![]()
![]()
![]()
![]()
![]()
Re = ![]()
![]()
![]()
![]()
![]()
Dimensionless time:
![]()
![]() Interface
Interface
![]() Segregation Number stanched zone stirred tank.
Segregation Number stanched zone stirred tank.
G = Stirred tank in the third model.
P = Stanched zone in the third model.
[1] URGUHART, D.H., Cacao La Habana, Editorial Revolucionaria – Instituto del Libro de la Habana, 322 p., 1963. [ Links ]
[2] BECKETT, S. T., Fabricación y Utilización Industrial del Chocolate, Editorial Acribia S.A., 422 p., 1988. [ Links ]
[3] MENIFIE, Bernard W., Chocolate, Cocoa and Confectionary: Science and Technology, Editorial Putnan Russ, 625 p., 1970. [ Links ]
[4] ASOCIACION AMERICANA DE SOYA, Manual de Procesamiento y Utilización de Aceite de Soya, Editorial Propia, 400 p., 1988.
[5] OSTROVSKI, A. Y COLABORADORES, Fundamentos de la Tecnología de los Productos Alimenticios, Editorial Mir, 322 p., 1980.
[6] KIRSCHENBAUER, H. G., Grasas y Aceites: Química y Tecnología, Compañía Editorial Continental S.A., 310 p., 1964.
[7] ANDERSEN, A. J. C., Refinación de Aceites Comestibles.
[8] BERNARDINI, E. , Tecnología de Aceites y Grasas.
[9] MORRISON, R. T., BOYD, R. N., Química Orgánica, Editorial Addison Wesley Iberoamericana, 1990.
[10] LEVENSPIEL, Octave, Ingeniería de las Reacciones Químicas, Editorial Reverté, 637 p., 1976.
[11] FROMENT, Gilbert F., BISCHOFF, Keneth B., Chemical Reactor Analysis and Design, Editorial John Wiley and Sons, 664 p., 1990.
[12] SMITH, J. M., Ingeniería de la Cinética Química, Compañía Editorial Continental, 774 p., 1986.
[13] CHEMICAL ENGINEERING JOURNAL, Chemical Engineering: Liquid Agitation, Re-impresión de los artículos de los años 1975 – 1976, 1996.
]]> [14] ALPER, Erdogan, “Mass Transfer with Chemical Reaction in Multiphase Systems”, en Vol I: Two Phase Systems, Editorial Martins Nijhoff Publishers, 1983, págs. 577 – 635.[15] CALO, Joseph M., RODRIGUEZ, Alicio R., SWEED, Norman H., “Multiphase Chemical Reactors” en Vol I: Reactor Fundamentals, Ecitorial Martins Nihoff and Noordhoff, 1981, págs. 133 – 223.
[16] SHAH, Kamlesh J., VENKATESAN, T. K., “Aqueous Isopropyl Alcohol for Extraction of Free Fatty Acids from Oils”, Journal of the American Oil Chemists Society, Boston, Vol. 66, No 6, 1989.
[17] GORE, W. L., Métodos Estadísticos para Experimentación Química y Tecnología, Editorial Tecnos S.A., 174 p., 1990.
[18] MILLER, J. C., MILLER, J. N., Estadística para Química Analítica, Editorial Addison – Wesley Iberoamericana, 211 p., 1993.
[19] VOGEL, Arthur, Química Analítica Cuantitativa, Editorial Kapelusz, 1960, págs. 537 – 680.
[20] COMISION VENEZOLANA DE NORMAS INDUSTRIALES, Cacao y Preparaciones de Cacao, Compendio de Norma Venezolana, Caracas. (Normas proporcionadas y utilizadas por IBNORCA, sin fecha de impresión).
[21] BERL, LUNGE, D´ANS, Métodos de Análisis Químico Industrial, Tomo II, 2da Parte, Editorial Labor S.A., 1945, págs. 895-907.
[22] RODIER, J., Análisis de las Aguas, Editorial Omega S.A., 1989, págs. 228-230.
[23] ASOCIACION AMERICANA DE PRODUCTORES DE OLEAGINOSAS Y ACEITE DE SOYA, Analisis de grasas y aceites, 623 p., 1980.
]]> [24] SANDLER, S. I., Chemical and Engineering Thermodynamics, 2da Ed., New York, Wiley Series for Chemical Engineering, 1989.[25] BALZHISER, Richard E., SAMUELS, Michael R., ELIASSEN, John D., Termodinámica Química para Ingenieros, Editorial Prentice Hall Internacional, 733 p., 1974.
[26] CHAISERI, Siree, DIMICK, Paul S. “Lipid Hardness Characteristics of Cocoa Butters from Different Geographic Regions”, Journal of the American Oil Chemists Society, Vol. 66, No 11, 1989, págs. 1771-1776.
[27] PERRY, Robert H. & Green, Don, Perry´s Chemical Engineers´ Handbook, 6ta Ed., Editorial McGraw Hill, 1984.
[28] BIRD, R. B., STEWART, W. E., LIGHTFOOT, E. N., Fenómenos de Transporte, Editorial Reverté, 1982.
[29] FRANKS, Rober G. E., Modeling and Simulation in Chemical Engineering, Editorial John Wiley and Sons, 411 p., 1972.
[30] JOURNAL BOLIVIANO DE CIENCIAS, Vol. I, No 1, Editorial UNIVALLE, 1998.
[31] TABOADA, Patricia, Informe de Práctica Industrial, Carrera de Ingeniería Química, Universidad Mayor de San Andrés, 1995.
[32] DUHNE, Carlos, “Viscosity – temperature correlations for liquids”, Chemical Engineering Journal (Reprint), Editorial McGraw-Hill, julio de 1979.
[33] DIMICK, Paul S., MANNING, Douglas M., “Thermal and Compositional Properties of Cocoa Butter”, Journal of the American Oil Chemists Society, Vol. 64, No 12, 1987, págs. 1663 – 1667.
]]> [34] BERGMAN, T. J., NAUMAN, E. B., “The Flash Devolatization of Cocoa Butter”, Journal of the American Oil Chemists Society, Vol. 63, No 11, 1976, págs. 1469 – 1473.[35] CERVO A. L., BERVIAN, P. A., Metodología Científica, Editorial McGraw – Hill, Sao Paulo, 1980.
]]>