Diversidade e prevalência das mutações de resistência genotípica aos antirretrovirais entre crianças infectadas pelo HIV-1
Diversity and prevalence of antiretroviral genotypic resistance mutations among HIV-1-infected children
Flávia J. Almeida1, Eitan N. Berezin2, Rosângela Rodrigues3, Marco A. P. Sáfadi4, Mariana V. Arnoni5, Cristina Oliveira6, Luis F. M. Brígido7
1. Médica assistente, Serviço de Infectologia Pediátrica, Santa Casa de São Paulo, São Paulo, SP.
2. Médico responsável, Serviço de Infectologia Pediátrica, Santa Casa de São Paulo, São Paulo, SP. ]]>
Como citar este artigo: Almeida FJ, Berezin EN, Rodrigues R, Sáfadi MA, Arnoni MV, Oliveira C, et al. Diversity and prevalence of antiretroviral genotypic resistance mutations among HIV-1-infected children. J Pediatr (Rio J). 2009;85(2):104-109.
Artigo submetido em 08.09.08, aceito em 21.01.09.
doi:10.2223/JPED.1877
(1) Artículo original de Brasil, publicado en el Jornal de Pediatría (Rio J.) 2009; 85: 104-9 y que fue seleccionado para su reproducción en la XV Reunión de Editores de Revistas Pediátricas del Cono Sur. Paraguay 2010
]]>
Abstract:
Objective: To evaluate genotyping and subtyping in antiretroviral (ARV) naïve and experienced children, as well as drug resistance profiles through genotyping in these children.
Methods: This retrospective study assessed ARV-naïve HIV children and HIV children failing highly active antiretroviral treatment (HAART) followed up at Santa Casa de São Paulo. Genotypingwas performed using purified polymerase chain reaction (PCR) products from retrotranscribed RNA using Kit Viroseq HIV-1 Genotyping System 2.0 or nested PCR in-house. Sequencing was performed using automatic equipment (ABI 3100). ARV resistance mutations were analyzed in the Stanford HIV Drug Resistance Database and subtypingwas performed at the National Center for Biotechnology Information (NCBI), using SimPlot analysis, together with phylogenetic analysis.
Results: No primary ARV resistance mutation was detected in the 24 ARV-naïve children, although there were mutations that may contribute to resistance to nucleoside analogue reverse transcriptase inhibitors (NRTI) (12.5%) and to protease inhibitors (PI) (95.8%). For the 23 children failing HAART, we found ARV resistance mutations to NRTI in 95.6% and to non-nucleoside analogue reverse transcriptase inhibitors (NNRTI) in 60.8%. For PI, we found ARV resistance mutations in 95.7%, 47.8% of which had only polymorfisms. In the subtyping analyses, 78.3% of the sequences clustered in HIV-1 subtype B, 4.3% in C, 13% in F and 4.4% in recombinant forms.
Conclusion: Our results show low rates of primary resistance in ARV-naïve children and high rates of resistance in children failing ARV treatment, which is compatible with ARV use in these patients.
Key words:
Rev Soc Bol Ped 2011; 50 (3): 186-93: HIV, resistance, genotype, child, antiretroviral therapy.
Resumo
]]> Objetivo: Avaliar a genotipagem e subtipagem em crianças experimentadas e virgens de tratamento, assimcomoperfis de resistência a medicamentos através da genotipagem nessas crianças.Métodos: Estudo retrospectivo de crianças HIV positivas virgens de tratamento e HIV positivas que não responderam ao tratamento pela terapia antirretroviral altamente ativa (HAART), acompanhadas na Santa Casa de São Paulo (SP). A genotipagem foi realizada com produtos purificados de reação em cadeia da polimerase (PCR) de RNA retrotranscrito, utilizando-se o kit comercial Viroseq HIV-1 Genotyping System 2.0 ou a técnica de nested PCR in-house. O sequenciamento foi realizado com equipamento automático (ABI 3100). As mutações de resistência antirretroviral (ARV) foram analisadas no Stanford HIV Drug Resistance Database e a subtipagem realizada no U.S. National Center for Biotechnology Information (NCBI), utilizando-se o programa de análises SimPlot, juntamente com a análise filogenética.
Resultados: Não foi detectada nenhuma mutação de resistência primária ARV nas 24 crianças virgens de tratamento, embora tenham ocorrido mutações que podem contribuir para a resistência aos inibidores da transcriptase reversa análogos de nucleosídeos (ITRN) (12,5%) e aos inibidores da protease (IP) (95,8%). Para as 23 crianças que não responderam à HAART, foram encontradas mutações de resistência ARV aos ITR Nem 95,6% e aos inibidores da transcriptase reversa não-análogos de nucleosídeos (ITRNN) em 60,8%. Para os IP, foram observadas mutações de resistência ARV em 95,7%, 47,8% das quais apresentavam apenas polimorfismos. Nas análises de subtipagem,78,3%das sequências agruparam-se no subtipo B do HIV-1, 4,3% no C, 13% no F e 4,4% em formas recombinantes.
Conclusões: Nossos resultados mostrambaixas taxas de resistência primária em crianças virgens de tratamento e altas taxas de resistência emcrianças que não responderamao tratamento ARV, o que é compatível com o uso ARV nesses pacientes.
Palabras Claves:
Rev Soc Bol Ped 2011; 50 (3): 186-93: HIV, resistência, genótipo, criança, terapia antirretroviral.
Introdução
A introdução da terapia antirretroviral altamente ativa (highly active antiretroviral treatment, HAART) tem reduzido as taxas de morbimortalidade em crianças infectadas pelo HIV-1. No Brasil, a terapia antirretroviral (ARV), assim como o acesso a testes de monitoramento como a contagem de células TCD4, carga viral e genotipagem, são oferecidos gratuitamente pelo governo a todos os pacientes infectados pelo HIV-11.
]]> O surgimento de vírus resistentes a medicamentos pode limitar a eficácia do tratamento ARV. Apesar do crescente número de agentes antirretrovirais disponíveis, as opções para as crianças infectadas pelo HIV-1 que não respondem à terapia ARV permanecem limitadas, especialmente para os inibidores da transcriptase reversa análogos de nucleosídeos (ITRN), que eram os antirretrovirais de primeira linha disponíveis e são os mais usados. Embora tenham sido demonstradas taxas elevadas de resistência ARV em crianças que não respondem à terapia ARV2-5, ainda são limitadas as informações sobre resistência a medicamentos na população pediátrica, assim como sobre o impacto dos testes de resistência.Nos últimos 10 anos, a prevalência de resistência primária ao HIV tem variado de 0 a 25%, com taxas mais elevadas e crescentesempaíses com acesso à terapia ARV6-12. No Brasil, apesar da terapia ARV ser gratuita, nos últimos 10 anos, ainda observamos baixas taxas de resistência primária13-19, mas não há informações disponíveis sobre as crianças.
O papel da diversidade viral na transmissão e resposta a tratamento permanece não esclarecido. O HIV-1 pode ser classificado em três grupos: M, O e N. O grupo M é responsável pela pandemia e, com base na distância genética, pode ser classificado em 11 subtipos e 37 formas recombinantes circulantes (circulating recombinant forms, CRF). No Brasil, predomina a infecção por HIV-1 do subtipo B, com a cocirculação do subtipo F como uma variante menor; com exceção da Região Sul, onde prevalece o subtipo C. Esse cenário favorece a recombinação intersubtipo e diferentes mosaicos, incluindo as CRF BF e BC, têm sido descritos.
Este estudo tem por objetivo comparar a genotipagem e subtipagem do HIV-1 em crianças experimentadas e virgens de tratamento e comparar os perfis de resistência aos medicamentos usados através da genotipagem nessas crianças.
Métodos
A Santa Casa é um hospital de referência, localizado na área central da cidade de São Paulo (SP), com 150 leitos pediátricos. Em seu ambulatório, em torno de 80 crianças e adolescentes infectados pelo HIV-1 são acompanhados. O protocolo de estudo foi aprovado pelo Comitê de Ética e Pesquisa da Santa Casa e do Instituto Adolfo Lutz. Todos pacientes assinaramumtermo de consentimento livre e esclarecido.
O estudo foi conduzido entre novembro de 2000 e março de 2004. Foram selecionadas crianças infectadas pelo HIV-1 virgens de tratamento e crianças previamente tratadas (experimentadas) que não responderam à terapia ARV (falha virológica).
As fichas dos pacientes foram examinadas para coleta de dados demográficos, contagem de células CD4, carga viral, genotipagem e histórico ARV.
O grupo virgem de tratamento ARV era composto por crianças de 0 a 16 anos no momento do diagnóstico de infecção pelo HIV. O grupo experimentado em tratamento ARV era composto por crianças de 0 a 16 anos em HAART (definida como, pelo menos, 3 ARV) por ao menos 3 meses e que apresentavam falha virológica (definida como uma redução < 1 log10 após 12 semanas do início da HAART ou um aumento > 0,5 log10).
]]> Provas laboratoriais- Quantificação do RNA do HIV-1: a carga viral foi medida utilizando-se o teste COBAS Amplicor HIV-1 Monitor Test 1.5, com uma quantificação de 400 cópias/mL.
- Genotipagem e subtipagem do HIV-1: a genotipagem foi realizada utilizando-se produtos purificados de reação em cadeia da polimerase (polymerase chain reaction, PCR) de RNA retrotranscrito, utilizando-se o kit comercial Viroseq HIV-1 Genotyping System 2.0 ou a técnica de nested PCR in-house20. O sequenciamento foi realizado com equipamento automático (ABI 3100). As mutações de resistência ARV foram analisadas no banco de dados de resistência do HIV a medicamentos da Universidade de Stanford (Stanford HIV Drug Resistance Database). A subtipagem foi realizada no Centro Nacional de Informação em Biotecnologia dos Estados Unidos (National Center for Biotechnology Information, NCBI), utilizando-se o programa de análises SimPlot, juntamente com a análise filogenética. Análises moleculares mais detalhadas de partes das amostras foram descritas em outro estudo21.
- Análise das mutações de resistência aos medicamentos: mutações da transcriptase reversa (TR) associadas à resistência foramidentificadas de acordocoma Sociedade Internacional de AIDS (International AIDS Society, IAS)22 e o Stanford HIV Drug Resistance Database23.
Resultados
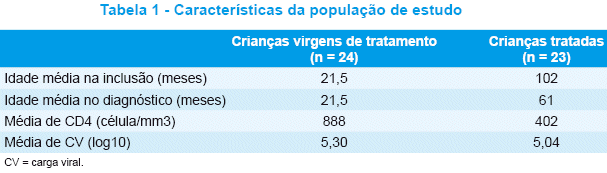
Nossa amostra total foi composta por 47 crianças, das quais 24 eram virgens de tratamento e 23 não responderam à terapia ARV. Todos os pacientes adquiriram o HIV por transmissão vertical. A Tabela 1 apresenta as características da população de estudo.

No grupo de crianças virgens de tratamento, todas as amostras foram obtidas na confirmação do diagnóstico. Os 24 pacientes eram filhos de mães não tratadas durante a gravidez. Apenas duas receberam AZT profilático nas primeiras 6 semanas de vida.
]]> No grupo que não respondeu à terapia ARV, a duração média do tratamento ARV foi de 60 meses (3-120) e o número médio de tratamentos ARV foi quatro (1-10). Sete pacientes receberam monoterapia, e 10 receberam terapia dupla anteriormente. Os seis restantes iniciaram tratamento pela HAART.No momento da genotipagem, todos os pacientes, exceto dois, estavam em tratamento pela HAART. Dezessete pacientes já haviam sido expostos aos inibidores da transcriptase reversa não-análogos de nucleosídeos (ITRNN) e 16 aos inibidores da protease (IP).
Todos os pacientes haviam recebido ou estavam recebendo AZT; 91,3%, DDI; 82,6%, 3TC e D4T; 47,8%, Efavirenz; 34,7%, Nevirapina; 54,1%, Ritonavir; 60,8%, Nelfinavir; 8,6%, Amprenavir; 4,3%, Indinavir, Saquinavir e Lopinavir. Os esquemas ARV observados no momento da coleta da amostra foram: esquema duplo de ITRN (AZT+DDI) em 8,6% das crianças, 2 ITRN + 1 ITRNN em 30,4%, 1 ITRN + 1 ITRNN + 1 IP em 17,3%, 2 ITRN + 1 IP em 43,4%.
No grupo de virgens de tratamento, não foram observadas mutações de resistência principal em nenhum dos tratamentos ARV, mas foram encontradas mutações secundárias que contribuem para a resistência aos ITRN (12,5%) e IP (95,8%). As mutações de resistência mais frequentes foram L10I (16,7%), K20R (12,5%), M36I (33%), L63P (37,5%), A71T (8,3%), V77I (25%), V82I (4%), I93L (12,5%), V118I (8,3%), K219N (4,1%).
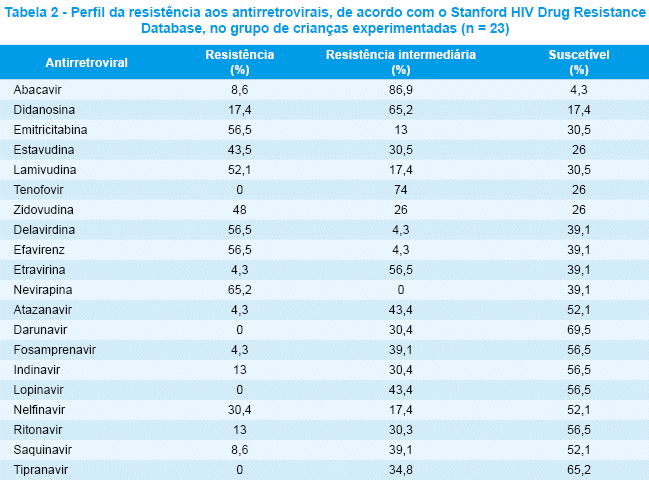
No grupo de crianças experimentadas, todas as amostras (95,6%), exceto uma, tiverammutações que conferem resistência aos ITRN e 14 amostras (60,8%) aos ITRNN. As mutações mais comuns aos ITRN foram T215 (69,6%), M184 (56,5%), D67 (47,8%), M41 (43,5%), K219 (34,8%); e aos ITRNN foram K103 (39,1%) e Y181 (17,4%). Encontramos mutação por excisão de nucleotídeos (nucleotide excision mutation, NEM) em 17 pacientes (73,9%), com uma média de três mutações/paciente (0-5). Na protease, 95,7% tiveram mutações, 47,8% das quais apresentaram apenas polimorfismos nos códons 10, 20, 36, 63, 71, 77, 93 e 47,8% tiveram mutações primárias que conferem resistência aos IP. As mutaçõesmais comuns foram V82, I54, L90 (21,7%), M46 (17,4%). A Tabela 2 mostra o perfil da resistência ARV de acordo com o Stanford HIV Drug Resistance Database.
Nas análises de subtipagem, 78,3% das sequências agruparam-se no subtipo B do HIV-1, 4,3% no subtipo C,13% no F e 4,4% nos mosaicos BF.
Discussão
]]> No grupo de crianças virgens de tratamento não foram encontradas mutações que confiram resistência primária aos ITRN, ITRNN ou IP, mas apenas mutações secundárias que contribuem para a resistência. No gene TR, dois pacientes tiveram V118I e outro teve K219N. Este último tinha recebido AZT profilático nos primeiros 45 dias de vida, o que pode explicar a presença dessa mutação.Na protease,umpaciente, agrupado como B, teve V82I; essa mutação é mais freqüente em subtipos não-B (1%), conferindo baixa resistência aos IP24-28.Os estudos que avaliaram a resistência primária ao HIV demonstraram uma prevalência que varia de 0 a 25%6-12. No Brasil, apesar da terapia ARV ser gratuita, nos últimos 10 anos, ainda observamos baixas taxas de resistência primária, com exceção de algumas cidades do país, como Santos (SP)13-19.
Nossos dados não mostraram taxas de resistência elevadas entre os pacientes virgens de tratamento.Um aspecto que pode ter contribuído para isso é o fato de que todos os pacientes eram filhos de mães não tratadas, que não haviam recebido o diagnóstico da infecção pelo HIV antes da gravidez.
No grupo que não respondeu à terapia ARV, observamos uma taxa elevada de resistência genotípica, com um padrão variável de mutações. Outros estudos brasileiros que também avaliaram crianças que não responderam à terapia ARV encontraram taxas similares2-4,19.
Considerando os ITRN, as mutações mais frequentes foram T215 (69,6%), M184 (56,5%), D67 (47,8%), M41 (43,5%), K219 (34,8%). É comum encontrar quatro, cinco e até seis NEM em pacientes que não respondem a muitos tratamentos ARV. Esse fato reduz a susceptibilidade a muitos ITRN, principalmente ao AZT, D4T e Abacavir; mas também ao DDI e Tenofovir22,23.
O AZT é o medicamento usado com mais frequência na população pediátrica. Todos os pacientes foram expostos ao AZT e 74% deles tiveram mutações associadas à resistência.
Outros estudos com crianças que não responderam à terapia ARV apresentaram taxas similares de resistência ao AZT (64-85%)22,23.
Também observamos uma alta taxa de mutações de resistência ao D4 Tempacientes expostos (68,4%) e não-expostos (74%). Resultados diferentes foram encontrados na literatura, com uma prevalência de resistência variando de 0 a 20% em crianças expostas4,5,29,30. Essa discrepância nos resultados pode ser explicada pelas diferenças nos critérios de inclusão com relação às mutações de resistência ao D4T.O primeiro estudo4 considerou apenas a mutação V75T; enquanto o segundo 5 considerou as mutações T215Y, M184V, K70R, D67N; o terceiro29 considerou as mutações Q151M e 69I; e o último30 não mencionou as mutações consideradas. A definição de resistência varia entre os estudos e algoritmos de interpretação desenvolvem-se ao longo do tempo. Em nosso estudo, consideramos as mutações 41, 210, 215, 75T, 151, 69SS, publicadas pela IAS22 e pela Universidade de Stanford23.
O 3TC é também um ITRN frequentemente usado para crianças. Nove de nossos pacientes foram expostos previamente ao 3TC, sendo que 11 pacientes estavam em uso do medicamento, porum período médio de 31 meses. Encontramos mutação M184 em 63% de nossos pacientes expostos ao 3TC e em 25% dos não-expostos. Essa baixa taxa de mutação em pacientes não-expostos com mutação de resistência ao 3TC pode ser explicada pela especificidade da M184 a esse ITRN.
Para o DDI, quando consideradas as mutações 65, 74, 75T, encontramos L74V em apenas um paciente exposto (4,8%), que estava em uso do medicamento no momento da genotipagem. Em pacientes não-expostos não foi encontrada nenhuma mutação. Foram observados resultados diferentes na literatura, com taxas mais altas de mutação de resistência emcrianças expostas e não-expostas2-5. Esse resultado também pode ser explicado por dificuldades nos critérios de inclusão com relação às mutações de resistência, como acontece com o D4T. A mutação M184 não foi considerada para o DDI, com base na publicação da IAS22. Nesse relatório, a M184 não está associada à resistência ao DDI in vivo. Esse fato foi demonstradoemdiferentes estudos31-34. Operfil de resistência ao DDI conforme o Stanford HIV Drug Resistance Database também revelou umabaixa taxa de resistência (17,4%), mas, quando considerada a resistência intermediária, chegou a 65,2%.
]]> Muitas mutações conferem resistência cruzada a diversos antirretrovirais dentro dos ITRN. Esse fato pode ser demonstrado através de nossos resultados, já que pacientes sem exposição ao Abacavir e Tenofovir tiveram mutações associadas à resistência a esses inibidores. Padrões similares de resistência também foram apresentados em outros estudos pediátricos5,29.Para os ITRNN, as mutações mais frequentes foram K103 (39,1%) e Y181 (17,4%). Essa classe também é frequentemente usada na população pediátrica. Tivemos 12 pacientes que não responderam à terapia ITRNN e todos apresentaram alguma mutação que confere resistência aos ITRNN, confirmando a baixa barreira genética dessa classe. Não encontramos mutações nos ITRNN em pacientes que nunca tinham feito uso do medicamento.
Para os IP as mutações mais frequentes foram V82, I54, L90 (21,7%) e M46 (17,4%). Os IP usados com mais frequência em nossas crianças, no momento da realização do estudo, foram o Ritonavir e Nelfinavir. Embora outros IP não tenham sido usados, observamos resistência cruzada a alguns deles, com exceção do Lopinavir/r, Tipranavir e Darunavir, sugerindo um potencial benefício dessas drogas na terapia de resgate. Os polimorfismos mais frequentes foram L63 (73,9%), M36, L10 e V77 (34,8%).Opolimorfismo L63 é realmente o mais freqüente na literatura, variando de 50 a 90%22,23.
O subtipo B do HIV-1 predomina na América Latina, incluindo o Brasil, mas outros subtipos, como o F e C, também circulam, favorecendo as CRF. Estudos brasileiros2-4 incluindo crianças apresentam taxas de 67 a 78% do subtipo B e de 6 a 15% do subtipo F; similares aos nossos resultados (78% do B e 13% do F). Entretanto, considerandose o subtipo C, observamos uma taxa mais alta (4,3%). Esse fato está ocorrendo na Região Sul do Brasil, com taxas de 29 a 70%13,35,36, revelando uma distinção epidemiológica naquela região. As CRF foram encontradas em 4,4% das crianças de nosso estudo (B/B-F em 2,2% e F/B em 2,2%), mas é necessário sequenciar o gene completo para descartar novas recombinações em regiões genômicas.
Em conclusão, foi observado um extenso perfil de resistência em crianças que não responderam aos esquemas ARV, com 95% das amostras apresentando resistência a pelo menos uma classe de medicamentos.
A falta de mutações principais em crianças virgens de tratamento está de acordo com trabalhos anteriores documentando baixa prevalência dessas mutações em nosso país, assim como a presença de polimorfismos.
São necessários estudos mais detalhados em crianças para confirmar esses achados e avaliar a prevalência e importância da resistência do HIV.
Referências
1. Programa Nacional de DST e AIDS [website]. http:// www.aids.gov.br/. Acesso: 07/09/2008. [ Links ]
2. Brindeiro PA, Brindeiro RM, Mortensen C, Hertogs K, De Vroey V, Rubini NP, et al. Testing genotypic and phenotypic resistance in human immunodeficiency virus type 1 isolates of clade B and other clades from children failing antiretroviral therapy. J Clin Microbiol. 2002;40:4512-9. [ Links ]
3. Machado ES, Lambert JS,Watson DC, Afonso AO, da Cunha SM, Nogueira SA, et al. Genotypic resistance and HIV-1 subtype in Brazilian children on dual and triple combination therapy. J Clin Virol. 2004;30:24-31. [ Links ]
4. Machado DM, Fernandes SC, Succi RC, Freire WS, Pannuti CS, Gouveia AB, et al. Analysis of HIV-type 1 protease and reverse transcriptase in Brazilian children failing highly active antiretroviral therapy. Rev Inst Med Trop S Paulo. 2005;47:1-5. [ Links ]
5. Mullen J, Leech S, O’Shea S, Chrystie IL, Du Mont G, Ball C, et al. Antiretroviral drug resistance among HIV-1 infected children failing treatment. J Med Virol. 2002;68:299-304.
6. Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, Collier AC, et al. Antiretroviral-drug resistance among patients recently infected with HIV. N Engl J Med. 2002;347:385-94. [ Links ]
7. Rubio A, Leal M, Pineda JA, Caruz A, Luque F, Rey C, et al. Increase in the frequency of mutation at codon 215 associated with zidovudine resistance in HIV-1-infected antiviral-naive patients from 1989 to 1996. AIDS. 1997;11:1184-6. [ Links ]
8. Duwe S, Brunn M, Altmann D, Hamouda O, Schmidt B, Walter H, et al. Frequency of genotypic and phenotypic drug-resistant HIV-1 among therapy-naive patients of the German Seroconverter Study. J Acquir Immune Defic Syndr. 2001;26:266-73. [ Links ]
9. UK Collaborative Group on Monitoring the Transmission of HIV DrugResistance. Analysis of prevalence of HIV-1 drug resistance in primary infections in the United Kingdom. BMJ.2001; 322:1087-88. [ Links ]
10. Gomez-Cano M, Rubio A, Puig T, Perez-Olmeda M, Ruiz L, Soriano V, et al. Prevalence of genotypic resistance to nucleoside analogues in antiretroviral-naive and antiretroviral-experienced HIV-infected patients in Spain. AIDS. 1998;12:1015-20. [ Links ]
11. Simon V, Vanderhoeven J, Hurley A, Ramratnam B, Louie M, Dawson K, et al. Evolving patterns of HIV-1 resistance to antiretroviral agents in newly infected individuals. AIDS. 2002; 16:1511-9. [ Links ]
12. Dietrich U, Raudonat I, Wolf E, Jager H, Husak R, Orfanos CE, et al. Indication for increasing prevalence of resistance mutations for protease inhibitors in therapy-naive HIV-1-positive German patients. AIDS. 1999;13:2304-5. [ Links ]
13. Brindeiro RM, Diaz RS, Sabino EC, Morgado MG, Pires IL, Brigido L, et al. Brazilian Network for HIV Drug Resistance Surveillance (HIV-BResNet): a survey of chronically infected individuals. AIDS. 2003;17:1063-9. [ Links ]
14. Pires IL, Soares MA, Speranza FA, Ishii SK, Vieira MC, Gouvea MI, et al. Prevalence of human immunodeficiency virus drug resistance mutations and subtypes in drug-naive, infected individuals in the army health service of Rio de Janeiro, Brazil. J Clin Microbiol. 2004;42:426-30. [ Links ]
15. Barreto CC, Nishiya AS, Busch M, Sabino E. HIV subtype and drug resistanceamongrecent and long standing infected blood donors of Sao Paulo Brazil. In: VI Simpósio Brasileiro de Pesquisa em HIV/AIDS, 2003; Rio de Janeiro.Rio de Janeiro: FIOCRUZ; 2003. p. 90. [ Links ]
16. Barreto CC, Nishiya AS, Alcalde R, Santos MAC, Casseb J, Gonsalez CR, et al. Prevalence of mutations that confer resistance to antiretroviral drug therapyamongHIV-1 infected subjects with virological failure or among drug naive individuals in Sao Paulo city. In: VI Simpósio Brasileiro de Pesquisa em HIV/AIDS, 2003; Rio de Janeiro. Rio de Janeiro: FIOCRUZ; 2003. p.91. [ Links ]
17. Olavarria VN, Spinola J, Brites C, Galvao-Castro B. Antiretroviral drug resistance mutations in HIV-1 infected patients from Salvador, Bahia. In: VI Simpósio Brasileiro de Pesquisa em HIV/AIDS, 2003; Rio de Janeiro. Rio de Janeiro: FIOCRUZ; 2003. p. 101. [ Links ]
18. Soares MA, Brindeiro RM, Tanuri A. Primary HIV-1 drug resistance in Brazil. AIDS. 2004;18 Suppl 3:S9-13. [ Links ]
19. Simonetti SR, Schatzmayr HG, Simonetti JP. Human immunodeficiency virus type 1: drug resistance in treated and untreated Brazilian children. Mem Inst Oswaldo Cruz. 2003; 98:831-7. [ Links ]
20. Snoeck J, Kantor R, Shafer RW, Van Laethem K, Deforche K, Carvalho AP, et al. Discordances between interpretation algorithms for genotypic resistance to protease and reverse transcriptase inhibitors of human immunodeficiency virus are subtype dependent. Antimicrob Agents Chemother. 2006; 50:694-701. [ Links ]
21. de Oliveira CM, Almeida FJ,Rodrigues R, Crozatti M, Vazquez CM, do Socorro Carneiro Ferrao M, ET al. High frequency of BF mosaic genomes among HIV-1-infected children from Sao Paulo, Brazil. Arch Virol. 2008;153(10):1799-806. [ Links ]
22. JohnsonVA, Brun-Vézinet F, ClotetB, Günthard HF,Kuritzkes DR, PillayD, et al. Update of the Drug Resistance Mutations in HIV-1: 2007. Top HIV Med. 2007;15:119-25. [ Links ]
23. Stanford University. HIV drug resistance database. [website]. http://hivdb.stanford.edu Acesso: 07/09/2008. [ Links ]
24. Gonzales MJ, Machekano RN, Shafer RW. Human immunodeficiency virus type 1 reverse-transcriptase and protease subtypes: classification, amino acid mutation patterns, and prevalence in a northern California clinic-based population. J Infect Dis. 2001;184:998-1006. [ Links ]
25. King RW, Winslow DL, Garber S, Scarnati HT, Bachelor L, Stack S, et al. Identification of a clinical isolate of HIV-1 with an isoleucine at position 82 of the protease which retains susceptibility to protease inhibitors. Antiviral Res. 1995; 28:13-24. [ Links ]
26. Descamps D, Apetrei C, Collin G, Damond F, Simon F, Brun-Vezinet F. Naturally occurring decreased susceptibility of HIV-1 subtypeGto protease inhibitors. AIDS. 1998;12:1109-11. [ Links ]
27. Brown AJL, Precious HM, Whitcomb J, Simon V, Daar E, DAquila RT, et al. Reduced susceptibility of HIV-1 to protease inhibitors from patients with primary HIV infection by 3 distinct routes. In: 8th Conference on Retroviruses and Opportunistic Infections, 2001; Chicago. http://www.retroconference.org/2001/abstracts/default.htm Acesso: 07/09/2008. [ Links ]
28. Rhee SY, Gonzales MJ,Kantor R, Betts BJ,Ravela J, Shafer RW. Human immunodeficiency virus reverse transcriptase and protease sequence database. Nucleic Acids Res. 2003; 31:298-303. [ Links ]
29. Johann-Liang R, Lee SE, Fernandez A, Cervia J, Noel GJ. Genotypic characterization of human immunodeficiency virus type 1 isolated from vertically infected children with antiretroviral therapy experience. Pediatr Infect Dis J. 2000;19:363-4. [ Links ]
30. Cohen NJ, Oram R, Elsen C, Englund JA. Response to changes in antiretroviral therapy after genotyping in human immunodeficiency virus-infected children. Pediatr Infect Dis J. 2002;21:647-53. [ Links ]
31. Molina JM, Marcelin AG, Pavie J, Heripret L, De Boever CM, Troccaz M, et al. Didanosine in HIV-1-infected patients experiencing failure of antiretroviral therapy: a randomized placebo-controlled trial. J Infect Dis. 2005;191:840-7. [ Links ]
32. Winters MA, Bosch RJ, Albrecht MA, Kazenstein DA, AIDS Clinical Trials Group 364 Study Team. Clinical impact of the M184V mutation on switching to didanosine or maintaining lamivudine treatment in nucleoside reverse-transcriptase inhibitor-experienced patients. J Infect Dis. 2003;188:537-40. [ Links ]
33. Eron JJ Jr, Bosch RJ, BettendorfD, Petch L, FiscusS, Frank I; Adult Clinical Trials Group 307 Protocol Team. Antiretroviral activity of didanosine in lamivudine-experiencied subjects in comparison to activity in subjects who were lamivudine-naive. Antivir Ther. 2002;7:S135. [ Links ]
34. Pozniak A, Gazzard BB, Peeters M, Hoetelmans R, Graham NM. Influence of the M184V mutation on virologic outcome of highly active antiretroviral therapy with or without didanosine. Antivir Ther. 2002;7:S164. [ Links ]
35. Soares EA, Santos RP, Pellegrini JA, Sprinz E, Tanuri A, Soares MA. Epidemiologic and molecular characterization of humanimmunodeficiency virus type 1 in southern Brazil. J Acquir Immune Defic Syndr. 2003;34:520-6. [ Links ]
36. Brígido LF, Nunes CC, Oliveira CM, Knoll RK, Ferreira JL, Freitas CA, et al. HIV type 1 subtype C and CB Pol recombinants prevail at the cities with the highest AIDS prevalence rate in Brazil. AIDS Res Hum Retroviruses. 2007; 23:1579-86. [ Links ]
Correspondência: Flávia J. Almeida
Rua Dr. José Rodrigues Alves Sobrinho, 150/42 Matisse
CEP 05466-040 - São Paulo, SP
Tel.: (11) 9656.3149, (11) 3569.9374, (11) 3813.9611 ]]>
]]>