MECHANISTIC VIEWS OF STEREOSELECTIVE SYNTHESIS OF TRI-AND TETRA-SUBSTITUTED ALKENES, PART I; THE ORGANIC CHEMISTRY NOTEBOOK SERIES, A DIDACTICAL APPROACH, N°3
José A. Bravo*, José L. Vila
Department of Chemistry, Instituto de Investigaciones en Productos Naturales IIPN, Universidad Mayor de San Andrés UMSA, P.O. Box 303, Tel. 59122792238, La Paz, Bolivia
*Corresponding author: jabravo@umsa.bo
As underlined in two previous papers in: "The Organic Chemistry Notebook Series, a Didactical Approach", the presentation of synthesis works in a verbal and graphical succinct manner, needs a didactical approach. Isomerically pure tri- and tetra-substituted alkenes are difficult to obtain as shown in several publications. We used a series of reactions to synthesize tri- and tetra-substituted alkenes as reviewed by W. Carruthers, and we have proposed didactical and mechanistic views for the reviewed reactions.
Keywords: Organic Chemistry, Stereoselective synthesis, Alkenes, Addition reaction, Mechanisms of Reactions.
ANALYSIS AND MECHANISTIC PROPOSALS
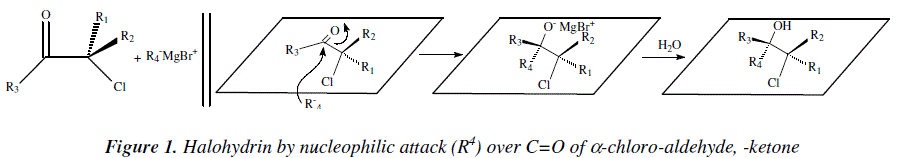
As academics we are concerned with the didactical importance of covering the needs of debutant students in organic synthesis. This is a third study in: "The Organic Chemistry Notebook Series, a Didactical Approach" [1,2]. The present article is an analytical and didactical approach to stereoselective synthesis of tri- and tetra-substituted alkenes as reviewed by W. Carruthers [3]. Tri- and tetra-substituted alkenes are difficult to obtain. Authors [3,4] signaled that the substrate a-chloro-aldehyde or -ketone, in its way to alkene, finds its critical step when reacting with Grignard reagent. Also, the most reactive conformation for the substrate is when the carbonyl group and the carbon-chlorine are antiparallel dipoles [3,4]. This conformation allows an addition of Grignard reagent very stereo-selectively and from the side less hindered by R1 and R2 groups, on the a-carbon atom with respect to carbonyl [3,4]. The nucleophilic attacking group R4 orients itself in an anti disposition with respect to the most hindering group between R1 and R2 [3,4]. The derived chlorohydrin is then submitted to stereoselective reactions to afford the alkene where three of the double bond substituents come from the alfa-chloro-aldehyde, -ketone and the fourth one comes from Grignard reagent [3,4]. Figure 1 shows the corresponding mechanistic view.
As an example let us examine the synthesis of (E)-3-methyl-2-pentene [3,4] (Figure 2), and let us propose the corresponding mechanistic view (Figure 3).
]]>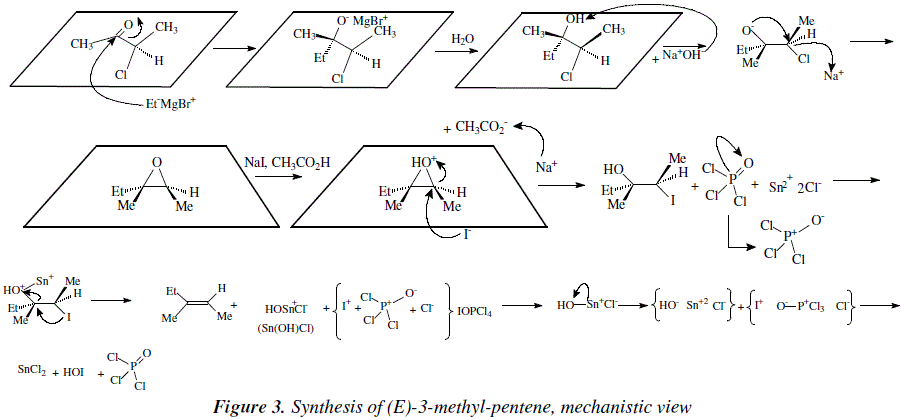
Similarly, a series of stereo-selective reactions with methylmagnesium iodide (Grignard reagent) onto substrate 2-chloropentane-3-one gave rise to (Z)-3-methyl-2-pentene [3,4] as shown in the mechanism of Figure 4. These reaction series are an elegant way to obtain pure stereo-isomers.
A different reaction to afford the stereo-specific obtaining of 2- or 3-alkylated allylic alcohol from a propargylic alcohol is achieved by reduction of propargylic alcohol into ![]() - or
- or ![]() -iodoallylic alcohols with aluminium hydride reagent (modified) and ulterior reaction of the reduction product with iodine [3,5], Figure 5.
-iodoallylic alcohols with aluminium hydride reagent (modified) and ulterior reaction of the reduction product with iodine [3,5], Figure 5.
Reduction is done with presence of NaOMe and the final product is only ![]() -iodoallylic alcohol. If reduction is carried out with LiAlH4 in the presence of AlCl3, final iodination gives final
-iodoallylic alcohol. If reduction is carried out with LiAlH4 in the presence of AlCl3, final iodination gives final ![]() -iodoallylic alcohol exclusively.
-iodoallylic alcohol exclusively.
The resulting iodo compounds with LiR2Cu afford the corresponding substituted allylic alcohol, where the original substituents of the propargyl alcohol, are now trans to each other (Figure 5) [3,5]. The explicit mechanism for these reactions are shown on Figure 6 and 7. Figure 6 shows the mechanism for the synthesis of ![]() -iodinated, allylic alcohol.
-iodinated, allylic alcohol.
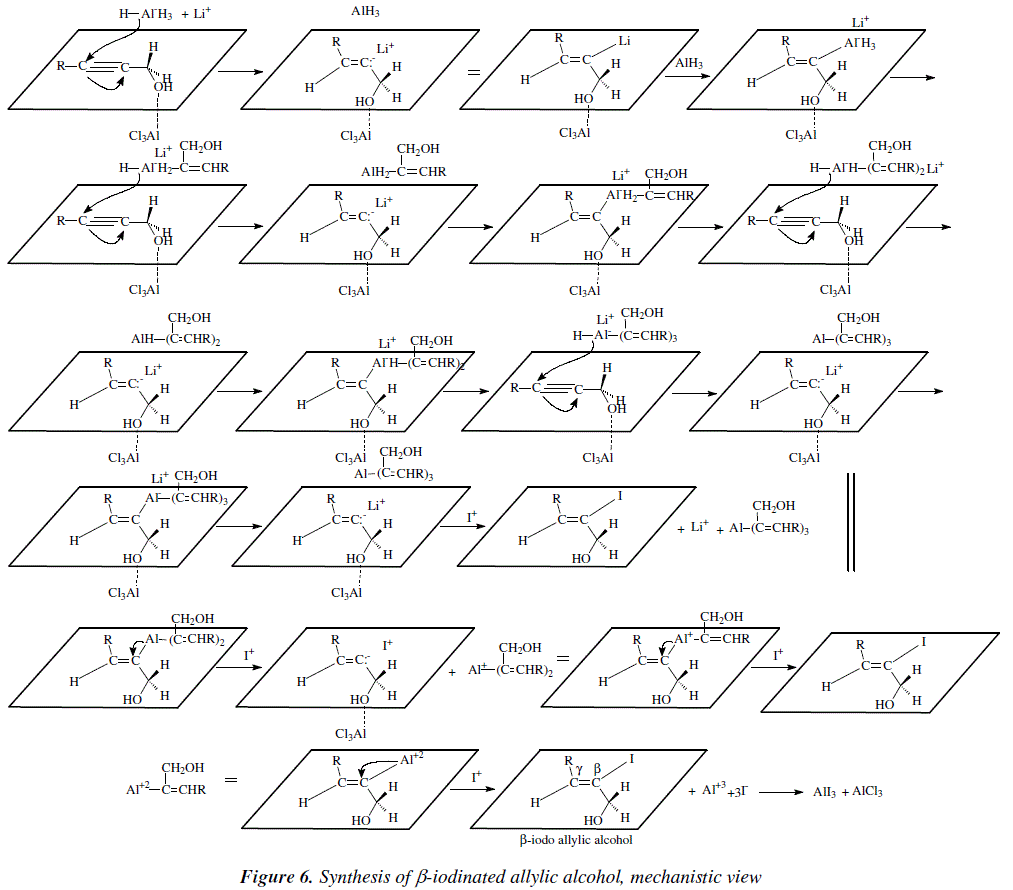
For the ![]() -iodinated allylic alcohol, NaOCH3 is employed (Figure 5) [3,5]. The corresponding mechanism is shown in Figure 7.
-iodinated allylic alcohol, NaOCH3 is employed (Figure 5) [3,5]. The corresponding mechanism is shown in Figure 7.
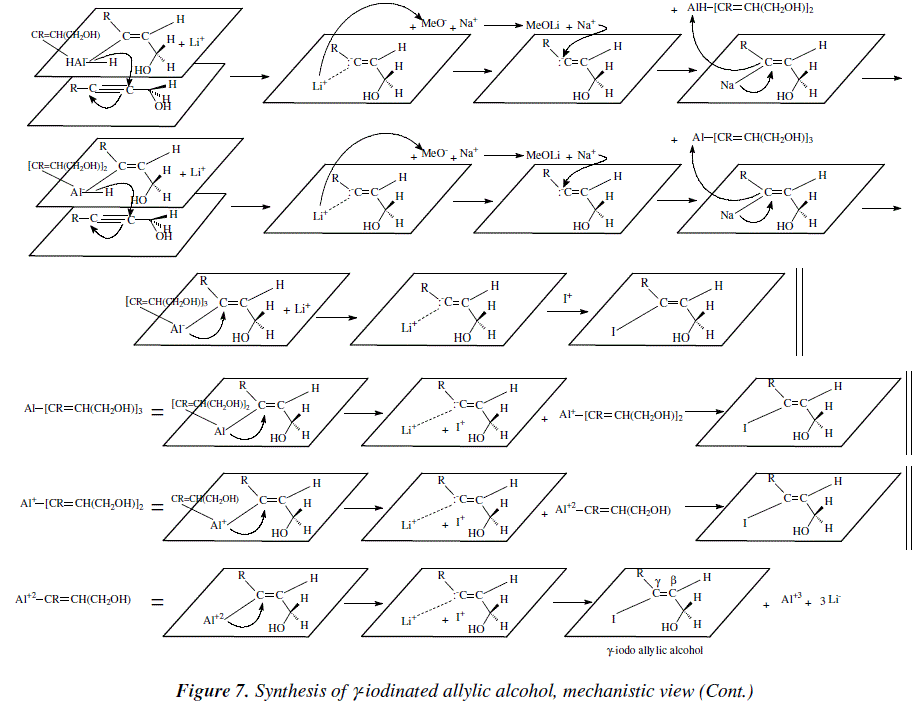
The ![]() -iodinated and
-iodinated and ![]() -iodinated allylic alcohols are easily transformed by action of lithium dimethylcuprate over substrates to afford 2-, 3-alkylated allylic alcohols as shown in Figure 8.
-iodinated allylic alcohols are easily transformed by action of lithium dimethylcuprate over substrates to afford 2-, 3-alkylated allylic alcohols as shown in Figure 8.

This method found application in the stereo-specific C=C bond formation in the synthesis of trisubstituted derivatives. This was the case for the key step in the synthesis of the dl-C18 Cecropia juvenile hormone, as reviewed by W. Carruthers [3,6], Figure 9.
Figure 10 is the mechanism corresponding to the key step in the synthesis of dl-C18 Cecropia juvenile hormone.
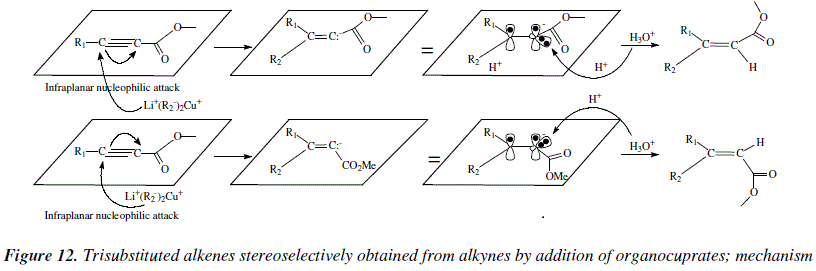
Organocopper and organoborane reagents are employed to obtain stereoselectively tri- and tetra-substituted alkenes by addition on alkynes [3,7]. Organocuprates lead to ![]()
![]() -dialkylacrylic esters by reaction with
-dialkylacrylic esters by reaction with ![]()
![]() -acetylenic esters (Figure 11) [3]. The structure of each stereoisomer depends on the temperature and the solvent [3]. High yield of
-acetylenic esters (Figure 11) [3]. The structure of each stereoisomer depends on the temperature and the solvent [3]. High yield of ![]() ,
,![]() -dialkylacrylic ester is achieved at -78°C in THF [3]. Contrasting with reactions employing propargyl alcohols, this reaction produces alkenes in which the substituents in the acetylenic precursor are cis to each other in the alkene [3].
-dialkylacrylic ester is achieved at -78°C in THF [3]. Contrasting with reactions employing propargyl alcohols, this reaction produces alkenes in which the substituents in the acetylenic precursor are cis to each other in the alkene [3].
The mechanistic views conducting to alkenes by this method are exposed in Figure 12.
]]>
Organocopper(I) reagents add readily to terminal alkynes giving rise to 1-alkenylcopper(I) compounds [3]. These organocopper(I) reagents are RCu.Alkyl copper(I) compounds that can be obtained from Grignard reagents and an equimolar quantity of copper(I) bromide or copper(I) bromide-dimethylsulphide complex [3]. Copper adds to the alkyne on the terminal carbon, adding the alkyl group of the alkylcopper(I) reagent, in a syn manner [3]. This kind of products, alkenylcopper(I) compounds, react with electrophiles like alkyl halides, ![]()
![]() -unsaturated ketones and epoxides to afford trisubstituted alkenes with almost complete retention of configuration [3,7,8]. See Figure 13. The elaborated mechanism is shown in Figure 14.
-unsaturated ketones and epoxides to afford trisubstituted alkenes with almost complete retention of configuration [3,7,8]. See Figure 13. The elaborated mechanism is shown in Figure 14.
The method was employed as shown in the examples of Figure 15.
The corresponding mechanistic views are exposed in Figures 16 and 17.
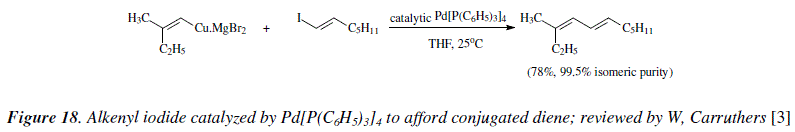
Alkenyl iodides also react in the presence of Pd(PPh3) as catalyst to give conjugated dienes [3,9], Figure 18.
]]>
The corresponding mechanism is exposed in Figure 19.
ACKNOWLEDGEMENTS
Authors express their gratitude to Prof. Eduardo Palenque from the Physics Department, Universidad Mayor de San Andrés, for bibliographic support.
REFERENCES
1. Bravo, J. 2005, Bol. J. ofChem., 23, 1-10. (http://www.bolivianchemistyournal.org, 2005). [ Links ]
2. Bravo, J.A., Mollinedo, P., Peñarrieta, J.M., Vila, J.L. 2013, Bol. J. ofChem., 30, 24-41 (http://www.bolivianchemistryjournal.org, 2013) [ Links ]
3. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 148-156. [ Links ]
4. Conforth, J.W., Conforth, R.H., Mathew, K.K. 1959, J. Chem. Soc., 112. [ Links ]
5. Corey, E.J., Katzenellenbogen, J.A., Posner, G.H. 1967, J. Am. Chem. Soc., 102, 1433. [ Links ]
6. Corey, E.J., Katzenellenbogen J.A., Gilman N.W., Roman, S.A., Erickson, B.W. 1968, J. Am. Chem. Soc., 90, 5618. [ Links ]
7. Carruthers, W. In Comprehensive Organometallic Chemistry, eds. Wilkinson, G., Stone F.G.A., Abel, E.W., Vol. 7, Pergamon Press, 1982, Oxford, U.K., pp. 716. [ Links ]
8. Marfat, A., McGuirk, P.R., Helquist, P. 1979, J. Org. Chem., 44, 3888. [ Links ]
9. Jabri, N., Alexakis, A., Normant, J.F. 1982, Tetraedron Lett. 23, 1589. [ Links ]
]]>