Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Investigación & Desarrollo
versión impresa ISSN 1814-6333versión On-line ISSN 2518-4431
Inv. y Des. vol.18 no.1 Cochabamba 2018
http://dx.doi.org/10.23881/idupbo.018.1-6i
ARTÍCULOS – INGENIERÍAS
ANALYSIS OF MINING ORE CONCENTRATES WITH A LOW COST PORTABLE LIBS SYSTEM
ANÁLISIS DE CONCENTRADOS MINEROS CON UN SISTEMA LIBS PORTABLE DE BAJO COSTO
Omar Ormachea1, Alex Villazón2 and Iván Terceros1
1Centro de Investigaciones Ópticas y Energías (CIOE)
2Centro de Investigaciones de Nuevas Tecnologías Informáticas (CINTI)
Universidad Privada Boliviana
(Recibido el 10 de mayo 2018, aceptado para publicación el 15 de junio 2018)
ABSTRACT
Identifying minerals in ore concentrates require costly techniques and preparation of samples, thus limiting their instant analysis. We present the development of a low-cost portable LIBS (Laser Induced Breakdown Spectroscopy) system, to identify atomic elements in ore concentrates in-situ and in real-time. The system consists of a YAG:Nd+++ pulsed laser, an optical diffraction spectrometer, and a measurement gun consisting of a laser head and an optical system for collecting light from the sample. We developed customized software for data acquisition, processing and analysis that stores and uses the principal spectral lines of referential atomic elements (Cu, Pb, Ag, Au, Li, Zn). We experimentally obtained spectra of selected samples using our LIBS system, and successfully matched them with the referential spectral lines. Finally, we tested our system with real samples of ore concentrates from a mining company, where the presence of Zn was successfully detected through its characteristic spectrum.
Keywords: LIBS, Atomic Emission Spectroscopy, Lasers, Spectrometers.
RESUMEN
La identificación de minerales en concentrados, requiere de técnicas costosas y preparación previa de muestras, lo que limita su análisis inmediato. En este artículo presentamos el desarrollo de un sistema LIBS (Laser-Induced Breakdown Spectroscopy) portátil y de bajo costo, para la identificación de elementos atómicos en concentrados mineros in-situ y en tiempo real. El sistema está compuesto por un láser pulsado del tipo YAG:Nd+++, un espectrómetro de difracción óptico; y una pistola de medición compuesta por el cabezal laser y un sistema óptico para la recolección de luz. Se desarrolló un software de adquisición, análisis y procesamiento de datos donde se almacenaron las líneas espectrales principales proveniente de fuentes referenciales, para los elementos atómicos seleccionados (Cu, Pb, Ag, Au, Li, Zn). Se compararon los espectros obtenidos experimentalmente con nuestro sistema, con los datos de referencia, encontrando una afinidad espectral alta. Finalmente, se realizaron pruebas en muestras reales de concentrados de una empresa minera, donde se detectó la presencia de Zn analizando el espectro característico obtenido.
Palabras Clave: LIBS, Espectroscopia de Emisión Atómica, Láseres, Espectrómetros.
1. INTRODUCTION
LIBS (Laser Induced Breakdown Spectroscopy) is a type of atomic emission spectroscopy induced by laser radiation [1]. LIBS is a very powerful technique for the real-time analysis of the atomic composition of a wide variety of materials, from metals and geochemical samples to biological agents [2, 6].
A LIBS system consists of a laser pulse of high intensity which is focused towards the sample to be analyzed, and positioned between a few centimeters to one meter of the sample. A laser pulse of 10 ns of life time and 10 mJ of energy are enough to excite a sample [7]. When the laser is fired, the high temperature of the concentrated pulse produces plasma. As the plasma cools (~1.0 µs after the laser shot), excited atoms in the plasma emit light with particular wavelengths that distinguish each atomic element of the periodic table, where practically all of them emit in the region 200-980 nm [1].
The LIBS technique has several advantages over other real-time techniques, such as X-ray fluorescence [8], or the Prompt-Gamma Neutron Activation Analysis (PGAA) [9]. For example, one of the main disadvantages of X-ray fluorescence in industrial applications, is that it cannot detect elements with an atomic number lower than Z = 16, in contrast to the LIBS technique that does not have this restriction. Likewise, PGAA has the disadvantage that a rigorous environmental control and nuclear health is needed, not required for LIBS. Therefore, LIBS is very attractive technique for identification and quantification control in-situ and in real-time of atomic elements in the mining industry [10]. In addition, the use of the LIBS technique does not require prior preparation of the sample and detection sensitivity can reach up to parts per million (ppm) [11].
In this article, we present the implementation of a low-cost and portable LIBS system and its application to the Bolivian mining industry. This work is based on previous research results in the development of pulsed lasers [12] and low-cost optical spectrometers [13, 14]. The portable LIBS functional prototype is based on a YAG:Nd+++ laser and a reflection optical spectrometer [14, 16].
Currently, in Bolivia 5 minerals represent 93.3% of the export of mining concentrates: Zn (36,5%), Ag (28,7%), Sn (15,0%), Au (7,4%) y Pb (5,7%), generating around 3,8 billion dollars in 2012 [17], constituting a very important percentage of the Bolivian Gross Domestic Product (GDP), equivalent to 14.3% [18].
The mining industry in Bolivia is largely composed by mining cooperatives. Minerals are exported mainly among mining ore concentrates, and companies declare the material and the percentage that is exported. Unfortunately, there is no rigorous government control of additional minerals that may be present in ore concentrates, such as Europium, Indio, Neodymium or others that are very expensive in international markets.
In order to respond to this need in the Bolivian mining industry, we propose the development of a completely portable and energy-autonomous LIBS system (Figure 1), which can be controlled wirelessly and has its own control software for the detection of minerals in mining concentrates in-situ and in real-time.
The rest of the article is structure as follows: Section 2 describes the complete proposed LIBS system. Section 3 presents the data acquisition scheme. In Section 4, we describe the software that we developed and the data processing. Results with real ore concentrates are presented in Section 5 and Section 6 shows an estimation of prototype costs. Section 0 concludes the article.
2. PORTABLE LIBS SYSTEM
The proposed portable LIBS system includes an optical spectrometer, an electronic power supply and control of the pulsed laser of the YAG type: Nd+++, and a measuring gun that collects the light produced by the plasma on the sample and sends it through a fiber optic cable to the spectrometer. Figure 2 shows the developed portable LIBS system.
2.1 OPTICAL SPECTROMETER
The developed optical spectrometer is based on a monochromatic Chameleon CCD camera of 1.2 MP (spectral range of sensitivity from 400-900 nm) as optical sensor and a reflective diffraction grating (1200 lines per millimeter and a size of 25x25mm) [19]. This diffraction grating is mounted in an opto-mechanical positioning system. The electronic control is carried out through a wireless system (Bluetooth), which provides a stepped adjustment of 4 spectral windows with steps of 80 nm, with a spectral resolution of 0.3 nm. This allows a spectral range of 400-720 nm, corresponding to the visible range of the electromagnetic spectrum [20] (see Figure 3).
2.2 ELECTRONIC POWER SUPPLY
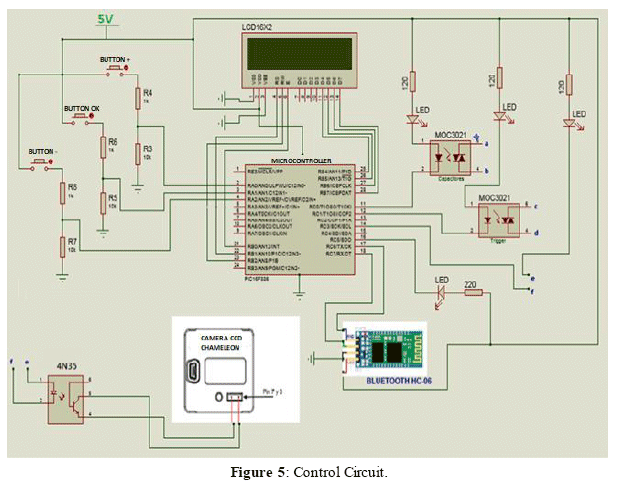
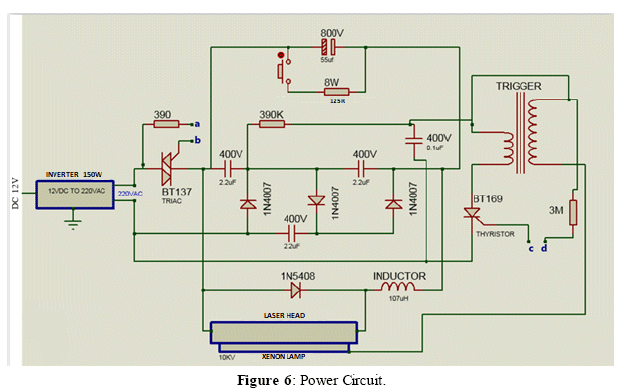
The power supply of the laser is composed of a lithium polymer battery of 18.5 V and 3300 mAh, regulated by a voltage regulation circuit that uses DC-DC Step Down converters (shown in Figure 4), and an electronic circuit of power and control (manual and wireless). The electronic circuit allows selecting the energy and firing frequency of the pulsed laser, values that can be visualized on the LCD 16x2 display and the selected parameters are saved and controlled by a microcontroller (shown in Figure 5). The power system consists of a capacitor block with a capacitance of 55 µF, 800 VDC and maximum storage energy of 17.6J (shown in Figure 6).
The pulsed laser is based on a crystal of the YAG:Nd+++ type, (l=1064 nm) that works in the passive Q-switch regime. A trigger system based on an external trigger scheme was chosen and the firing frequency of the laser system is 0.16 Hz. The pulse has a duration time and estimated power of: t=10 ns, P=10 MW/cm2 [7].
2.3 MEASURING GUN
Inside the measuring gun is located the laser head and an optical system for collecting the light produced by the laser ablation in the examined material.
The design of the measuring gun allows focusing the laser radiation on the sample, which simply approaches the light insertion hole. The optical design of the measuring gun is configured in such a way that the viewing plane of the light collection system coincides with the laser focus point and the position of the sample (Figure 7). After the laser shots the sample, the measuring gun transports the emitted light emitted to the spectrometer through an optical fiber cable for further spectral analysis. Finally, between the laser source and the measuring gun, there is a power cable that transports the electrical energy and the control pulses to the laser head for firing.
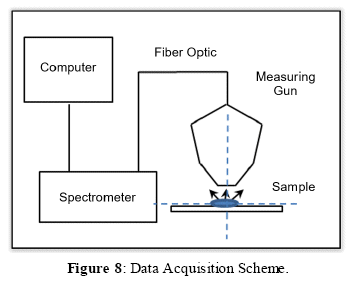
3. DATA ACQUISITION SCHEME
For the sampling of different materials, as shown in Figure 8, the following data acquisition scheme was used:
The samples to be analyzed can be raw solid samples or be pulverized (as samples of the mining concentrates are usually available). Since the measuring gun is directed downwards, the geometry of the proposed scheme allows the sampling and analysis of pulverized samples, which are deposited in a glass base and the laser shot is made from the upper position, as shown in Figure 8.
Figure 9 shows a real sample of a pulverized mining concentrate. We can observe that after several laser shots, a mini-crater is generated in the sample due to the intense electro-magnetic radiation.
4. ANALYSIS AND PROCESSING SOFTWARE
Processing software was developed for the acquisition, processing and control of the spectrum produced by the samples analyzed. The software controls a Chameleon CCD digital camera and processes the image, which is captured at the moment that the light coming from the sample ablated by the laser radiation.
The program has a database of referential spectra of some elements of the periodic table. These references were taken from the National Institute of Standards and Technology [21] and the Optical Science Center for Applied Research [22]. The developed software compares this database (main spectral lines of atomic elements), with the spectra obtained by the proposed LIBS system. These operations allow visual comparison of coincidence and qualitatively identify the existence of the element in the analyzed sample.
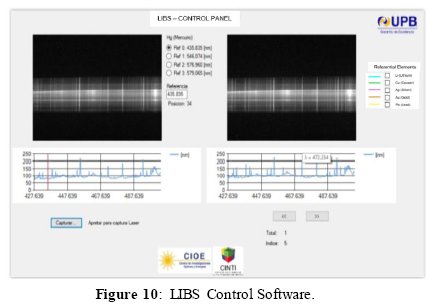
Figure 10 shows the main window of the control panel of our LIBS software. The program allows to see in real time the images captured from the camera. It also allows calibration of the spectrometer according to the wavelength. Finally, it is possible to select elements of the database and perform the processing of the images to obtain the characteristic spectra of the analyzed samples and compare them with the referential data. For the calibration of the spectrometer, we use a fluorescent lamp containing mercury. As seen in Figure 11(a), the user can select different main lines of mercury, which correspond to specific wavelengths. Each of these lines allows calibrating an 80 nm spectral window in the spectrometer.
Once the spectrometer is calibrated, the use can select a referential element from the database (see Figure 11(b)), and the laser pulse is triggered on the sample to be analyzed. The software generates the main lines of the spectrum of the chosen element, and the spectrum obtained from the real sample. In this way, one can visually see the correspondence of the elements spectral lines from the sample ablated by laser radiation.
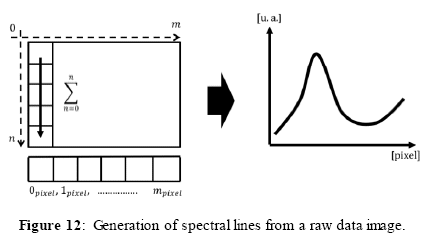
To generate and display the spectra, the images are captured from the digital camera in raw format, and processed by the software as a pixel matrix (with n columns and m rows). Figure 12 shows the generation of spectral lines from a raw image. To obtain the spectral lines from the image, for each column of the image matrix, we sum the RGB (Red, Green, Blue) values into a single data array of size n (the RGB values are already in gray scale as we use a monochromatic camera). The obtained array contains the spectrum as a set of values of the intensity of signal for each pixel in the x-axis, in arbitrary units [u.a.]. The software plots the values of the array as a continuous line representing the spectrum. Since positioning the camera and the diffraction grating in a perfect horizontal line is difficult, we use a control reference to verify the correct position.
The calibration step based on the reference values allows to associate wave-length values to each index of the array. A minimum of two reference values are needed to extrapolate the wave-length values for each pixel in the x-axis. Because the camera sends images at a rate of 30 fps (frames per second), and the processing of every images can be time consuming, we apply two optimizations: one consisting in analyzing only the central part of the image (where the signal is more intense), and the other is to analyze the whole image, but skipping some of the rows. In both cases, we noticed similar results, without affecting the obtained results. For convenience, the images and their spectra can be recorded for post-mortem analysis.
The software runs on Windows and was implemented in C# using Chameleons Software Development Kit (SDK)[1] drivers and Application Programming Interfaces (API) to control de camera from the software.
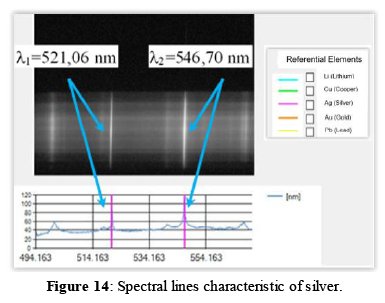
For the validation of the proposed LIBS system, we carried out tests with standard samples of atomic elements. For example, Figure 13 shows a sample of silver used for testing, where shots were made with the laser device and the corresponding LIBS spectra were obtained that can be seen in Figure 14.
In the control software, we select the atomic element to be analyzed (i.e. Ag) from the database and the corresponding reference lines are displayed on top of the obtained spectrum. The software compares the actual spectrum obtained with the reference lines of the database (i.e. wavelengths l1=521,06 nm and l2=546,70 nm for silver [22]). It can be clearly seen in Figure 14, the coincidence of the spectral lines obtained with our LIBS system with the reference lines.
In addition, satisfactory tests were performed with gold, lead, copper and lithium standards to validate the proposed LIBS system.
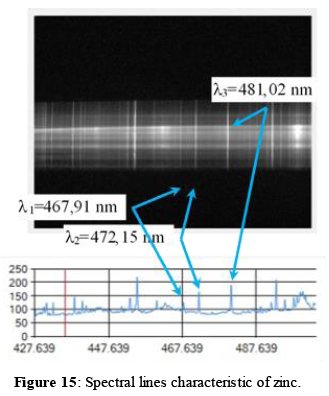
The measurements on real samples from a mining company where performed using an ore concentrate containing zinc in a percentage of 2.44% (Figure 9). This value was measured by the company providing the samples, with the atomic absorption technique. Once the sample was ablated with our LIBS system and the information was processed, the respective emission spectrum was obtained. Figure 15 shows the obtained lines corresponding to the main zinc wavelengths (l1=467,91 nm, l2=472,15 nm and l3=481,02 nm [22]). The other lines obtained in the spectrum (on both sides of the zinc lines) correspond to other atomic elements present in the matrix containing the sample analyzed, which are not relevant for the present study.
The results obtained experimentally with real samples, demonstrate the validity of the proposed LIBS system, for the identification of atomic elements of interest in mining concentrates in real time (Zn in our case study).
6. COST ESTIMATION OF THE PROTOTYPE
For the construction of the proposed LIBS system, all non-specialized electronic and optical components (i.e. capacitors, micro-controllers, photographic lenses, focusing lenses) are easily acquired in local commerce, which drastically reduces the costs.
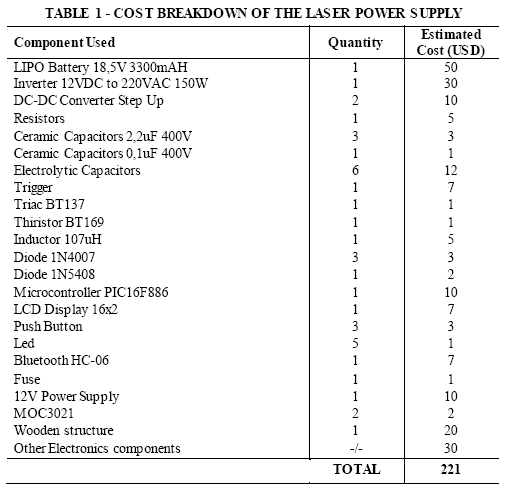
TABLE 1, TABLE 2 TABLE 3, show the list of components that were used to build every part of the proposed LIBS system. TABLE 1 describes the electronic components of the laser power supply and control. TABLE 2 shows the elements used to build the spectrometer together with its wireless electronic control used to positioning the diffraction grating. TABLE 3 shows the elements required to build the measuring gun, composed by the laser head (controlled by the electronic power supply), and the components to collect the light from the analyzed samples.
The total cost of the prototype is less than 2000 USD, which does not includes research and development of the software and hardware of the proposed LIBS system. Note that existing portable commercial LIBS systems are at least 20X more expensive (between 20000 and 60000 USD [23, 24]). Also, it would be possible to use a commercial laser and/or spectrometer in our prototype. However, such commercial products do not include the source code of their software, and the licenses are also costly (~15000 USD for a commercial laser system [25] and 2000 USD for a basic spectrometer [26]).
7. CONCLUSION
The work presented in this article describes the development of a low cost portable LIBS system, for the identification of atomic elements in mining ore concentrates, and its application in the Bolivian mining industry. We also described our control and analysis software that processes the data produced by the LIBS system, and provides features for calibrating the device and identifying atomic elements. Identification tests were carried out with copper, lead, silver, gold and lithium. Our case study shows that the proposed device successfully identified zinc in real sample of a mining ore concentrates. The proposed combination of self-made optical elements and software into a compact and low-cost LIBS system is an initial step towards the availability of LIBS devices that are universally accessible, portable, and easy to use.
8. REFERENCES
[1] D. A. Cremers y L. J. Radziemski, Handbook of Laser-Induced Breakdown Spectroscopy, Wiley, 2006.
[2] J. D. Hybl, A. G. Lithgow y G. S. Buckley, «Laser-induced breakdown spectroscopy detection and classification of biological aerosols,» Appl. Spectrosc., vol. 57, pp. 1207-1215, 2003.
[3] A. I. Whitehouse, J. Yong, I. M. Botheroyd, S. Lawson, C. P. Evans y J. Wright, «Remote material analysis of nuclear power station steam generator tubes by laser-induced breakdown spectroscopy,» Spectrochimica Acta Part B: Atomic Spectroscopy, vol. 56, nº 6, pp. 821-830, 2001.
[4] C. F. Alvira, O. J. Orzi and M. G. Bilmes , "Surface Treatment Analyses of Car Bearings by Using Laser-Induced Breakdown Spectroscopy," Applied Spectroscopy, vol. 6, no. 2, pp. 192-198., 2009.
[5] P. F. Rull, «Nuevas Fronteras en Mineralogía y Geoquímica de Campo,» Revista de la sociedad española de mineralogía, vol. 13, pp. 189-190, 2010.
[6] R. S. Russell, «Laser-induced breakdown spectroscopy (LIBS) – an emerging field-portable sensor technology for real-time, in-situ geochemical and environmental analysis,» Geochemistry: Exploration, Environment, Analysis, vol. 5, pp. 21-28, 2005.
[7] O. Ormachea y M. Delgado, «Diseño y construcción de un láser pulsado portátil del tipo YAG: Nd+++ para aplicaciones LIBS,» Investigación & Desarrollo, vol. 11, pp. 85-97, 2011.
[8] IMA-Engineering, "Innovative Mineral Analyzers," 2016. [Online]. Available: http://www.ima.fi/products. [Accessed 18 march 2016]. [ Links ]
[9] ProgramNuclearRadiationEngineering, "Prompt Gamma Activation Analysis," 2016. [Online]. Available: https://nuclear.engr.utexas.edu/index.php/netl/services/prompt-gamma-activation-analysis. [Accessed 14 april 2018]. [ Links ]
[10] M. Gaft, L. Nagli, Y. Griosman y A. Barishnikov, «Industrial Online Raw Materials Analyzer Based on Laser-Induced Breakdown Spectroscopy,» Applied Spectroscopy, vol. 68, nº 9, pp. 1004-1015, 2014.
[11] E. Tognoni, V. Palleschi, M. Corsi y G. Cristoforetti, «Quantitative micro-analysis by laser-induced breakdown spectroscopy: a review of the experimental approaches,» Spectrochimica Acta Part B, vol. 57, pp. 1115-1130, 2002.
[12] O. Ormachea, «Diseño y construcción de un laser solido pulsado del tipo YAG: Nd+++,» Revista Boliviana de Física, vol. 13, nº 13, pp. 54-57, 2007.
[13] O. Ormachea, O. Urquidi y J. Cisneros , «Desarrollo de un espectrómetro de difracción y su programa de control utilizando elementos comerciales de bajo costo,» Revista Cubana de Física, pp. 93-98, 2013.
[14] I. Terceros y O. Ormachea, «Control inalámbrico de un sistema LIBS portátil basado en tecnología bluetooth,» Investigación & Desarrollo, pp. 108-117, 2014.
[15] O. Ormachea, O. Urquidi and D. Casazola, "Development of a portable low-cost LIBS system," Proc. of SPIE 8th Ibero American Optics Meeting/11th Latin American Meeting on Optics, Lasers, and Applications, pp. 87851D-87851D-8, 2013.
[16] O. Urquidi y O. Ormachea, «Desarrollo de un sistema de análisis fisicoquímico, basado en espectroscopia de emisión atómica inducida por radiación láser,» Investigación & Desarrollo, vol. 13, pp. 5-12, 2013.
[17] J. Espinoza, «Minería boliviana 2001-2011,» El Diario, p. 23, 7 marzo 2012.
[18] Banco-Mundial, "Banco Mundial," 2016. [Online]. Available: http://datos.bancomundial.org/indicator/NY.GDP.MKTP.CD?locations=BO. [Accessed 11 may 2018]. [ Links ]
[19] Chamaleon-CCD, "Poin gray," 2016. [Online]. Available: https://www.ptgrey.com/chameleon-usb2-cameras. [Accessed 5 may 2018]. [ Links ]
[20] I. Terceros, O. Ormachea, A. Villazon y O. Urquidi, «Desarrollo de un espectrómetro de bajo costo con una rejilla ajustable y control electrónico inalámbrico,» Revista Politécnica, pp. 113-119, 2016.
[21] NIST, "NIST Atomic Spectra Database Lines Form," 2016. [Online]. Available: http://physics.nist.gov/PhysRefData/ASD/lines_form.html. [Accessed 13 march 2018]. [ Links ]
[22] OSCAR, "Optical Science Center for Applied Research - Laser Induced Breakdown Spectroscopy Elemental Spectra," 2016. [Online]. Available: https://oscar.desu.edu/libs/. [Accessed 4 March 2018]. [ Links ]
[23] Portable low cost analyzer LIBS 2018. [Online]. Available: https://www.laser2000.se/Photonics/Optical-Instrumentation/Spectroscopy/Portable-low-cost-analyzer-LIBS.html. [Accessed 5 may 2018].
[24] Z-300 LIBS Analyzer 2018. [Online]. Available: https://www.sciaps.com/libs-handheld-laser-analyzers/z-300/. [Accessed 2 may 2018].
[25] LS-2131M Compact pulsed Nd:YAG laser 2018. [Online]. https://www.lotis-tii.com/eng/productid69.php. [Accessed 15 may 2018].
[26] USB4000 (Custom) 2018. [Online]. https://oceanoptics.com/product/usb4000-custom/. [Accessed 15 may 2018].
[1] https://www.ptgrey.com/Downloads/