Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
Compartir
Investigación & Desarrollo
versión impresa ISSN 1814-6333versión On-line ISSN 2518-4431
Inv. y Des. vol.1 no.17 Cochabamba 2017
http://dx.doi.org/10.23881/idupbo.017.1-4i
ARTÍCULOS–INGENIERÍAS
DEGRADATION OF SYNTHETIC ORGANIC DYES IN SOLUTION BY FERRATE – HYPOCHLORITE OR CALCIUM HYPOCHLORITE
DEGRADACIÓN DE TINTES SINTÉTICOS ORGÁNICOS EN SOLUCIÓN POR FERRATO-HIPOCLORITO O HIPOCLORITO DE CALCIO
Pablo Marcelo Pérez Garcíaa,b, Sandra L. Ibáñez-Calerob and Ramiro Escalera Vásqueza
aCentro de Investigaciones en Procesos Industriales – CIPI
bCentro de Investigaciones Fitoquímicas-CIF
Universidad Privada Boliviana
(Recibido el 18 mayo 2017, aceptado para publicación el 16 de junio 2017)
ABSTRACT
This study investigated the use of ferrate – hypochlorite solution or solid Ca(OCl)2 as treatment options to remove color and COD from solutions of the synthetic organic dyes: Reactive blue 19, Allura red and Reactive black 5. It was found that doses around 1000 ppmCa(OCl)2 had color removal efficienciesaround 90 % for all the dyes after 15 minutes reaction time; this treatment had a COD removal efficiency of 97 % forazo dyeAllura red. The ferrate – hypochlorite (0.06 mM - <400 ppm) treatment resulted in a 50% color removal for Reactive blue 19 dye after 1 hour reaction time. The COD removal of Reactive blue 19 dye was improved from 40% to 84% when the ferrate – hypochlorite solution was used.
Keywords: Ferrate, Synthetic Organic Dyes, Hipochlorite, COD Removal.
RESUMEN
Este estudio investigó el uso de una solución de ferrato – hipoclorito o Ca(OCl)2 sólido como opciones de tratamiento para la remoción del color y de la DQO de soluciones de los tintes orgánicos industriales: Reactive blue 19, Allura red y Reactive black 5. Se encontró que dosis de alrededor 1000 ppm de Ca(OCl)2 generaron eficiencias de remoción de color de alrededor el 90 % para todos los tintes luego de 15 minutos de reacción; este tratamiento tuvo una eficiencia de remoción de la DQO de 97 % para el tinte azoAllura Red. El tratamiento con ferrato – hipoclorito (0.06 mM - <400 ppm) resultó en una remoción de color de 50 % para el tinte Reactive blue 19 después de 1 hora de reacción.La remoción de la DQO del tinte orgánico industrial Reactive blue 19 fue mejorado del 40% al 84% cuando la solución ferrato – hipoclorito fue utilizada.
Palabras claves: Ferrato, Tintes Orgánicos Industriales, Hipoclorito, Remoción DQO.
1. INTRODUCTION
Synthetic organic dyes are among the major chemicals used as manufacture goods by the textile industry[1]. These dyes are recalcitrant chemicals and represent a major concern for the treatment of industrial textile effluents because conventional wastewater treatment systemshave been inefficient eliminating them[2], [3]. During the last decades, the treatment technologies based on Advanced Oxidation Process (AOP) have garnered the great interest of the scientific community for the treatment of industrial effluents containing high concentration of recalcitrant chemicals [4], [5]. During these processes, very reactive chemical species are introduced orproduced in situto oxidize organic matter,ideally only generating CO2, H2O and biodegradable organic products. For instance, the application of the Fenton reagent (a mixture of H2O2/Fe2+) generates hydroxyl radicals under the action of UV light triggering a complex chain reaction provoking the organic matter decompositiondeclining the chemical oxygen demand (COD)[6].Our group reported[7] that a treatment based onphoto-Fenton process whichresults inhigh removal efficiencies of color (77 %) and COD (92 %) from industrial effluents containing high content of synthetic organic dyes. The process was conceived in a solarlight concentrating photo-reactor system with an average reaction time of 2.5 hours.
Another highly oxidant reagent based on iron is ferrate (FeO42-), where iron has an oxidation state of +6. In aqueous solution the ferrate is readily oxidized to Fe3+ having standard half-reduction potentials of +2.2 V and +0.72 V in acidic and basic medium respectively, these potential values are comparable to thosefrom common oxidizers as ozone or chlorine[8]. The advantages of using ferrate as oxidizing agent in wastewater treatment are its low toxicity and its flocculating activity which increasesthe removal percentage of COD[8], [9]. Moreover its synthesis is simple from affordable chemical compounds[10], [11] and the oxidation of organic matter by ferrate only produces innocuous organic by products[12]; this is not always the case when ozone or chlorine derivatives are applied[13].
The use of ferrate as oxidizing agent for the removal of synthetic organic dyes form industrial effluents was reported by Li et al.[14]. The authors declared a 95 % decolorization of a 50 ppm azo dye Orange II solution by batch reacting with 0.17 mMferrate– hypochlorite solution after 30 minutes; this result demonstrated an oxidizing synergy between the ferrate and hypochlorite since the treatment with only ferrate (10 ppm) led to 67% decolorization. Similar removal efficiencies were reported by Xu et al.[15] and GC-MS analysis demonstrated that the first step of the decomposition of the azo dye was the cleavage of the azo bond. On the other hand, Massoudinejad et al.[16]studied the treatment of effluents from polyacrylic textile industry by Ca(OCl)2 direct application. A dose treatment of 2000 ppm led to >95% color – COD removalwith a reaction time of only 10 minutes. The authors declared that the absence of oxygen and humic materials in the effluents avoid the formation of any hazardous chlorinated products such trihalomethanes (THM).
This work aims to study the decomposition treatment of three synthetic industrial organic dyesused by the textile industry in Cochabamba[7], using ferrate – hypochlorite solution or solid calcium hypochlorite as oxidizing agents. This report also includes the complete description of the synthesis and characterization of pure ferrate and ferrate – hypochlorite solutions made from cheap and readily available starting materials. To the best of our knowledge this is the first work concerning the use of ferrate – hypochlorite solution or Ca(OCl)2for the removal treatment of the synthetic organic dyes: Reactive blue 19, Allura red and Reactive black 5.
2. MATERIALS AND METHODS
2.1 General
The UV-vis spectra analyses were carried out using a Shimadzu UV-1601 spectrometer using quartz cells with 1 cm length. The COD analysis were carried out by the Centro de Aguas y SaneamientoAmbiental – CASA - UMSS using the standard method 5220 D. The synthetic organic dyes were provided by the local industry[7]. The rest of the chemicals,in UPB laboratories, had technical grade qualities and were purchased in local stores.
2.2 Characterization of organic dyes
The UV-vis spectra were registered for 100 ppm solutions of each dye: blue, red and black. The solutions were prepared using distilled water as solvent.
2.3 Wet chemical synthesis of ferrate
The used experimental procedure was developed by Ibañez et al.[10]6 g of NaOH pulverized were placed into a 100 mL beaker containing 80 mL of distilled water and magnetic stirred until total dissolution. 4 g of Ca(OCl)2 were added to the basic solution and magnetic stirred at 60°C until the formation of an homogeneous white suspension. 500 mg of pulverized FeCl3 were added to the suspension and the heating temperature was increased to 80 °C. The resulting suspension was stirred at 80°C until a homogenous violet color appeared. The suspension was then cooled slowly to room temperature. The suspension was placed in centrifugal tubes and centrifuged at 2000 rpm during 2 minutes. The supernatant was removed and placed in a beaker. The final clear violet solution was UV-vis analyzed.
2.4 Electrochemical synthesis of ferrate
The used experimental procedure was developed by Ibañez et al.[10]30 g of NaOH were dissolved in 200 mL of distilled water. This solution was placed inside an U shaped glass tube vertically installed using an utility clamp held to a ring stand. An iron coiled wire was installed inside each extremity of the U-tube and connected to a voltage generator using jump wires. 12 V were applied to the system until inside the anode extremity a clear violet solution appeared. The voltage source was then switched-off and the iron electrodes were carefully removed. The ferrate solution was recovered and placed in a beaker to further UV-vis analysis.
2.5 Color removal tests by ferrate – hypochlorite solutions
● Preparation of 200 ppm dye solution: 50 mg of dye were dissolved in distilled water insidea 250 mL volumetric flask filled to the calibration mark.
● Preparation of basic hypochlorite solution: 6 g of NaOH pulverized were placed into a 100 mL beaker containing 80 mL of distilled water and magnetic stirred until total dissolution. 4 g of Ca(OCl)2 were added to the basic solution and magnetic stirred at 60°C until the formation of an homogeneous white suspension. The suspension was then cooled slowly to room temperature. The suspension was placed in centrifugal tubes and centrifuged at 2000 rpm during 2 minutes. The supernatant was removed and placed in a beaker.
● Color removal test with ferrate hypochlorite solution: 20 mL of 200 ppm blue dye solution were placed into a 100 mL beaker and 20 mL ferrate hypochlorite solution (absorbance: 0.65) were added under magnetic stirring. The resulting solution was stirred during 15 minutes and its UV-vis spectrum was recorded.
● Color removal test with basic hypochlorite solution: 20 mL of 200 ppm blue dye solution were placed into a 100 mL beaker and 20 mL basic hypochlorite solution was added under magnetic stirring. The resulting solution was stirred during 15 minutes and its UV-vis spectrum was recorded.
● Color removal test with Ca(OCl)2: 40 mL of 200 ppm dye solution were placed into a 100 mL and an specific amount of Ca(OCl)2 was added until magnetic stirring. The resulting solution was stirred during 15 minutes and its UV-vis spectrum was recorded. Three tests were carried out for each dye using increasing quantities of Ca(OCl)2: approximately 50, 100 and 200 mg.
2.6 COD removal tests
● COD removal with Ca(OCl)2: This test was carried out using separately blue and red dye. 50 mg of dye were dissolved in water inside a 250 mL volumetric flask filled to the calibration mark. 100 mL of this 200 ppm solution were separated for COD analysis as blank. 100 mL were treated with 100 mg Ca(OCl)2 (1000 ppm dose) under magnetic stirring during 15 min. A little portion of the final solution was UV-vis analyzed and the remainder was sent to COD analysis.
● COD removal with ferrate – hypochlorite solution: 50 mg of blue dye were dissolved in water inside a 250 mL volumetric flask filled to the calibration mark. 100 mL of this 200 ppm solution were separated for COD analysis as blank. 100 mL were treated with 10 mL of ferrate – hypochlorite solution (A=0.758, c=0.66 mM) during 1 hour. A little portion of the final solution was UV-vis analyzed and the remainder was sent to COD analysis.
3. RESULTS AND DISCUSSION
3.1 Characterization of industrial organic dyes
Three colors of synthetic organic dyes were analyzed: blue, red and black. The chemical nature of each one was characterized by UV-vis spectrometry and identified by comparison of the absorption spectra to those reported in the literature. The absorption spectra were recorded in a wavelength range of 190 – 900 nm.
Figure 1 shows the absorption spectrumfor the blue dye showing a maximum absorption at 590 nm. A comparison to reported spectra[17] let us to conclude that the commercial name of this dye is Reactive blue 19.
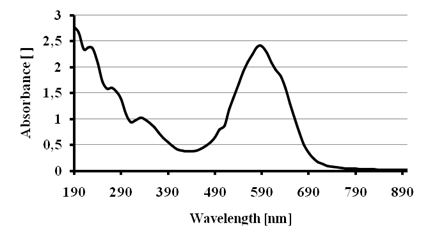
Figure 1 - Absorption spectrum for the dye Reactive blue 19.
The chemical structure of this dye is presented in Figure 2. The molecular formula of this compound is C22H16N2Na2O11S3 with a molecular weight of 626.5 g/mol. The compound is an anthraquinone dye (quinoidchromophore)[18] and has auxochromes such as amine, sulfone and sulfonate.
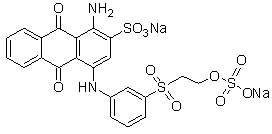
Figure 2 -Chemical structure of the dye Reactive blue 19.
The absorption spectrum of the red dye was recorded (Figure 3) showing a maximum absorbance at 490 nm. Its comparison with the literature[19] let us to conclude that the commercial name of this dye is Allura red.
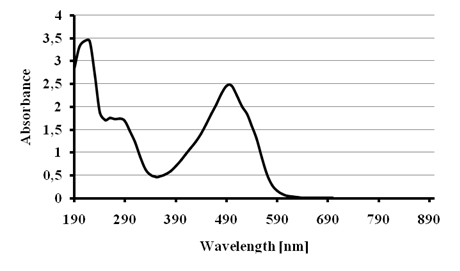
Figure 3 - Absorption spectrum for the dye Allura red.
The dye Allura red has the molecular formula C18H14N2Na2O8S2 and a molecular weight of 496.4 g/mol. The chemical structure is showed in Figure 4. The compound is an azo dye[18], the azo chromophore is formed by tautomerism from the hidrazone group. The auxochromes are methoxy and sulfonate groups.
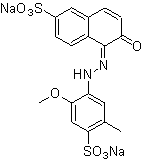
Figure 4 - Chemical structure of the dye Allura Red.
Figure 5 shows the absorption spectrum for the black dye with a maximum absorption at 600 nm, the shape of the spectrum is very broad. We compared this spectrum with the literature[20] concluding that the commercial name of the dye is Reactive black 5.
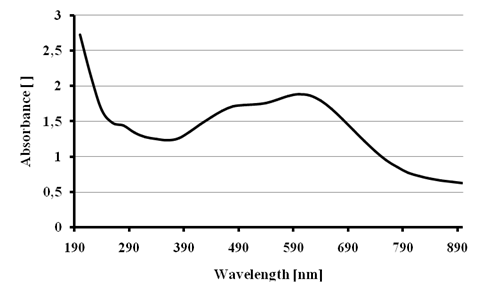
Figure 5 - Absorption spectrum for the dye Reactive black 5.
The dye Reactive Black 5 has a molecular formula C16H21N5Na4O19S6 with a molecular weight of 991.8 g/mol. Figure 6 shows the chemical structure. The compound is an azo dye[18] with two azo chromophores (one is the hidrazone tautomer); the auxochromes are sulfone, amine and sulfonate groups.
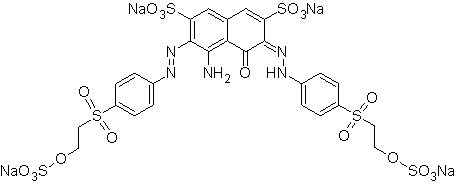
Figure 6 - Chemical structure of the dye Reactive Black 5.
3.2 Characterization of ferrate
Ferrate was synthesized in solution by chemical wet and electrochemical methods. Previous works reported[21] that aqueous ferrate solutions generates a maximum absorbance at approximately 510 nm. We show in figure 7, the UV/vis spectra of our ferrate solutions obtained by wet chemical and electrochemical method. As can be seen, the maximum absorbance matches at 510 nm for both synthetic routes. Themaximum absorbance valueswere converted in ferrate solution concentrations using1150 L∙mol-1∙cm-1 as extinction coefficient [21]. The ferrate concentration in the solution made by wet chemical method (0.66 mM) is around seven times higher than the concentration obtained by electrochemical method (0.09 mM). A plausible explanation is that the iron rods used as electrodes did not offer a convenient active surface to ensure an optimum mass transfer during the electrochemical synthesis.
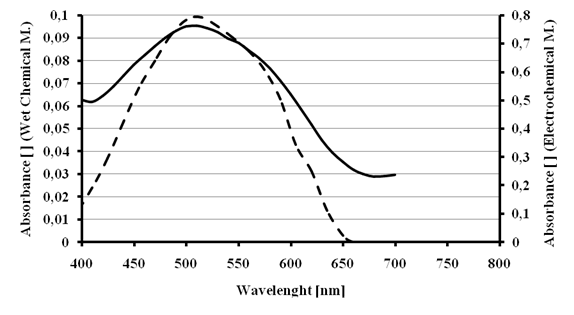
Figure 7 - Absorption spectra for ferrate solutions.Solid line: Wet Chemical MethodDashed line: Electrochemical Method.
We studied the stability of our ferrate solutions following the kinetics of decomposition at room temperature. A marked visual color change characterizes the decomposition of ferrate form strong violet to yellow brown.In order to compare our results to those described in the literature[21], we assumed a first order kinetics. Figure 8 shows the kinetic decomposition profile for the ferrate produced by both applied synthetic methodologies and their respective exponential regression; the decomposition rate constants are 3×10-5s-1(wet chemical method) and 8×10-4 s-1(electrochemical method). From these values, we calculated the decomposition halftime of ferrate ion in solution: 6.5 hours (wet chemical method) and 17 minutes (electrochemical method). The kinetic values for the ferrate solution synthesized by electrochemical method correlate perfectly with those previously reported[21]. The higher stability of ferrate solution made by wet chemical method might be due to the presence of hypochlorite; both species have in basic environment similar redox potentials (around +0.8 V) so hypochlorite helps to avoid afast reduction from iron +6 to iron +3.
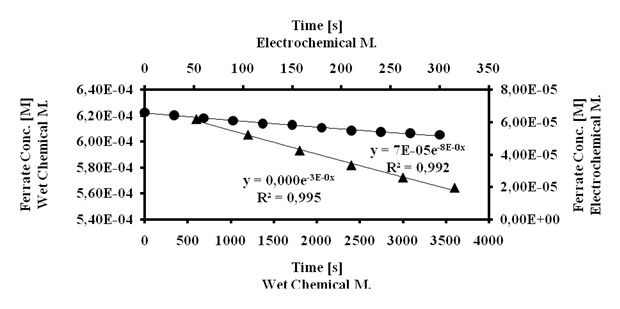
Figure 8 - Kinetic profiles of ferrate solutions.
Dots: Electrochemical Method and Triangles: Wet Chemical Method.
3.3 Color removal tests
The color removal efficiencywas tested for the Reactive blue 19 dye using the ferrate – hypochlorite solution and a basic saturated hypochlorite solution as blank test. The tests were carried out at room temperature over 20 mL of 200 ppm dye solution. The addition of 20 mL of ferrate – hypochlorite solution or 20 mL of basic saturated hypochlorite solutionproduced a color removal of>95 % after 1 hour agitation. In figure 9, a total disappearance of the signals at visible region was observed; this might mean that the quinoidchromophore is completely cleaved. On the hand, the spectra after treatment show strong signals at the UV region, this might indicate that the aromatic rings could not be decomposed during the process.
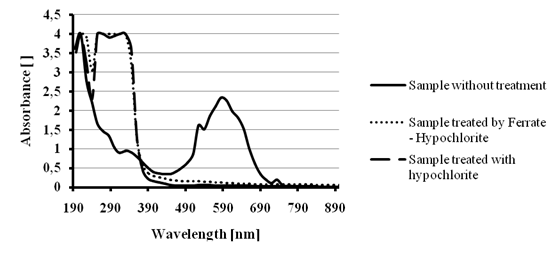
Figure 9 -Color removal test for 200 ppm blue dye solution.
Considering that the ferrate – hypochlorite and basic saturated hypochlorite solutions have the same color removal efficiency, we decided then to try a treatment directly applying solid calcium hypochlorite to the dye solution. We applied Ca(OCl)2 concentrations similar to those previously reported[16]. Figure 10 shows the spectra comparison for the treatment of 40 mL of 200 ppm solution of Reactive blue 19 dye. The color removal was around 50 % after 15 minutes reaction for all the Ca(OCl)2 doses: 1375 ppm, 2443 ppm and 5700 ppm; but more than 95 % after 60 minutes of treatment with the lower dose of 1375 ppm. These results could indicate that the kinetics of the color removal process is only depending on the dye concentration. The increase of signals at UV region might also indicate that stable aromatic compounds stays after the cleavage of the quinoidchromophore.
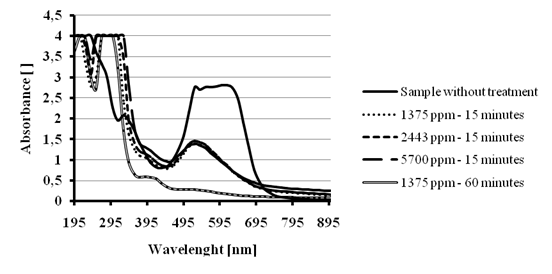
Figure 10 - Color removal tests for 200 ppm Reactive blue 19 solution treated with several Ca(OCl)2 doses.
Encouraged by these results, we decided to treat solutions of the other synthetic dyes by increasing doses of Ca(OCl)2. The treatment of 40 mL of 200 ppm Allura red dye solution during 15 minutes resulted in a 50 % color removal with a dose of 1275 ppm and 65 % for a dose of 2443 ppm or 5165 ppm. After one hour treatment, the lower dose 1275 ppm resulted in >95 % color removal.The spectra in Figure 11 shows that once the azo chromophore is cleaved stable aromatic residuals might stay in solution.
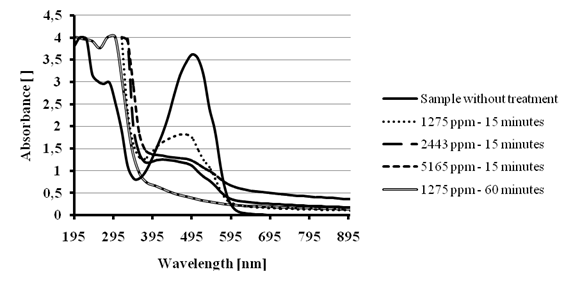
Figure 11 - Color removal tests for 200 ppm Allura Red solution treated with several Ca(OCl)2 doses.
Similar results were obtained when a 200 ppm Reactive black 5 dye solution was treated with increasing doses of Ca(OCl)2. The treatment of 40 mL of this solution by 1330 ppm, 2480 ppm or 5050 ppm resulted in 58 % color removal using any mentioned dose after 15 minutes treatment. After one hour treatment, the lower dose resulted once again in a color removal >95 %. It seems that the kinetic of the decomposition process is also independent of the concentration of Ca(OCl)2 as for the case of Reactive blue 19 dye. The absorption spectra after treatment (Figure 12) also indicate that aromatic compounds might stay in solution after the cleavage of the azo chromophores.
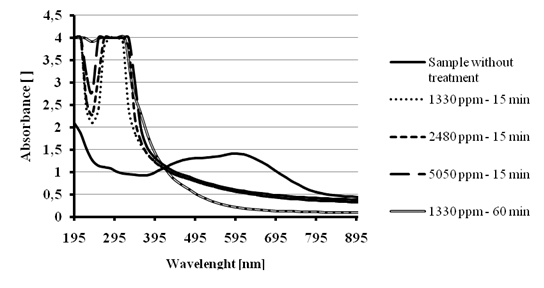
Figure 12 - Color removal tests for 200 ppm Reactive Black 5 solution treated with several Ca(OCl)2 doses.
3.4 COD removal tests
We tested first the COD removal efficiency by Ca(OCl)2 treatment for a 200 ppm Allura Red solution. The volume treated was 100 mL with a dose of 1000 ppm and15 minutes reaction time. The decolorization degree was >90% as it is showed in figure 13. The COD charges for the non-treated and the treated samples were 2602 mg O2/L and 76 mg O2/L respectively, this represents a 97 % COD removal.
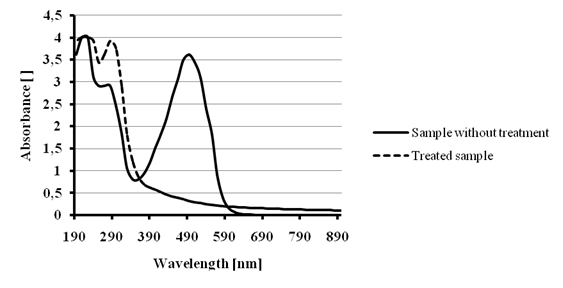
Figure 13 - Color removal tests for 200 ppm Allura Red solution with 1000 ppm Ca(OCl)2 dose.
We decided also to test the treatment COD removal efficiency for the anthraquinone dye Reactive Blue 19.Anthraquinone dyes are more resistant to biodegradation than azo dyes[22]; for this reason, we decided to compare the treatments with Ca(OCl)2 and ferrate – hypochlorite solution. 100 mL of 200 ppm Reactive blue 19 dye solution were treated with 1000 ppm dose of Ca(OCl)2 during 15 minutes (Sample 1) and 100 mL of 200 ppm Reactive blue 19 dye solution were treated with 10 mL ferrate – hypochlorite solution (A=0.758, c=0.66 mM) during 1 hour (Sample 2). As it can be seen in Figure 14, the decolorization efficiency for the treatment of sample 1 was around 80 % and for the treatment of sample 2 was around 50 %.
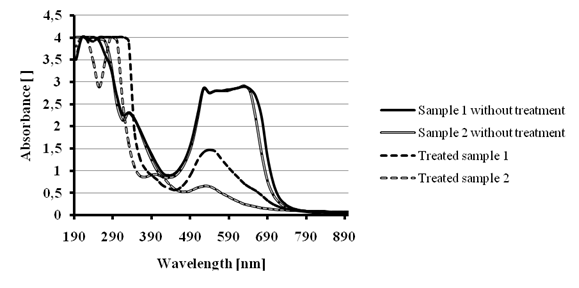
Figure 14 - Color removal tests for 200 ppm Reactive Blue 19. Sample 1: ferrate – hypochlorite treatment.
Sample 2: Ca(OCl)2 treatment.
It might seem that the treatment with only Ca(OCl)2 is more efficient than the treatment with ferrate – hypochlorite solution at the concentrations accessible in our experimental conditions. It is important to mention that the effective concentrations of the treating ferrate – hypochlorite solution are 0.06 mM - <400 ppm, which are much lower than those previously reported[14], [16]. Before treatment, the experimental analysis of sample 1 and sample 2 indicated different COD values (100 and 375 mg O2/L respectively) and the COD value after treatment was 60 mg O2/L for both samples. The difference between the initial CODs, despite the same initial dye concentrations (UV-vis spectra totally superpose in figure 14), might be due to the partial anthraquinone dye oxidation by dichromate during COD analysis[23]. Another plausible explanation is the samples were analyzed at different times by the CASA laboratory which could lead to a partial oxidation of the dye by the atmospheric oxygen. Nevertheless, the reference and treated sample were always analyzed at the same time; therefore the COD removal percentages are valid. The above described results are very encouraging since the efficient decolorization of Reactive Blue 19 dye by the two treatment methods, implies the cleavage of the quinoidchromophore; which represents the key step for the total decomposition of this kind of molecules[15].
4. CONCLUSION
In summary, we have presented a preliminary study for the removal treatment of three common used dyes (Reactive Blue 19, Allura red and Reactive black 5) by Ca(OCl)2 or ferrate – hypochlorite solution. Both treatments efficiently removed the color of 200 ppm dye solution in short time reaction (15 min). The COD removal efficiency for azo dye Allura Red by reaction with Ca(OCl)2 (1000 ppm) was 97 %, this is very promising for the treatment of effluents having high charges of dyes from this chemical family. For anthraquinone dyes,variability was observed for the COD values before treatment (despite same dye concentrations), making inconsistent the evaluation of the treatment COD removal efficiencies for this kind of dyes. This COD value erraticism might be eliminated measuring the organic content concentration with the Standard Method 5520 B, open reflux method, as used in other reported studies[24]. To our knowledge, this work is the first one studying the decomposition of the three above mentioned industrial synthetic dyes by treating them with Ca(OCl)2 and ferrate – hypochlorite solution. We are preparing further detailed studies to understand the chemical treatment mechanisms and to optimize COD analysis for anthraquinone dyes, building solid scientific bases for a future technology transfer to local textile industries.
5. BIBLIOGRAPHY
[1] C. ONeill, F. R. Hawkes, D. L. Hawkes, N. D. Lourenço, H. M. Pinheiro, and W. Delée, Colour in textile effluents – sources, measurement, discharge consents and simulation: a review, J. Chem. Technol. Biotechnol., vol. 74, no. 11, pp. 1009–1018, 1999.
[2] S. Arslan, M. Eyvaz, E. Gürbulak, and E. Yüksel, A Review of State-of-the-Art Technologies in Dye-Containing Wastewater Treatment – The Textile Industry Case, in Textile Wastewater Treatment, E. P. A. Kumbasar and A. E. Körlü, Eds. Rijeka: InTech, 2016.
[3] S. Barışçı, O. Turkay, and A. Dimoglo, Review on Greywater Treatment and Dye Removal from Aqueous Solution by Ferrate (VI), in Ferrites and Ferrates: Chemistry and Applications in Sustainable Energy and Environmental Remediation, pp. 349–409.
[4] M. A. Oturan and J.-J. Aaron, Advanced Oxidation Processes in Water/Wastewater Treatment: Principles and Applications. A Review, Crit. Rev. Environ. Sci. Technol., vol. 44, no. 23, pp. 2577–2641, Dec. 2014.
[5] M. Antonopoulou, E. Evgenidou, D. Lambropoulou, and I. Konstantinou, A review on advanced oxidation processes for the removal of taste and odor compounds from aqueous media, Water Res., vol. 53, pp. 215–234, 2014.
[6] A. Babuponnusami and K. Muthukumar, A review on Fenton and improvements to the Fenton process for wastewater treatment, J. Environ. Chem. Eng., vol. 2, no. 1, pp. 557–572, 2014.
[7] A. Zaconeta Piva and R. Escalera vasquez, Desarrollo de un sistema de reciclaje de aguas residuales textiles coloreadas mediante la utilización de un fotoreactor solar, Investig. Desarro., vol. 1, no. 1, pp. 36–47, 2010.
[8] J.-Q. Jiang, The Role of Ferrate(VI) in the Remediation of Emerging Micro Pollutants, Procedia Environ. Sci., vol. 18, pp. 418–426, 2013.
[9] J.-Q. Jiang, Advances in the development and application of ferrate(VI) for water and wastewater treatment, J. Chem. Technol. Biotechnol., vol. 89, no. 2, pp. 165–177, 2014.
[10] J. G. Ibanez, M. Tellez-Giron, D. Alvarez, and E. Garcia-Pintor, Laboratory Experiments on the Electrochemical Remediation of the Environment. Part 6: Microscale Production of Ferrate, J. Chem. Educ., vol. 81, no. 2, p. 251, 2004.
[11] V. K. Sharma, L. Chen, and R. Zboril, Review on High Valent FeVI (Ferrate): A Sustainable Green Oxidant in Organic Chemistry and Transformation of Pharmaceuticals, ACS Sustain. Chem. Eng., vol. 4, no. 1, pp. 18–34, 2016.
[12] S. J. DeLuca, A. C. Chao, and C. Smallwood, Ames Test of Ferrate Treated Water, J. Environ. Eng., vol. 5, no. 109, pp. 1159–1167, 1983.
[13] K. L. D. Katiyar, A. Singh, P. Bose, and V. Dutta, Similarities of Trihalomethanes Formation during Chlorination of Diluted Waste Water and Ganga River Water at Kanpur, Pollut. Res., vol. 34 (2), no. AUGUST, pp. 289–296, 2015.
[14] G. Li, N. Wang, B. Liu, and X. Zhang, Decolorization of azo dye Orange II by ferrate(VI)–hypochlorite liquid mixture, potassium ferrate(VI) and potassium permanganate, Desalination, vol. 249, no. 3, pp. 936–941, 2009.
[15] G. R. Xu, Y. P. Zhang, and G. B. Li, Degradation of azo dye active brilliant red X-3B by composite ferrate solution, J. Hazard. Mater., vol. 161, no. 2, pp. 1299–1305, 2009.
[16] M. Massoudinejad, M. Ghaderpoori, and M. Rezazadeh Azari, The removal of COD and Color from Textile Industry by Chlorine Hypochlorite, Int. J. Adv. Sci. Technol., vol. 76, pp. 35–42, 2015.
[17] D. C. dos Santos et al., Application of Carbon Composite Adsorbents Prepared from Coffee Waste and Clay for the Removal of Reactive Dyes from Aqueous Solutions, J. Braz. Chem. Soc., vol. 26, pp. 924–938, 2015.
[18] D. R. Waring and G. Hallas, Eds., The Chemistry and Application of Dyes. New York: Plenum Press, 1990.
[19] M. H. V Werts et al., Quantitative full-colour transmitted light microscopy and dyes for concentration mapping and measurement of diffusion coefficients in microfluidic architectures, Lab Chip, vol. 12, no. 4, pp. 808–820, 2012.
[20] M. S. Roriz, J. F. Osma, J. A. Teixeira, and S. Rodríguez Couto, Application of response surface methodological approach to optimise Reactive Black 5 decolouration by crude laccase from Trametes pubescens, J. Hazard. Mater., vol. 169, no. 1, pp. 691–696, 2009.
[21] C. Li, X. Z. Li, and N. Graham, A study of the preparation and reactivity of potassium ferrate, Chemosphere, vol. 61, no. 4, pp. 537–543, 2005.
[22] M. Celebi, M. Z. Akdeste, and Y. Hüseyin, Determination of decolorization properties of Reactive Blue 19 dye using Horseradish Peroxidase enzyme, Turkish J. Biochem., vol. 2, no. 37, pp. 200–206, 2012.
[23] A. M. Dessouki and S. E. Abdel-Aab, Radiation Degradation of some Commercial Dyes in Wastewater, in International Conference on Hazardous Waste: Source effect and Management, 1998, pp. 1331–1345.
[24] S. Chakrabarti and B. K. Dutta, Photocatalytic degradation of model textile dyes in wastewater using ZnO as semiconductor catalyst, J. Hazard. Mater., vol. 112, no. 3, pp. 269–278, 2004.














