OLEANANE-TYPE TRITERPENES AND DERIVATIVES FROM SEED COAT OF BOLIVIAN CHENOPODIUM QUINOA GENOTYPE “SALAR”
Yonny Flores a,b, Carlos Díaz a, Fidel Garay a, Osmar Colque c, Olov Sterner b and Giovanna R. Almanza a,*
aLaboratorio de Productos Naturales, Instituto de Investigaciones Químicas, Universidad Mayor de San Andrés, Calle 27 Cota Cota, P.O.Box. 303 La Paz-Bolivia.
bDepartment of Organic Chemistry, Lund University, P.O.Box 124, SE-22100 Lund-Sweden.
cIrupana Andean Organic Food S.A., Av. Ecuador Nº 2249, La Paz-Bolivia
*Corresponding author: giovyalmanza@yahoo.com.ar
]]>
ABSTRACT
Bolivia is the first world exporter of quinoa (Chenopodium quinoa Willd) genotype “salar”. In its natural state quinoa has a coating of bitter-tasting (saponins), making it essentially unpalatable. Most of quinoa exported from Bolivia has been processed to remove this coating. These seed coat residues contain a high level of oleanane-type saponins. From those residues two oleanane-type triterpenes were found as the major sapogenins and their structures were identified as: oleanolic acid and deoxyphytolaccagenic acid in base of NMR means. Furthermore, two derivatives of oleanolic acid were obtained: 28-Omethyl and 3-O-acetyloleanolic acid. The importance of oleanolic acid content in this industrial residue is discussed.
Key words: Chenopodium quinoa, sapogenines, oleanolic acid, deoxyphytolaccagenic acid, oleanolic acid derivatives, Bolivian highland crops.
Bolivia es el primer exportador de quinua (Chenopodium quinoa Willd) genotipo “salar” del mundo. En su estado natural las semillas de quinua tienen una cobertura amarga (saponinas), que causan un sabor desagradable. Por esto, la mayor parte de quinua exportada es previamente procesada para separar esta cobertura. Estos residuos de escarificación contienen un alto nivel de saponinas tipo oleanano. De estos residuos, dos triterpenos tipo oleanano fueron encontrados como sapogeninas mayoritarias y sus estructuras fueron identificadas como ácido oleanólico y ácido deoxifitolaccagénico, principalmente en base a técnicas de RMN. Además, dos derivados del ácido oleanólico fueron obtenidos: los ácidos 28-O-metil y 3O-acetiloleanólico. La importancia del contenido ácido oleanólico en estos residuos industriales es discutida.
Quinoa (Chenopodium quinoa Willd) is a pseudocereal which grows at elevations up to 5.000 m.a.s.l. in the Andean region of South America. Its grains have been used as food in the region since more than 5.000 years ago. For the Indian natives quinoa seeds have been more valuable than gold [1]. Nowadays, there is renewed interest in quinoa because it contains more protein than any other grain. In fact, it is considered a complete protein since it contains all eight essential amino acids [2]. For this reason it has a socioeconomic impact for the Andean people, particularly it is very important in Bolivia which is the main supplier country in the world.
This plant has an important genetic variation in agreement of its environmental conditions producing several genotypes grouped in five: “Sureño” which grows in the South of Chile; “Yungueño” which grows in “Los Yungas” beginning the Amazonic region; “Valle” which grows in the valleys of Colombia, Ecuador, Peru and Bolivia; “Altiplano” which grows in the border of Lake Titicaca and “Salar” which grows in the saline soils of the Bolivian Highlands. The last one is the most important since the economic point of view because it produces the biggest grains for export [3]. Those grains have high levels of saponins in their coat which are the responsible of their bitter flavour but also of the protection of this plant against noxious insects [4].
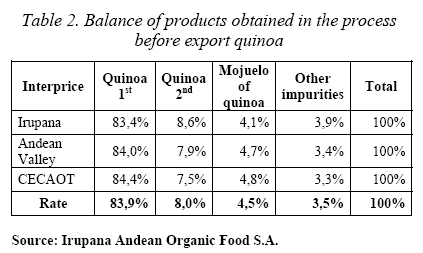
The bitter principles of quinoa must be removed before export or cooking. For export, the food Bolivian industries removed the seed coat by two processes: the first one is a mechanic process (dry way) where the grains are rubbed among them producing a residue called “mojuelo” in a percentage of 4.5%; the second process is done with water (wet way) to wash the last bitter impurities (Irupana Andean Organic Food S.A.).
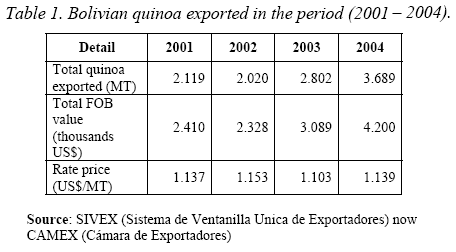
The increasing attention in these grains raised the exports of this product in Bolivia, with the consequent increase in the production of residues (Tables 1 and 2).
]]>
The residues of the dry process were previously analyzed doing around 50% of saponins (source Irupana Andean Organic Food S.A.). Other studies [4,5,6] showed that one of the main sapogenins in seeds of quinoa is the oleanolic acid. On the other hand, the oleanolic acid was widely studied for several research groups determining that it is a valuable compound possessing several biological and pharmacological activities [7,8]. In fact, our research group found that it is one of the major secondary metabolites in Bolivian Highland Plants which plays an important role against the environmental conditions [9]. For its pharmacologic properties the cost of oleanolic acid is going up, costing today approximately 60 U$D per 100 mg (97% pure) (Sigma Aldrich), being already commercialized in China as drug for liver disorders [7].
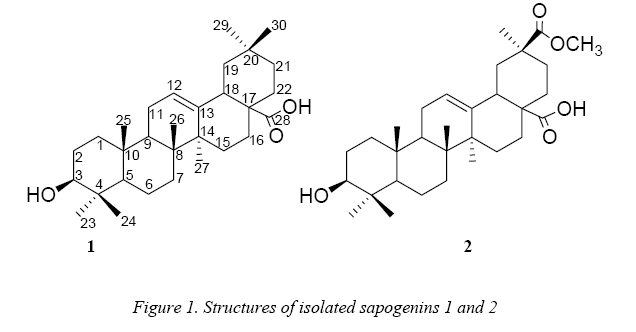
All of those things made us to think in study the presence of sapogenines in the dry residues of quinoa real for export, genotype “salar”, to see their potential uses or benefices. In this sense, herein we report the separation and identification of two oleanane-type triterpenes: oleanolic acid (1) and deoxyphytolaccagenic acid (2), as well as the synthesis of two derivatives of the obtained oleanolic acid: 28-Omethyl and 3-O-acetyloleanolic acid, as one of the potential chemical uses of the oleanolic acid.
RESULTS AND DISCUSSION
1000 g of the industrial residue of Chenopodium quinoa Willd genotype “salar” was extracted with EtOH/H2O (75/25). The obtained EtOH extract was subjected to an acid hydrolysis with HCl giving 38 g of sapogenin extract. So, the total sapogenin content in this industrial residue is very high (3.8%), this result is in agreement with other studies that showed the high content of saponins in the seed coat of quinoa [4,6].
]]> 3,8 g of sapogenin extract was subjected to a chromatographic analysis giving the isolation of two compounds: Compound 1 (1,4 g) and compound 2 (375 mg). Compound 1 was identified as oleanolic acid by comparison of NMR data (Table 3) with bibliographic data [9], as well as by comparison in TLC with authentic patrons of laboratory.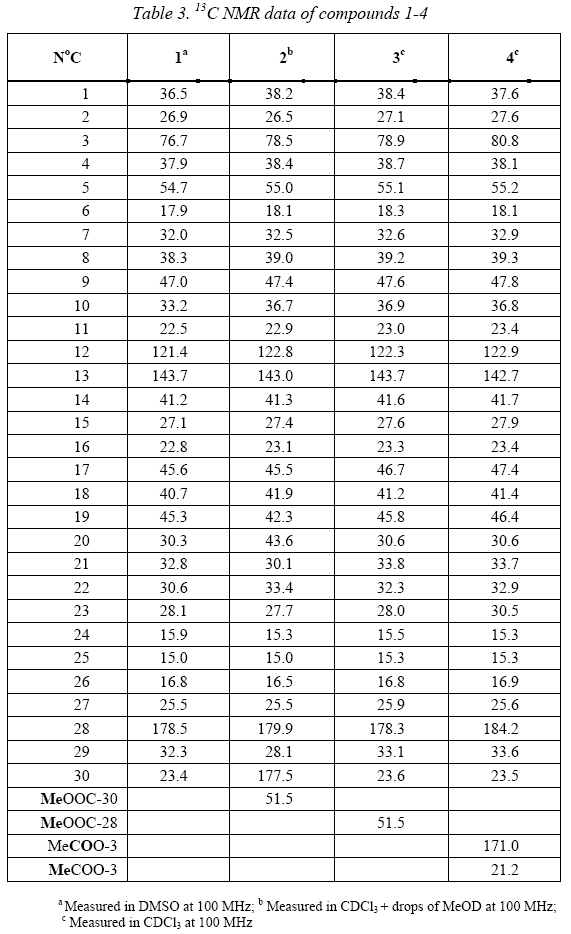
Compound 2 showed similar 1H and 13C NMR spectra that compound 1. In agreement of previous reports [6] the major sapogenins in seeds of Chenopodium quinoaare: the oleanolic acid, hederagenine, 30-O-methylspergulagenate and phytolaccagenic acid. So, at the first time we though in these compounds but the study of NMR data showed that the isolated compound was the deoxyphytolaccagenic acid 2.
13C NMR signals of compound 2 show a great similarity with those of 1 except by the presence of an extra ester group at δ 177.5 and 51.5. This ester group was located at C-30 because of its effect on carbons C-19, C-20, C-21 and C-30 (Table 3). Particularly is highlighted the shifted downfield effect of the ester group in C-20, suggesting a direct substitution in this position. This substitution was confirmed by HMBC correlations (Figure 2) which showed the long-range coupling of carbonyl C-30 (δ 177.5) and the methyl protons at C-29 (δ 28.1) which also coupled with the carbons C-19, C-20 and C-21 in the HMBC spectrum. This substitution just could be also located at C-4, but the long-range correlations among the methyl protons at C-23 (δ 27.7) with the carbons C-3, C-4, C-5 and C24 finally confirmed the proposal structure. In addition, the 13C NMR data was compared with bibliographic data [5].
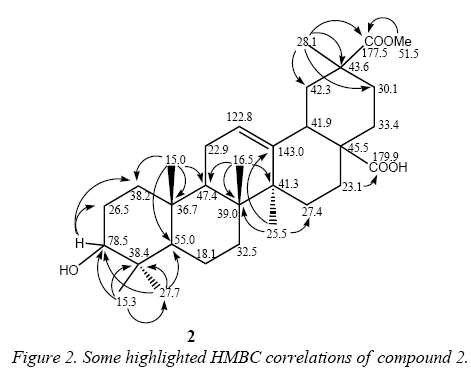
We know that the export of quinoa in Bolivia is going up, arriving to 3.689 TM in 2004 (Table 1) with the consequent increment in the production of residues (Table 2). So, it is very important, since the environmental and economic point of view, to think in use the industrial residues. In this study of sapogenins present in the industrial residue called “mojuelo” we obtained a very good yield of oleanolic acid 1 (1,4% table 4).


In addition, the oleanolic acid was submitted to several chemical modifications mainly to study the pharmacological effect of its derivatives [12,13].. So, thinking in complement our chemical study of the compound 1, we obtained the 28-O-methyl and 3-Oacetyl derivatives of oleanolic acid (3 and 4).
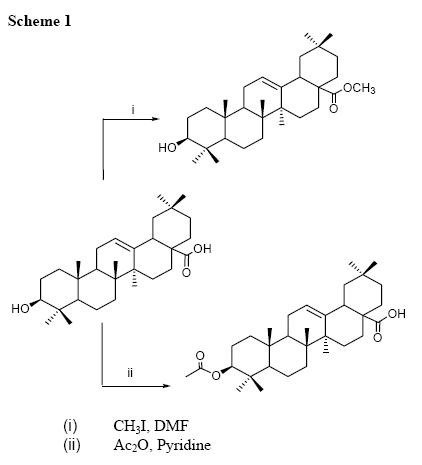
The structures of derivatives 3 and 4 were identified based on the 1H NMR (Figure 3) and 13C NMR (Table 3) spectra. So, table 3 shows that the 13C NMR signals of compound 3 are very similar to those assigned for 1, the little differences could be caused by the change of solvent, because the compound 3 was soluble in CDCl3 instead of 1 which was not soluble in it. The main difference in the NMR data was the presence of a methoxy group (MeO) at 51.5 ppm in 13C and 3.58 ppm in 1H NMR spectra. This methoxy group could be located in the hydroxyl group at C-3 or in the acid group at C-28. Its 1H NMR spectrum did not show an effect of this group over H-3 (Figure 3), so it was located in the group at C-28. This substitution was confirmed with HMBC correlations whish showed a long range coupling between the methyl protons of the methoxy group and the carbonyl C-28 at δ 178.3.
Compound 4 was clearly identified as 3-Oacetyloleanolic acid, because the 1H NMR spectrum (Figure 3) shows a high downfield shifted on H-3, so H-3 appears at δ 4.5 as a typical proton geminal to an acetate group, in addition one acetate methyl group is observed at δ 2.05, confirming the acetylation of compound 1. The 13C NMR data of 4 (Table 3) is in agreement with the established structure showing also the downfield shifted effect on C-3 (δ 80.8) and the acetyl group at δ 171.0 and 21.2.
MATERIALS AND METHODS
General Experimental Procedures.
]]> Crude extracts were concentrated under vacuum (Labora Rota Evaporator), 1H and 13C NMR spectra and two-dimensional experiments were recorded in a Bruker DRX-400 NMR spectrometer, operating at 400 MHz for 1H and 100 MHz for 13C. The raw data were transformed and the spectra were evaluated with the standard Bruker XWIN-NMR software package; chemical shifts are reported in δ units (ppm) and coupling constants (J) in Hz, using DMSO d6, CDCl3 and MeOD as the deuterated solvents and their residual solvent signals as reference, 13C-DEPT, HMQC, and HMBC experiments were carried out using the conventional pulse sequences described in the literature. The Vacuum Liquid Chromatography (VLC) was performed on silicagel G-60. The CC column chromatographies were performed on Silicagel 60 (70230 mesh) and TLC analyses were made on Silicagel F 254 (Merck) plates and visualized with sulphuric acid and heating. The organic solvents were used after purification by distillation, the other inorganic and organic reagents used were all commercially guaranteed. Plant materialThe plant material “mojuelo de quinua real” is an industrial residue of Chenopodium quinoa Willd genotype “salar” for export. This material was supplied by the Bolivian Industry Irupana Andean Organic Food S. A.. The plant was collected in Bolivian Highlands near to “Salar de Uyuni” (Prov. Ladislao Cabrera, Oruro) at 3.725 m.a.s.l. of altitude. The residue was obtained by an industrial mechanic process to remove the seed coat of quinoa before export. It looks as fine white powder with bitter flavour.
Extraction and acid hydrolisis of saponinsThe white powder of “mojuelo de quinua real” (1000g) was extracted by maceration with EtOH/H2O (75/25) for 72 h at room temperature with solvent renovation. The obtained extract was rotaevaporated until to obtain 1.6 L of a concentrated EtOH extract. The last extract was submitted to an acid hydrolysis adding 320 mL of HCl 2N, this solution was refluxed for 4h giving a reaction mixture constituted by two phases. After being cooled the two phases were separated and the solid phase was washed with H2O and then extracted with AcOEt. The AcOEt layer was dried with anhydrous Na2SO4 and evaporated to dryness giving 38 g of sapogenin extract.
Isolation of sapogenins 1 and 23,8 g of the AcOEt sapogenin extract was subjected to VLC chromatography on Silicagel G-60 using mixtures of AcOEt and petrol ether, in increased order of polarity, to give 5 fractions (C-I to C-V). The fractions were compared by chromatography TLC with patron samples of oleanolic acid from the Lab showing that it was present in the fractions C-II and C-III. Fraction C-II was submitted to a crystallization process in MeOH giving 850 mg of compound 1. Fraction CIII was submitted to a further CC chromatography on Silicagel 60 (70-230 mesh) eluted with AcOEt : petrol ether 50:50 giving, after purification by recrystallization in MeOH, 550 mg of compound 1 and 375 mg of compound 2
Compound 1White needles: M.P. > 312 ºC; C30H48O3; FW 456,7; 1H RMN (DMSO, 400 MHz) δ 5.16 (1H, t, H-12), 2.99 (1H, t, H-3), 2.73 (1H, dd, H-18), 1.90 (1H, ddd, H16a), 1.81 (2H, d, H-11a y H-11b), 1.08 (3H, s, Me), 0.90 (3H, s, Me), 0.87 (6H, s, 2 x Me), 0.85 (3H, s, Me), 0.73 (3H, s, Me), 0.68 (3H, s, Me); RMN13C (DMSO, 100 MHz) see Table 3.
Compound 2
White needles: M.P. > 312 ºC, C31H48O5; FW 500.71; 1H RMN (CDCl3+CD3OD, 400 MHz) δ 5.21 (1H, t, H12), 3.06 (1H, dd, H-3), 2.54 (1H, dd, H-18), 1.75 (1H, ddd, H-16a), 1.01 (6H, s, Me-29 and Me-24), 0.84 (3H, s, Me-27), 0.77 (6H, s, Me-25), 0.77 (6H, s, Me-26 and Me-23), 3,57 (3H, s, Me ester 30); RMN13C (CDCl3+MeOD, 100 MHz) see Table 3.
Methylation of compound 1The methylation of compound 1 (200 mg) was done with CH3I in presence of DMF for 12 h at room temperature (22ºC) giving a product. The product was purified by flash CC on Silicagel using CHCl3 as solvent to give the methyl ester of oleanolic acid 3 (160 mg, 80%).
Derivative: Me ester, compound 3
White powder: M. P.: 197-199°C; C31H50O3; FW 470.73; 1H NMR (CDCl3, 400 MHz) δ 5.26 (1H, t, H12), 3.20 (1H, dd, H-3), 2.84 (1H, dd, H-18), 1.11 (3H, s,Me), 0.97 (3H, s, Me), 0.91 (3H, s, Me), 0.89 (3H, s, M), 0.88 (3H, s, Me), 0.76 (3H, s, Me), 0.70 (3H, s, Me), 3,61 (3H, s, Me ester 30); 13C NMR (CDCl3, 100 MHz) data see Table 3.
Acetylation of compound 1 ]]> The acetylation of compound 1 (200 mg) was done with pyridine (2 ml) and acetic anhydride (2 ml) at room temperature (22 ºC) overnight and then evaporated to dryness after addition of toluene (3x5 ml). The product was purified by flash CC on Silicagel using CHCl3 as solvent to give the acetyl-oleanolic acid 4 (120 mg 60%).Derivative: Ac, compound 4
White powder: M.P. 165-167 ºC; C32H50O4; FW 498.74; 1H NMR (CDCl3, 400 MHz); δ 5.24 (1H, t, H12), 4.47 (1H, dd, H-3), 2.79 (1H, dd, H-18), 1.12 (3H, s,Me), 0.91 (3H, s, Me), 0.90 (3H, s, Me), 0.84 (3H, s, M), 0.83 (3H, s, Me), 0.79 (3H, s, Me), 0.72 (3H, s, Me), 2.02 (3H, s, MeCOO); 13C NMR (CDCl3, 100 MHz) data see Table 3.
ACKNOWLEDMENTS
We are indebted to SIDA/Sarec for financial support of the project “Research on Plant Biodiversity” and we also would like to thank to Irupana Andean Organic Food S.A. for all the information about quinoa business in Bolivia.
1. Tapia M.; Cultivos andinos subexplotados y su aporte a la alimentación. FAO, Santiago de Chile, (1990) [ Links ]
2. Koziok M. J.; Chemical composition and nutritional evaluation of quinoa (Chenopodium quinoa Willd); Food Comp. Anal., 5, 35-68, (1992). [ Links ]
3. Tapia M., Gandarillas H., Alandia S., Cardozo A, Mujica A., Ortíz R., Otazú V., Rea J. y Zanabria E.; La quinua y la kañihua. Serie cultivos andinos. CIID/IICA, Bogotá, (1979) [ Links ]
4. Mizui, F.; Kasai, R; Ohtani, K.;Tanaka, O.; Saponins from the bran of quinoa, Chenopodium quinoa Willd. II; Chem. Pharm. Bull, 38, 375-377, (1990) [ Links ]
5. Mizui, F.; Kasai, R; Ohtani, K.;Tanaka, O.; Saponins from the bran of quinoa, Chenopodium quinoa Willd. I; Chem. Pharm. Bull,36, 1415-1418, (1988) [ Links ]
6. Mastebroek H.D., Limburg H., Gilles T., Marvin H., Ocurrence of sapogenins in leaves and seeds of quinoa (Chenopodium quinoa Willd), J. Sci. Food Agric., 80, 152-156, (2000) [ Links ]
7. Liu J., J. Ethnopharmacol., 49, 57, (1995) [ Links ]
8. Hye Gwang Jeong, Inhibition of cytochrome P450 2E1 expression by oleanolic acid and its hepatoprotective effects against carbon tetrachloride-induced hepatic injury, Toxicology, 105 215-222, (1999) [ Links ]
9. Flores Y., Sterner O., G. R. Almanza, Oleanoic acid, presence and importance in highland bolivian plants, Rev. Bol. Quim. 21, 31-35, (2004) [ Links ]
10. García-Granados López de Hierro A., Patent US1998000043318, Universidad de Granada, Spain, (2000)
11. Gerold Gruenwald J., Patent US2001000024978, E. & J. Gallo Winery, Germany (2004) [ Links ]
12. Chao-Mei Ma et al, Chemical Modification of Oleanane Type Triterpenoids and Their Activity Against HIV-1 Protease Dimerization, Chem. Pharm. Bull, 48 (11), 1681-1688, (2000) [ Links ]
13. Kashiwada Y., et al.; Anti-AIDS Agents 30. Anti-HIV Activity of Oleanolic Acid and Structurally Related Triterpenoids, J. Nat. Prod, 61, 1090-1095, (1988) [ Links ]
]]>