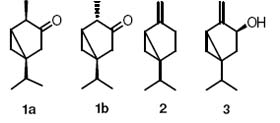
MAIN COMPONENTS OF THE ESSENTIAL OIL OF MENTA NATIVE
Marcelo Bascope,a,b* Lucio Alejob and Olov Sternera,
aDivision of Organic Chemistry, Lund University, P.O.Box 124, S-22100 Lund, Sweden.
bCentro de Tecnología Agroindustrial, Universidad Mayor de San Simón, Cochabamba Bolivia.
ABSTRACT
β-Thujone (1), sabinene (2), and sabinol (3) were identified as the major constituents of the essential oil of Menta native (identification in progress), with β-Thujone being present at a concentration higher than 90 %. The compounds were identified by NMR techniques and quantified by GC means. Remarkable is the fact that the isomer α-thujone is absent in the essential oil, plants containing α-Thujone and β-thujone are important herbal medicines and food additives, and their essential oils are appreciated in the commercial market.
Corresponding author: Marcelo.bascope@hotmail.com
RESUMEN.
β-Thujona (1), sabineno (2), y sabinol (3) se identificaron como los componentes mayoritarios del aceite esencial de Menta Nativa (identificación en progreso), con β-Thujona estando presente a una concentración mayor al 90 %. Los compuestos han sido identificados por técnicas de RMN y cuantificados por cromatografia Gaseosa. Es notorio el hecho que el isomero α-Thujona esta ausente en el aceite esencial, Plantas conteniendo α-Thujona y β-thujona son importantes medicinas hierbales y aditivos alimenticios, sus aceites esenciales son apreciados en el mercado comercial.
During the exploration of potential vegetal sources for the extraction of essential oils, we found that the aerial parts of Menta native (identification in progress) give a yellowish oil with strong mint fragrance. Preliminary GC studies of the essential oil shows the presence of one major component accounting for 90 % of the oil, and we decided to identify the components by NMR. We report in this paper the identification and structure determination of; β-thujone, sabinene, and sabinol as the major components of the essential oil of menta native. Plants containing α-thujone and β-thujone are important herbal medicines and food additives1.2.3., therefore their essential oils are appreciated in the commercial market. Both monoterpenes are isomers with different stereochemistry of the C-4 methyl group. α-Thujone is considered to be the principal active ingredient of wormwood oil and toxic principle of absinthe4. The content of β-thujone often exceeds that of α-thujone depending on the plant source, but the β isomer is considered to be less toxic5.

Figure 1. monoterpenoids from the family of the thujane skeleton.
RESULTS, DISCUSSION
The steam distilled oil of Menta native was analyzed by GC means, the chromatogram clearly shows three main constituent, whit concentrations of; 90.8, 6.1 and 0.7 % respectively, the remaining 3.6 % were 12 peaks which we didnt consider important for the characterization of the oil. The essential oil of was submitted to chromatography on straight phase, using hexane as eluent. The fractions were joined in three groups; fractions before the main constituent, containing the main component, and fractions after the main component. Its proton spectrum shows two proton signals (6-H2) at δ -0.09 ppm (dd, J=5.7, 4.10 Hz, H6-β) and δ 0.54 ppm (ddd, J=2.0, 5.7, 7.8 Hz, H6-α) typical from a cyclopropyl ring. The 13CNMR spectrum indicates a carbonyl group at δ 217.6 ppm, but no double bonds. Analysis of the 2D NMR spectra lead us to the structure of thujone, either 1a or 1b, and the stereochemistry was deduced from comparison of the experimental data with those reported in the literature 6, see Table 1. The main component of the essential oil of Menta native was consequently identified as β-Thujone (1b).
]]>
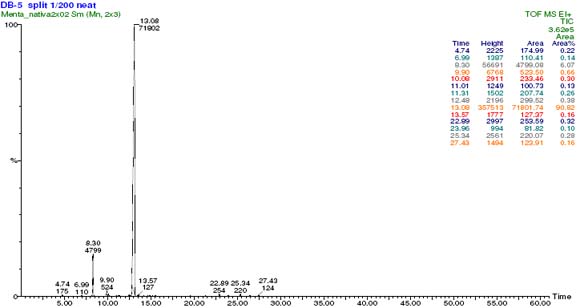
Figure 2. Gas chromatogram of the essential oil of the aerial parts of Menta native, showing mainly one constituent.
Compounds 2 and 3 present two geminal protons in an exocyclic double bond, in both situations the geminal protons do not present noticeable coupling, and they show up in the spectra as broader singlets than expected. The typical coupling constants for geminal double bonds vary between -4 and 4 Hz, and the range is reduced to -2.5 to 2.5 Hz for monosubstituted ethylenes whit no electronegative groups in the substituent, this reduced range explains the fact that the proton signals are observed as broad singlets. Other compounds containing exocyclic double bonds show the similar behaivor; Achilleol A7, Nakamurol A8 and Annulide.9 Compound 2 has two proton signals with the chemical shift δ 4.80 ppm (br s, H10 a) and δ 4.62 ppm (br s H10 b) corresponding to a exocyclic double bond C-10 δ 101.5, two methyl groups at C-8, C-9 δ 19.76 and 19.67 ppm with protons δ .95 ppm (d, J=6.8, Hz, 3H) and δ 0.88 ppm (d, J=6.8, Hz, 3H) respectively. A proton signal for H-6 at δ 0.65 (d, J=5.4 Hz, 2H) and C-6 at δ 16.0 ppm suggest the cyclopropyl moiety. Deep analysis of 2D NMR lead us to suggest the formula 2 which later was confirmed by literature reports10. Compound 3 present the same features of compound 2 and an additional proton signal at δ 4.40 ppm (d, J=7.4 Hz, H-1) and a 13C NMR signal at δ 75.1 ppm suggesting the presence of an hydroxyl group at the position 1. After analysis of 2D NMR we suggested the structure 3 which later was confirmed with literature reports. Noticeable is the high concentration of β-thujone in the essential oil and the relative absence of αthujone, whose presence couldnt be confirmed, Revision of commercial sources of the thujone isomers shows that it is possible to find α-thujone with a high purity but not β-thujone, where the best source is wormwood oil with 35 % content of β-thujone and 3-2 % of αthujone. This essential oil is therefore an attractive source for the β isomer.
Table 1. H and 13 C NMR of α-thujone and β-thujone

EXPERIMENTAL SECTION
]]> General GC analisys were performed using an Agilent Tecnologies Model 6890N Gas chromatograph, equipped with a DB-5 fused silica column (25x0.25 mm i.d., film thickness 0.25μm). Oven temperature was held at 60 C for 2 min and was raised up to 250 C at a rate of 5 C/min, where it was kept for 20 min. Injector temperature was 250 C and helium was used as carrier gas with a flow of 1.0 ml/min. The detector used was micromass GCT. 1H (400 MHz) and 13C NMR (100 MHz) spectra were recorded at room temperature with a Bruker DRX500 spectrometer with an inverse multinuclear 5 mm probehead equipped with a shielded gradient coil. The spectra were recorded in CDCl3, and the solvent signals (7.26 and 77.0 ppm, respectively) were used as reference. The chemical shifts (δ) are given in ppm, and the coupling constants (J) in Hz. COSY, HMQC and HMBC experiments were recorded with gradient enhancements using sine shaped gradient pulses. For the 2D heteronuclear correlation spectroscopy the refocusing delays were optimised for 1JCH = 145 Hz and nJCH = 10 Hz. The raw data were transformed and the spectra were evaluated with the standard Bruker XWIN-NMR software (rev. 010101).Plant material
The aerial parts of Menta native were collected in the location of Sapanani - Cochabamaba, Bolivia, during January 2003. The identification is in process, by the National herbarium Martin Cardenas Cochabamba, a voucher of the specimen is also deposited at the Centro de Tecnologia Agroindustrial.
Isolation procedure
Fresh aerial parts of menta nativa were subjected to hydro-distillation during 5 h. Steam was generated in a laboratory boiler to produce saturated steam at atmospheric pressure (565 mm Hg). Steam was conducted trough an isolated 10 lts cone chamber were the sample was placed. The oil was dried over anhydrous calcium chloride and stored at low temperature before analysis. The yield was 0.5 %. Each fraction was further purified by flash chromatography.
ACKNOWLEDGEMENTS
The authors are grateful to the Swedish International Development Agency (Sida) and its research division SAREC, for the economical support of this work.
REFERENCES.
1. Gruenwald, J.; Brendler, T.; Jaenicke, C., Eds. PDR for Herbal Medicines; Medical Economics: Montvale, NJ, 1998; pp 1181-1183. [ Links ]
2. Anonymous. Medicinal herbs: NTP extracts the facts. Environ. Health Perspect. 1999, 107, A604-A605.
]]>3. Blumenthal, M., Ed.; American Botanical Council: Austin, TX, 1998; pp 664-665. [ Links ]
4. Arnold,W.N.J. Am. Med. Assoc. 260, 3042-3044 [ Links ]
5. Hold, K; Sirisoma, N; Ikeda, T; Narahashi, T; Casida, J: Ï PNAS 97.8 (2000): 3826-31. [ Links ]
6. Sirisoma, Nilantha S.; Hoeld, Karin M.; Casida, John E. Journal of Agricultural and Food Chemistry (2001), 49(4), 1915-1921. [ Links ]
7. Barrero, A. F.; Alvarez-Manzaneda Roldan, E. J.; Alvarez-Manzaneda R., R. Achilleol A: Tetrahedron Letters (1989), 30(25), 3351-2. [ Links ]
8. Shoji, Noboru; Umeyama, Akemi; Teranaka, Miyuki; Arihara, Shigenobu. Journal of Natural Products (1996), 59(4), 448-50. [ Links ]
9. Brown, Geoffrey D. Annulide, a sesquiterpene lactone from Artemisia annua. Phytochemistry (1993), 32(2), 391-3.
10. Ferreira, M. J. P.; Emerenciano, V. P.; Linia, G. A. R.; Romoff, P.; Macari, P. A. T.; Rodrigues, G. V. Progress in Nuclear Magnetic Resonance Spectroscopy (1998), 33(3,4), 153-20. [ Links ] ]]>