 1
1
Determination of the phenolic contents and evaluation of the Antityrosinase activity, and the antioxidant indexes of four Bolivian Quinoa varieties
Determinación del contenido fenólico, y evaluación de la actividad Antitirosinasa, y de los índices antioxidantes de cuatro variedades de Quinoa Boliviana
Alejandra Navia, Peggy Ormachea, Lily Salcedo, Maribel Lozano, Santiago Tarqui, Yonny Flores, Giovanna R. Almanza*
Laboratorio de Bioorgánica, Instituto de Investigaciones Químicas IIQ, Carrera de Ciencias Químicas, Facultad de Ciencias Puras y Naturales FCPN, Universidad Mayor de San Andrés UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, phone +59122792238, La Paz, www.iiq.umsa.bo ]]>
Abstract
Four varieties of Bolivian quinoa (QJG Quinoa Jacha Grano, QRB Quinoa Real Blanca, QRN Quinoa Real Negra and QRR Quinoa Real Roja) have been studied in order to valorize them through the establishment of the phenolic contents and consequently by their antioxidant indexes. In addition, and in this pathway, the antityrosinase activity of the four varieties was measured. One important quinoa-endemic glycosylated flavonoid, mauritianin 1 (Kaempferol-3-O-(2,6-di-O-α-ramnopyranosyl-β-galactopyranoside) was isolated, and identified by cromatographic and spectroscopic methods and it was quantified. The four quinoa samples were treated by means of solid-liquid extractions with hydro-alcoholic 8:2 solvents. Quantifications and evaluations were done in the samples of the four varieties of quinoa and in phenolic rich fractions from the extract of QJG, and in compound 1, coming from the QJG extract. In a series of in vitro tests, antioxidant indexes, by means of the total phenolic content and ABTS methods, and antityrosinase activities, by using the fungal tyrosinase method, of phenolic rich fractions and of the pure compound 1 were evaluated, 1 showed an important antityrosinase activity (74.73% I at 1.67 mg/mL) and antioxidant (826.68 mg GAE/g and 1141.38 µM Trolox/g), as well as the phenolic rich fraction from the extract of QJG named as EEW-1 that showed 69.89% I of the enzyme tyrosinase and significant antioxidant activity (246.08 mg GAE/g and 569.21 uM trolox/g), suggesting that these products could have a potential application in dermatology, cosmetics and food processing.
Keywords: Quinoa, Chenopodium quinoa, Antityrosinase, Antioxidant, Total Phenolic Contents, Mauritianin.
Resumen
Se han estudiado cuatro variedades de quinua boliviana (QJG Quinoa Jacha Grano, QRB Quinoa Real Blanca, QRN Quinoa Real Negra y QRR Quinoa Real Roja) para valorizarlas mediante el establecimiento de los contenidos fenólicos, y en consecuencia, por sus índices antioxidantes. Además, y en este sentido, se midió la actividad antitirosinasa de las cuatro variedades. Se aisló un importante flavonoide glicosilado endámico de la quinua, la mauritianina 1 (Kaempferol-3-O- (2,6-di-O-α-ramnopiranosil-β-galactopiranosido), que se identificó por métodos cromatográficos y espectroscópicos y se cuantificó. Se trataron las cuatro muestras de quinua por medio de extracciones sólido-líquido con solventes hidroalcohólicos 8:2. Se realizaron cuantificaciones y evaluaciones en las muestras de las cuatro variedades de quinua y en fracciones ricas en fenólicos del extracto de QJG, y en el compuesto 1, proveniente del extracto QJG. En una serie de pruebas in vitro, los índices de antioxidantes, por medio del contenido fenólico total y los métodos ABTS, y las actividades antitrosinasa, mediante el método de la tirosinasa fúngica, de fracciones ricas en fenólicos y del compuesto puro 1 fueron evaluados, 1 mostró una importante actividad antitrosinasa (74.73% I a 1.67 mg / mL) y antioxidante (826.68 mg GAE / gy Trolox 1141.38 µM / g), así como la fracción rica fenólica del extracto de QJG nombrado como EEW -1 que mostró 69.89% I de la enzima tirosinasa y actividad antioxidante significativa (246.08 mg GAE / gy 569.21 uM trolox / g), lo que sugiere que estos productos podrían tener una aplicación potencial en dermatología, cosmética y en el procesamiento de alimentos.
Palabras clave: Quinoa, Chenopodium quinoa, Antitirosinasa, Antioxidante, Contenidos fenólicos totales, Mauritianin.
INTRODUCTION
Quinoa (Chenopodium quinoa Willd), is the main crop of western Bolivia, particularly of the south highlands of the country. It is resistant to various pests and can be grown in conditions of high environmental stress, such as dry, saline soils, high ultraviolet radiation, strong winds and frost [1]. Additionally, it is a pseudo-cereal with high benefits for human health and nutrition, not only because of its important content of high quality proteins, which contain essential amino acids such as histidine and lysine, that are deficient in most grain crops, but also because its grains are rich in phenolic compounds, dietary fiber, and minerals such as calcium, iron and zinc, with a fundamental role in physiological functions and with pharmacological properties [2-4].
Various phenolic compounds, particularly flavonoids, have been previously reported in quinoa grains [2-6]. Among the flavonoids identified in quinoa we found the flavonol derivatives that are present mostly as glycosidic derivatives of kaempferol and quercetin [2,5,6]. Phenolic compounds are known as antioxidants, or molecules capable of preventing or retarding the oxidation of biological molecules such as proteins, lipids and nucleic acids [3]. The structure-activity relationship of flavonol derivatives isolated from quinoa and its antioxidant activity, showed that the ability to inhibit free hydroxyl radicals increased with the amount of hydroxyl groups in ring B, so that quercetin is the antioxidant stronger among flavonoids, however kaempferol glycosides are the major phenolics in quinoa. The study conducted by Zhu et al [2] showed a moderate antioxidant activity by DPPH of four kaempferol glycosides, while two quercetin glycosides showed a strong antioxidant activity. Which suggests that quinoa could represent an important source of free radical inhibitors.
Studies on phenolic compounds have shown that they may have antityrosinase activity [7-9]. Tyrosinase inhibitors are chemical agents capable of inhibiting the enzymatic reactions that produce the browning of food and melanin in human skin. Therefore, these agents have good commercial potential in both food processing and industrial cosmetics. Phenolic compounds can act as tyrosinase inhibitors through hydroxyl groups that bind to the active site in tyrosinase, resulting in steric hindrance or change conformation. Thus, the phenolic compounds proved to be effective inhibitors of tyrosinase activity as previously reported [7].
The aim of this study was to contribute to evaluate the nutritional, cosmeceutical and food processing advantages of four Bolivian quinoa varieties (three of Quinoa Real and Quinoa Jacha Grano) of great importance in the national market. In this sense, we have obtained extracts rich in phenolic compounds, quantified their composition of total phenols, and as well, we have isolated, characterized and quantified the major component mauritianin 1, and we have evaluated the antioxidant and antityrosinase activity of all of them.
RESULTS AND DISCUSSION
]]> Three varieties of Quinoa Real (Blanca-White QRB, Negra-Black QRN and Roja-Red QRR) from the Bolivian southern altiplano, acquired from the company Irupana Andean Organic Food SRL, and a large grain variety from the Bolivian central altiplano, called Quinoa Jacha Grano (QJG), acquired from the Municipality of Umala, were submitted to extraction processes to obtain fractions rich in phenolic compounds and for the isolation of a major flavonoid.The extraction process began with the separation of fats with petroleum ether, from the pulverized dry material (flour), followed by a maceration with EtOH 96°/H2O (80:20) for 24 h at room temperature. This process gave the hydroalcoholic extracts (HAE) of the four samples. The QJG HAE was the one with the highest yield (5,8 % with respect to 3.7 – 5.0% for the other quinoas). Additionally, it was the variety with the highest amount of raw material, 1591 g regarding the other Quinoa Real varieties of around 100 g each. For such reasons, QJG was the one chosen to obtain the fractions rich in phenolic compounds and for the isolation of the major flavonoid 1.
The hydroalcoholic maceration can lead to the extraction of some primary metabolites, such as sugars and proteins, trace elements and polar secondary metabolites, such as saponins and glycosylated flavonoids. The last two are less polar than the others, so to obtain the fraction rich in glycosylated flavonoids, successive extractions were performed with a mixture of EtOAc/EtOH 96°/H2O (EEW 50:45:5), obtaining successively three fractions with the following yields with respect to the HAE of QJG: EEW-1 (23.1%), EEW-2 (18.8%), EEW-3 (22.5%). The presence of phenolic compounds in these fractions was verified by a TLC analysis revealed with H2SO4, where it was observed that the phenolic compounds were mostly present in the first fraction (EEW-1), in the EEW-2 there was a little and in the third nothing. Therefore, it was decided to isolate the majority flavonoid from the first extraction.
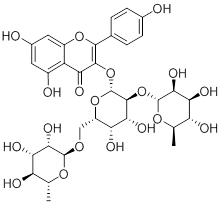
The EEW-1 fraction was submitted to a molecular exclusion chromatography (Sephadex LH-20), where the first non-flavonoid fractions were separated from the latter fractions with flavonoids, obtaining a fraction rich in flavonoids. From this fraction a separation by HPLC-prep was performed, obtaining the majority flavonoid (1), which was identified as mauritianin (kaempferol-3-O-(2,6-di-O-α-rhamnopyranosyl-β-galactopyranoside) by NMR spectroscopic analysis, and comparison with bibliographic data [2].
 1
1
Figure 1. Structure of compound 1 mauritianin
Compound 1 is a glycosylated flavonoid with kaempferol as aglycone and three monosaccharides linked to the C-3 via an O-glycosidic bond. This compound has been previously reported in other quinoa varieties, as one of the major flavonoids [10], and its antioxidant properties were evaluated by the DPPH free radical inhibition method, showing a moderate activity. Considering the potentialities of this flavonoid, glycosylated or as aglycone, which can be translated into advantages of the varieties under study, mauritianin (1) was quantified in the four varieties of quinoa as well as in extracts and fractions rich in phenols obtained in this study. The results are shown in Table 1, where it can be observed that in the Quinoa Real varieties of the south altiplano (QRB, QRN and QRR) there is a higher concentration of this flavonoid than in the variety of the central altiplano QJG. The highest concentration is observed in the QRN , which is consistent with the consideration that the flavonoids participate in the coloration of plants. In addition, it is observed that the fraction rich in phenolic compounds, EEW-1, has the highest concentration of 1 (Figure 2).
Flavonoids are mostly present as glycosides in plants. During intestinal absorption, these glycosides are mostly hydrolyzed to their aglycones, then the bioactivity is attributed to the aglycone structures and not to the sugar moieties. Thus, it is important to consider the properties of kaempferol. This flavonol is widely distributed in the plant kingdom. Numerous edible plants contain kaempferol, and it has been estimated that the human dietary intake of this polyphenol can be up to approximately 10 mg/day [11]. Epidemiological studies have found a positive result between the consumption of foods containing kaempferol and a reduction in the risk of developing cardiovascular diseases and some types of cancer [12]. Likewise, numerous in vitro studies and some in vivo support the role of kaempferol in the prevention and/or treatment of the mentioned and other diseases, such as neurodegenerative, infectious diseases, diabetes, osteoporosis, inflammations and pain. However, most in vitro studies have been performed at much higher concentrations than those found in plasma and tissues after oral administration of kaempferol [11]. This means that some biological effects induced by kaempferol in vitro may not be relevant in vivo when this flavonoid is taken orally. However, it is a potential drug that still requires bioavailability studies [11].
]]>Table 1. Contents of the flavonoid 1 in quinoa grains, extracts and fractions rich phenolic compounds
Figure 2. Contents of mauritianin 1 in quinoa grains, extracts and fractions rich phenolic compounds
Then, the antityrosinase activity, the total phenolics contents and the antioxidant activity (ABTS method) were evaluated of the HAE from QJG and QRB, the fractions rich in phenolic compounds (EEW-1, EEW-2) and of the isolated flavonoid mauritianin (Table 2). Among the Quinoa Real varieties only the QRB was evaluated because it is the one with the highest production in the Bolivian Southern Highlands and there is more availability of the plant material.
Tyrosinase is the speed-limiting enzyme of the biosynthesis of melanin pigments responsible for the coloration of the skin, hair and eyes, as well as the browning of foods. Tyrosinase inhibitors have good potential both dermatologically and in food processing, they can thus be clinically useful for the treatment of some dermatological disorders associated with hyperpigmentation due to high melanin production; as well as in cosmetic for depigmentation after burns [13]. Kojic acid was used as a positive control, because it shows a high inhibition of this enzyme [13,14]. Table 2 shows that the percentage of tyrosinase inhibition of flavonoid 1 is comparable to that of kojic acid, although somewhat less, and the inhibition of the EEW-1 fraction is also interesting. Some flavonoids, such as kaempferol, quercetin and morin, showed an inhibitory activity of tyrosinase [15,16]. The studies showed that flavonoids containing an α-keto group show potent tyrosinase inhibitory activity [15]. This inhibition ability may be explained in terms of similarity between the dihydroxyphenyl group in L-DOPA and the α-keto group in flavonoids. But, the activity of some flavonols possessing a 3-hydroxy-4-keto moiety, such as kaempferol and quercetin, seems to be related to their ability to chelate the copper in the active site, leading to irreversible inactivation of tyrosinase, that’s why some 3-O-glycosides derivatives didn’t show any activity. However, although the hydroxyl group in C-3 somehow relates to the activity, it may not be essential because several flavones, such as luteolin and luteolin 7-O-glucoside which lack this 3-hydroxyl group [17], or 3-O-glycosides like quercetin-3-O-(6-O-malonyl)-β-D-glucopyranoside and kaempferol-3-O-(6-O-malonyl)-β-D-glucopyranoside [18], still showed inhibition of tyrosinase, but with other inhibition mechanisms, mainly associated with phenolic hydroxyl groups that could form a hydrogen bridge bond with amino acids of the active tyrosinase site [18,7]. Anyway, mauritianin (1) and the fraction rich in phenolic compounds EEW-1, showed an interesting potential as tyrosinase inhibitors but more studies are needed to demonstrate their usefulness in this field.
]]> Table 2. Evaluation of antityrosinase and antioxidant activity of extracts, fractions rich in phenolic compounds and compound 1
The derivatives radical of oxygen (ROS) are the most important free radical in biological systems and harmful byproducts generated during normal cellular functions. Increasing intake of natural antioxidants may help to maintain a tolerable antioxidant status. The phenolic compounds and in particular the flavonoids are one of the most important groups inhibitors of free radicals [19]. In this sense we evaluated the total phenolic contents by Folin-Ciocalteu, and the inhibition of free radicals by the ABTS method. Table 2 shows that the highest amount of Gallic Acid Equivalents (GAE) is found in mauritianin, this was expected if we consider that it is a pure compound and has three phenolic hydroxyl groups in its structure. The fractions EEW-1 and EEW-2 show higher concentration of phenols than the HAE, this proves that the process to obtain fractions rich in phenolic compounds was successful. Finally, we observed that the concentration of phenols in QRB is bigger than in QJG, which can also contribute to the bitterness of Quinoa Real. Finally, in the ABTS method, the pure compound is also the one that presents the highest free radical inhibitory activity, which is in accordance with previous studies where it was already shown that this compound had antioxidant properties due to its ability to inhibit radicals free. Numerous studies have shown that kaempferol and some glycosides of kaempferol have antioxidant activity not only in vitro, but also in vivo, those studies have shown that the presence of a double bond in C2-C3 in conjunction with an oxo group in C4, and the presence of hydroxyl groups in C3, C5 and C4 ', are important structural features involved in the antioxidant activity of kaempferol [19, 20]. Mauritianin has all these structural features except the OH group in C-3, so probably its antioxidant activity is somewhat lower than that of kaempferol, but that should be confirmed. It is also interesting to note that the EEW-1 fraction shows interesting antioxidant properties, all of which increase the functional properties of these quinoa varieties. However, we need more studies to prove the effectiveness of the fraction EEW-1 and the pure compound 1 as antioxidants, using other antioxidant methods in vitro and in vivo.
EXPERIMENTAL
Plant material
The Irupana Andean Organic Food society provided with the three varieties of Quinoa Real: Blanca-White QRB, Negra-Black QRN and Roja-Red QRR coming from the Bolivian southern altiplano. The variety Quinoa Jacha Grain QJG coming from the Bolivian central altiplano was offered by the Municipality of Umala, La Paz. In both cases the suppliers identified by the name the varieties given.
Extraction
Quinoa grains were subsequently subjected to grinding with a grain grinder-doser (Brand: La Pavoni SpA, Model: Zip, Series: Automatic-Base), for subsequent sieving with a 250 µm sieve (No. 60) (Retschun, ASTM E11), obtaining flour from the four varieties.
Quinoa flour, of the four varieties, were weighed: QJG (1591 g), QRB (76.5 g), QRR (100.0 g) and QRN (100.0 g) and extracted with petroleum ether, fraction 40-60ºC, to separate the fats. From the degreased flour of the four varieties, the hydroalcoholic extracts (HAE) were obtained, by maceration at room temperature for 24 hours with EtOH 96°/H2O (80:20), in a ratio 1:10 (weight: volume). The extracts were concentrated by rotaevaporation until complete removal of EtOH and the resulting aqueous extracts were dried by lyophilization, obtaining the dry extracts, the yield of each extract respect to the starting material was calculated.
]]> To obtain fractions rich in phenolic compounds, a first extraction from the HAE of the QJG, was carried out using a solvent mixture: EtOAc/EtOH 96°/H2O (EEW 50:45:5), with constant stirring for five hours at 50°C, following similar processes employed to obtain glycosylated flavonoids [21, 22]. After reposing, two phases were evident, the supernatant or liquid phase and the precipitate insoluble solid phase. These two phases were separated by filtration in filter paper. The obtained liquid fraction was concentrated by rotaevaporation until total elimination of the solvent and then by lyophilization, until the elimination of the water residues, obtaining the fraction EEW-1. The residue of the first extraction with EEW, or solid phase, was subjected to two successive extractions more, made with the same mixture of solvents in the same manner, giving the fractions EEW-2, EEW-3, which were analyzed by TLC revealed with H2SO4 (5%), for the determination of phenolic compounds.Isolation of compound 1
The isolation of the majority flavonoid 1 was performed from the EEW-1 fraction. 2 g of EEW-1 were initially submitted to a separation by a molecular exclusion chromatography Sephadex LH-20, which gave two fractions: FI fractions without flavonoids and FII (598 mg) with flavonoids. The FII fraction was subjected to a further separation by a preparative HPLC chromatography using a Shimadzu HPLC-prep equipment, consisting of a 5 µmx100x 10 mm Restek Ultra C-18 column; a UV-Vis detector, model SPD-20 A; and a isocratic pump LC-20AP coupled to a fraction collector. For the separation, an isocratic mixture of acetonitrile/ 0.1% formic acid (17:83) and a flow of 4 ml/min were used. The chromatogram was observed at a wavelength of 280 nm, getting the separation of the compound 1 (23.6 mg) in the intermediate fractions.
Mauritianin 1 (kaempferol-3-O- (2,6-di-O-α-ramnopyranosyl-β-galactopyranoside)
1H NMR (300 MHz, DMSO) Kaempferol: d 6.36 (1H, d, J = 1.8 Hz, H-6), 6.14 (1H, d, J = 1.8 Hz, H-8), 8.03 (2H, d, J = 10.2 Hz, H-2', H-6’), 6.85 (2H, d, J = 8.4 Hz, H-3', H-5’) Important signals of sugars: Gal- d 5.55 (1H, d, J = 7.2 Hz, anomeric H), Rha- 5.08 (1H, d, J = 6.6 Hz, anomeric H), 0.77 (3H, d, J = 6.6 Hz), Rha– 4.76 (1H, br s, anomeric H), 0.97 (3H, d, J = 6.0 Hz).
13C NMR (75 MHz, DMSO) Kaempferol δ 156.9 (C-2), 133.0 (C-3), 177.5 (C-4), 160.3 (C-5), 99.6 (C-6), 161.6 (C-7), 94.4 (C-8), 156.7 (C-9), 103.9 (C-10), 121.4 (C-1'), 131.2 (C-2'), 115.5 (C-3'), 156.9 (C-4'), 115.5 (C-5'), 131.2 (C-6'), Gal - 100.5 (C-1''), 75.3 (C-2''), 74.8 (C-3''), 70.9 (C-4''), 74.3 (C-5''), 68.9 (C-6''), Rha - 101.0 (C-1'''), 71.1 (C-2'''), 71.0 (C-3'''), 73.7 (C-4'''), 65.8 (C-5'''), 17.7 (C-6'''), Rha - 99.5 (C-1''''), 68.7 (C-2''''), 68.6 (C-3''''), 72.3 (C-4''''), 65.2 (C-5''''), 18.4 (C-6'''').
Quantification of compound 1
The quantification of compound 1 was carried out on a HPLC Agilent Technologies 1100 equipment consisting of a quaternary pump (Quat Pump-G1311A), a continuous vacuum degasser (G1322), a manual injector and a diode array detector (DAD). Separations were conducted at 40oC on an Agilent Eclipse Plus RP-C18 column (4.6 mm x 250 mm length, 5 µm particle size) with a pre-column filter of 0.5 µm (Agilent Technologies). The mobile phase was a binary solvent system using (C): 0.1% formic acid in ultrapure water and (D): acetonitrile, based in the method described by Paśko et. al. [23]. For the analysis the samples were dissolved in MeOH, the injection volume was 25 µl, at a flow rate of 1.0 ml/min. The elution gradient applied was: 95%C and 5% D from 0 to 21 min, 75%C and 25% D from 22 to 31 min, 45% C and 55% D from 32 to 35 min. The eluates were detected at 360 nm.
For the quantification of 1, a calibration curve was done using the mauritianin isolated from QJG. Figure 3 shows the HPLC-DAD chromatogram of 1 using the described method where the analyte appears at retention time tR= 19.8 min. The calibration curve, was obtained using the peak areas of the standard with different concentrations (25, 50, 75, 100, 125 y 150 ppm). The area for every point was the average of three runs. The linear correlation equation of the Area vs. Concentration and the linear coefficient were: 8.6036x + 15.277 (R2= 0.998).
]]> The content of mauritianin in the extracts and fractions of the different varieties of quinoa was determined by using the calibration curve. With this purpose, 30.0 mg of each sample was dissolved in 5 ml of MeOH, subsequently passed through a 0.2 µm filter and 25 µl of the solution was injected. Then, the area of the compound at retention time tR= 19.8 min was recorded by triplicate, and the average was used for the calculation of the amount of mauritianin in the HAE and fractions rich in phenolic compounds.
Figure 3. HPLC chromatogram of mauritianin 1
Antityrosinase activity
The tyrosinase inhibition assay was carried out by means of an enzymatic colorimetric test based on a previous reported method [24,25], which gives a quantitative data by spectrophotometric analysis using L-tyrosina as substrate. The percentage of tyrosinase inhibition of the samples (HAE-QJG, EEW-1, EEW-2, HAE-QRB and mauritianin) were determined, using 70 µL of the sample (5.0 mg/mL phosphate buffer pH 6.5; 50 mM), with 30 µL of the enzyme L-tyrosinase (0.20 mg/mL phosphate buffer pH 6.5; 50 mM)), this mixture was stirred in a vortex and incubated for 60 min at 37 °C, then 110 µL of the L-tyrosine substrate (3.6 mg/10 mL phosphate buffer pH 6.5; 2 mM)) was added, again stirred and incubated for 15 min more at 37 ° C, the absorbance reading was measured at 492 nm. Kojic acid was used as a positive control (5.0 mg/mL phosphate buffer (pH 6.5; 50 mM) and phosphate buffer (pH 6.5; 50 mM) as blank.
The percentage of tyrosinase inhibition (I %) was calculated as follows: I % = [(Acontrol – Asample)/Acontrol] × 100 where Acontrol is the absorbance of the blank and Asample is the absorbance of the samples. Analyses were run in triplicate and the result was expressed as average values with standard error mean (SEM).
Total phenolic content
The total phenolic content in the samples (HAE-QJG, EEW-1, EEW-2, HAE-QRB and mauritianin) was determined by the Folin-Ciocalteu method given in the literature [26] with slight modifications. 10 µL of the sample (30.0 mg/5 mL of methanol for extracts and 4.0 mg/5 mL of methanol for mauritianin) was taken with a mixture of 500 µL of the Folin Ciocalteu reagent (1:10 v/v), 400 µL of 7.5% sodium carbonate (m/v) and 90 µL of distilled water. This mixture was stirred in a vortex and incubated for 15 min at 45 ° C, and the absorbance was measured at 765 nm.
]]> For the determination of total phenolic contents, we used a calibration curve with gallic acid constructed using different concentrations: 40, 80, 120, 160 and 200 mg/mL under the same conditions as above, corresponding to a linear equation y= 0.0014x + 0.0027 (R2= 0.999). Where y = absorbance at 765 nm and x= mg GAE (Gallic Acid Equivalents)/mLAntioxidant evaluation by ABTS cation radical scavenging activity
The scavenging activity against the radical-cation ABTS•+ (2,2_-azino-bis(3-ethylbenzothiazoline)-6-sulfonic acid) was measured according to the method given in the literature [27] with slight modifications. The ABTS•+ cation radical was produced by the reaction between 7 mM of ABTS in H2O and 2.42 mM of potassium persulfate, and stored in the dark at room temperature for 12-16 h. On the day of analysis, the ABTS•+ solution was diluted with ethanol to an absorbance of 0.70 (±0.02) at 734 nm. Extracts and fractions were prepared by dissolving 30.0 mg of each in 5 mL of methanol, and mauritianin was prepared by dissolving 4.0 mg of the compound in 5 mL methanol. For the measurement of the activity 10 µL of the sample with 1000 µL of ABTS•+ solution was stirred in a vortex and the absorbance at 734 nm was recorded.
Methanol was used as blank. The Trolox amount was estimated from the standard Trolox curve made by use of Trolox at 100, 250, 500 and 1000 µM under the same conditions described above, which gave a linear equation y= 0.0005x + 0.6512 (R2= 0.998). Where y= absorbance at 734 nm and x= µM Trolox equivalent concentration. The ABTS radical cation scavenging activity was expressed as µM Trolox equivalent/g sample.
ACKNOWLEDGEMENTS
We are grateful to SIDA Swedish Agency for the financial support of the project “Biomolecules of industrial and medicinal interest. Anticancer”. We also would like to thank to Irupana Andean Organic Food S.A. and the Municipality of Umala, La Paz for providing the plant material of this study.
REFERENCES
1. Jacobsen, S.E. 2001, The situation of quinoa and its production in southern Bolivia: From the economic success environmental, J. Agron. And Crop Sc. 197, 390-399. [ Links ]
2. Zhu, N., Sheng, S., Li, D., Lavoie, E.J., Karwe, M.V., Rosen, R.T.,Ho, C. 2001, Antioxidative flavonoid glycosides from quinoa seeds (Chenopodium quinoa Willd), Journal of Food Lipids, 8, 37-44. [ Links ]
3. Hirose, Y., Fujita, T., Ishii, T., Ueno, N. 2010, Antioxidative properties and flavonoid composition of Chenopodium quinoa seeds cultivated in Japan, Food Chemistry, 119 (4), 1300-1306. [ Links ]
4. Maradini, A.M., Ribeiro M., Da Silva J.T., Pinheiro H.M., Paes J. B., Dos Reis J.S. 2017, Quinoa: Nutritional, Functional and Antinutritional Aspects, Critical Reviews in Food Science and Nutrition, 57, 1618-1630. [ Links ]
5. Lin, M., Han, P., Li, Y., Wang, W., Lai, D., Zhou, L. 2019, Quinoa Secondary Metabolites and Their Biological Activities or Functions, Molecules, 24, 2512-2559. [ Links ]
6. Hernández-Ledesma, B. 2019, Quinoa (Chenopodium quinoa Willd) as a source of nutrients and bioactive compounds: a review, Bioactive Compounds in Health and Disease, 2 (3), 27-47. [ Links ]
7. Salleh, W.M.N.H.W., Ahmad, F., Yen, K.H. 2014, Antioxidant and Anti-tyrosinase Activities from Piper officinarum C,DC (Piperaceae), Journal of Applied Pharmaceutical Science, 4 (5), 87-91. [ Links ]
8. Zuo, A., Dong, H., Yu, Y., Shu, Q., Zheng, L., Yu, X., Cao, S. 2018, The antityrosinase and antioxidant activities of flavonoids dominated by the number and location of phenolic hydroxyl groups, Chin Med, 13, 51-63. [ Links ]
9. Sun, L., Guo, Y., Zhang, Y., Zhuang, Y. 2017, Antioxidant and Anti-tyrosinase Activities of Phenolic Extracts from Rape Bee Pollen and Inhibitory Melanogenesis by cAMP/MITF/TYR Pathway in B16 Mouse Melanoma Cells, Frontiers in Pharmacology, 8, Article 104, doi: 10.3389/fphar.2017.00104 [ Links ]
10. De Simone, F., Dini, A., Pizza, C., Saturnino, P., Schettino, 0. 1990, Two flavonol glycosides from Chenopodium quinoa, Phytochemistry, 29 (11), 3690-3692 [ Links ]
11. Calderon-Montano, M., Burgos-Moron, J., Perez-Guerrero, E., Lopez-Lazaro, C.M. 2011, Review on the Dietary Flavonoid Kaempferol, Mini Reviews in Medicinal Chemistry, 11 (4), 298-344, [ Links ]
12. Gates, M.A., Tworoger, S.S., Hecht, J.L., De, V.I., Rosner, B., Hankinson, S.E., 2007, A prospective study of dietary flavonoid intake and incidence of epithelial ovarian cancer, Int. J. Cancer, 121, 2225-2232. [ Links ]
13. Almeda, F., Astorga, L., Orellana, A., Samuel, L., Gaitan, I., Cáceres, A. 2015, Piper genus: source of natural products with antityrosinase activity favored in phytocosmetics, International Journal of Phytocosmetics and Natural Ingredients. 2:6. doi:10.15171/ijpni.2015.06 [ Links ]
14. Bochot C., Gouron, A., Bubacco, L., Milet, A., Philouze, C., Re´glier, M., Serratrice, G., Jamet, H, Belle C. 2014, Probing kojic acid binding to tyrosinase enzyme: insights from a model complex and QM/MM calculations, Chem. Commun., 50, 308-310 [ Links ]
15. Kim, Y.J., Uyama, H. 2005, Tyrosinase inhibitors from natural and synthetic sources: structure, inhibition mechanism and perspective for the future, Cellular and Molecular Life Sciences, 62 (15), 1707–1723
16. Lee, S.Y., Baek, N., Nam T. 2016, Natural, semisynthetic and synthetic tyrosinase inhibitors, Enzyme Inhib Med Chem, 31 (1), 1–13.
17. Badria F.A. and El Gayyar M.A. 2001, A new type of tyrosinase inhibitors from natural products as potential treatments for hyperpigmentation, Boll. Chim. Farma., 140, 267–271.
18. Yang, Z., Zhang, Y., Sun, L., Wang, Y., Gao, X., Cheng, Y., 2012, An ultrafiltration high-performance liquid chromatography coupled with diode array detector and mass spectrometry approach for screening and characterizing tyrosinase inhibitors from mulberry leaves, Analytica Chimica Acta, 719, 87– 95.
19. Sarian, M.N., Ahmed, Q.U., Mat So’ad, S.Z., Alhassan, A.M., Murugesu, S., Perumal, V., Mohamad, S.N.A.S., Khatib, A., Latip, J., 2017, Antioxidant and Antidiabetic Effects of Flavonoids: A Structure-Activity Relationship Based Study, BioMed Research International, Article ID 8386065, 1-14.
20. Cid-Ortega, S., Monroy-Rivera, J.A. 2018, Extraction of Kaempferol and Its Glycosides Using Supercritical Fluids from Plant Sources: A Review, Food Technol Biotechnol., 56 (4), 480–493.
21. Braca, A., Sortino, C., Politi, M., Morelli, I., Mendez, J. 2002, Antioxidant activity of flavonoids from Licania licaniaeflora, Journal of Ethnopharmacology 79, 379–381.
]]> 22. Yang, R., Guan, Y., Wang, W., Chen, H., He, Z., Jia, A-Q. 2018, Antioxidant capacity of phenolics in Camellia nitidissima Chi flowers and their identification by HPLC Triple TOF MS/MS, PLoS ONE 13(4): e0195508. https://doi.org/10.1371/journal.pone.0195508.23. Paśko, P., Sajewicz, M., Gorinstein, S., Zachwieja, Z. 2008, Analysis of selected phenolic acids and flavonoids in Amaranthus cruentus and Chenopodium quinoa seeds and sprouts by HPLC, Acta Chromatographica, 20 (4), 661–672.
24. Momtaz, S., Mapunya, B., Houghton, P., Edgerly, C., Hussein, A., Naidoo, S., Lall, N. 2008, Tyrosinase inhibition by extracts and constituents of Sideroxylon inerme L. stem bark, used in South Africa for skin lightening, Journal of Ethnopharmacology, 119, 507–512.
25. Lim, T.Y., Lim, Y.Y., Yule C.M. 2009, Evaluation of antioxidant, antibacterial and anti-tyrosinase activities of four Macaranga species, Food Chemistry, 114, 594–599.
26. Labiad, M.H., Harhar, H., Ghanimi, A.,Tabyaoui, M. 2017, Phytochemical Screening and Antioxidant Activity of Moroccan Thymus satureioïdes Extracts, JMES, 8 (6), 2132-2139. [ Links ]
27. Tejeda, L. Peñarrieta, M., Alvarado, J., Ǻkesson, B. y Bergenståhl, B. 2008, Determination of total antioxidant capacity and total phenolic compounds in andean grains (Quinua, Cañihua, Amaranth and Qentu), Revista Boliviana de Química, 25 (1), 70–74.
]]>