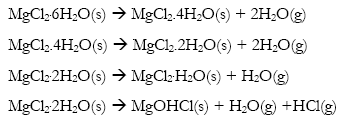Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
Compartir
Acta Nova
versión On-line ISSN 1683-0789
RevActaNova. v.9 n.5-6 Cochabamba nov. 2020
Artículo Científico
Development of shape stabilized thermal energy storage materials based on inorganic chloride salts by direct sol-gel method
Desarrollo de materiales de almacenamiento de energía térmica estabilizados en forma basados en sales de cloruro inorgánico mediante el método directo sol-gel
Yanio E. Milián, Svetlana Ushak*
Center for Advanced Research in Lithium and Industrial Minerals (CELiMIN), Universidad de Antofagasta y Departamento de Ingeniera Quimica y Procesos de Minerales de Universidad de Antofagasta, Avenue Universidad de Antofagasta 02800, Antofagasta, Chile.
Recibido: 13 de mayo 2020
Aceptado: 18 de septiembre 2020
Abstract: The potential of several improved materials was analyzed for thermal energy storage in this work, specifically for latent heat storage (LHS) and for thermochemical energy storage (TCS). The use of a direct sol-gel process, using tetraethyl orthosilicate as monomer, was applied to obtain shape stabilized -thermal energy storage materials (SS-TES material) based on different inorganic salts (MgCl2·6H2O, MnCL and LiCl) and wastes from non-metallic mining industry: bischofite (95% of MgCl2·6H2O) and carnallite (73% of KCl-MgCl2·6H2O). A detailed analysis was offered for the developed materials based on x-ray diffraction, scanning electronic microscopy and thermal analyses. A new low-cost material of carnallite/SiO2 showed an atypical absorption peak at 50 °C, which increase with the content of carnallite in the final material, indicating a possible potential as a thermochemical storage medium, for low temperature applications, below 100 °C. However, the application of the sol-gel method under the conditions studied in this work did not allow to obtain SS-TES materials based on MgCl2·6H2O, MnCl2 and carnallite for LHS, which indicates that future adjustments are necessary to obtain satisfactory results.
Keywords: shape stabilized thermal energy storage materials, bischofite, carnallite, sol-gel, tetraethyl orthosilicate, silicon dioxide.
Resumen: En este trabajo se analizo el potential de varios materiales mejorados para el almacenamiento de energía térmica, específicamente para el almacenamiento de calor latente (LHS) y para el almacenamiento de energía termoquímica (TCS). Se aplico el uso de un proceso directo sol-gel, utilizando tetraetil ortosilicato como monómero, para obtener materiales de almacenamiento de energía térmica estabilizada de forma (material SS-TES) a base de diferentes sales inorgánicas (MgCl2·6H2O, MnCl2 y LiCl) y residuos de Industria minera no metálica: bischofita (95% de MgCl2·6H2O) y carnalita (73% de KCl.MgCl2·6H2O). Se ofreció un análisis detallado de los materiales desarrollados basado en difracción de rayos X, microscopía electrónica de barrido y análisis térmicos. Un nuevo material de bajo costo de carnalita / SiO2 mostro un pico de absorción atípico a 50 ° C, que aumenta con el contenido de carnalita en el material final, lo que indica un posible potencial como medio de almacenamiento termoquímico, para aplicaciones de baja temperatura, por debajo de 100 ° C. Sin embargo, la aplicación del método sol-gel en las condiciones estudiadas en este trabajo no permitió obtener materiales SS-TES a base de MgCl2·6H2O, MnCl2 y carnalita para LHS, lo que indica que son necesarios ajustes futuros para obtener resultados satisfactorios.
Palabras clave: materiales de almacenamiento de energía térmica estabilizados en forma, bischofita, carnalita, sol-gel, ortosilicato de tetraetilo, dióxido de silicio.
1 Introduction
The solar energy technologies can be combined with thermal energy storage (TES) systems, which can be classified as thermochemical heat (TCS), sensible heat (SHS) and latent heat (LHS) storage [1]. The search of potential materials for TES systems and the optimization of their thermal performance are still a challenge. The availability and low-cost of these TES materials are important factors to consider for the sustainable development of any kind of solar based storage system [2]. Inorganic materials, including salt hydrates, metals and alloys are recently investigated for TES since have high thermal conductivity, energy storage densities, high working temperature range and low-cost [3]. Moreover, various inorganic wastes and byproducts from non-metallic mining industry are available without any application, accumulating in mining lands and constituting a serious risk to the environment. In recent years, the potentiality of these materials for TES applications has increased due to its cost being close to zero [4, 5]. Some of these wastes were proposed for SHS like astrakanite (Na2SO4·MgSO4·4H2O) [6], kainite (KCl·MgSO4·3H2O) [6] and NaCl [7] as by-products from the obtained processes of nonmetallic mining industry. Among the materials displaying potential as thermochemical material for TCS can be mentioned astrakanite and potassium carnallite (KCl·MgCl2·6H2O), which exhibited release of water below 300 °C [5,9]. Similarly, the dehydration reaction of bischofite [10] and carnallite [11] were analyzed as low-cost TCS materials. Compounds employed for LHS and called as phase change materials (PCMs) had been studied in several works, including those from inorganic wastes [5-13]. A dehydrated product obtained from astrakanite showed potential to be applied as PCM at high temperature (550 °C - 750 °C) [5,6]. Bischofite, with a phase change point at 100 °C and a latent heat (AH) around 115 kJ kg-1, was identified as a potential PCM with similar thermophysical characteristics to synthetic magnesium chloride hexahydrate, besides with a lower cost [12]. In fact, the successful application of bischofite impregnated in expanded graphite (EG) was demonstrated for thermal regulation on lithium batteries, absorbing the produced heat from batteries and, therefore, extending battery life and enhancing their performance [13]. However, all these inorganic materials present some adverse effects, as subcooling (around 35 °C) and phase segregation [12,14], which reduce the usefulness of the materials and, in some cases, could prevent entirely the heat recovery from the material.
Among the methods used to reduce the degree of subcooling are the addition of nucleating agents, mechanical agitation, chemical modifications, and the encapsulation of the PCMs, among others [14-16]. In a consecutive work, bischofite subcooling degree was reduced to 23.4 °C by the formulation of an inorganic eutectic mixture based on 40 wt.% bischofite and 60 wt.% Mg(NO3)2·6H2O, with a melting point (Tm) of 58.2 °C and Δh around 117.0 kJ kg-1 [16]. Nevertheless, lower subcooling degrees need to be achieved for real applications.
Encapsulation methods are among the most used techniques to improve thermal properties of inorganic TES materials and can be performed in two different ways: core-shell encapsulation and shape-stabilized thermal energy storage materials (SS-TES materials) [14,15]. Only few works related to the encapsulation of non-metallic industrial wastes and by-products [18,19] are available. Bischofite was micro-encapsulated by a fluidized bed method using acrylic acid as polymer and chloroform as solvent, after compatibility studies of several solvents and several polymers. The final microcapsules had excellent melting temperatures and latent heat (104.6 °C and 95 kj.kg-1, respectively), resulting in a decrease in subcooling degree and avoiding leakage of the hydrate when it melts [18]. MgCl2·6H2O-Mg(NO3)2·6H2O/fumed silica composites was obtained as SS-TES material by impregnation method, basically mixing the eutectic salts with dried fumed silica (85 °C, 2 h). Thermal conductivity, thermal stability and cycle stability were improved for the final material, compared with the eutectic salt [19]. However, the SS-TES materials currently developed are still insufficient to meet the demand and the requirements for practical and tangible applications. The search for economic options of support materials with a practical thermal performance and an effective-simple method to obtain SS-TES materials are still required. Support materials (SMs) based on silicon dioxide were used successfully to obtain SS-TES materials, since they are characterized for the presence of pores and a large internal surface area [20-22]. A one-step direct sol-gel technique was employed to develop lithium salts (LiCl and LiNO3) based shape stabilized phase change materials (SS-PCMs) with tetraethyl orthosilicate (TEOS), improving cycling stability and sub-cooling related with the pure salts [23]. As well, a Na2SC4 based SS-PCM was developed by the same technique, and the influence of pH (acid and basic hydrolysis) and PCM content were also analyzed [24]. Henceforward, the main objective of this work was to develop new TES materials with stabilized shape based on inorganic salts (MgCl2·6H2O, MnCl2 and LiCl) and low-cost wastes from non-metallic mining industry (bischofite and carnallite), by the sol-gel technique previously developed, with TEOS as monomer and via acid hydrolysis. Thus, the influence of the chemical nature of the inorganic TES material was evaluated. The potential of the developed materials was analyzed for LHS and TCS.
2 Materials and Procedures
2.1 Materials
Tetraethyl orthosilicate TEOS (reactant grade, Sigma-Aldrich, China) and ethanol (absolute grade for analysis, Merck Ka) were employed in the synthesis of SiO2 materials as SMs. The syntheses were produced by acid hydrolysis using hydrochloric acid (37 %) of analysis grade, obtained from Merck KGaA, Germany. Magnesium chloride hexahydrate MgCl2·H2O, manganese chloride MnCl2 and lithium chloride LiCl as reference material, (all grade salts for analysis), were obtained from Merck KGaA, Germany. Carnallite and bischofite were provided by the Rockwood Lithium mining company, Antofagasta, Chile. Distilled water was obtained in the laboratory using a water deionizing system SIMS600CP Mllipore Simplicity Personal Ultrapure Water System.
2.2 Experimental procedure
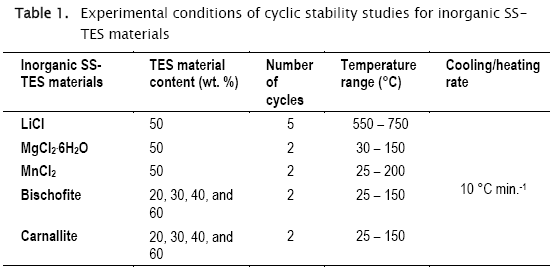
The used sol-gel procedure was based on the method proposed by Milian et al. [23, 24], via acid hydrolysis of the monomers to initiate the polymerization reaction. Briefly, tetraethyl orthosilicate was mixed with the same amount of ethanol under continuous stirring, HCl (30 %) and distilled water were added, and this solution was continuously stirred over two hours to produce the -Si-O-Si- pre-polymer. This pre-polymeric solution was employed straight to develop the SS-TES materials. The selected inorganic materials (Table 1), previously dissolved in deionized water, were mixed with the prepolymer and then stored for 10 days. Once the SS-TES materials were achieved in solid phase, materials were dried in an oven at 60 °C and finally mechanically pulverized. In this way, sol-gel procedure was employed to obtain SS-TES materials based on wastes from non-metallic mining industry: bischofite (MgCl2·6H2O) and carnallite (KMgCl3·6H2O). Eight new stabilized materials based on bischofite and carnallite with different inorganic salt contents (Table 1) were prepared.
2.3 SEM-EDX and XRD characterization of the SS-TES materials
The characterization of the new materials was performed by a Scanning Electron Microscopy (SEM) with Energy Dispersion X-ray Spectroscopy (EDS), with a Jeol machine, Model JSM6360 LV; a Siemens X-ray diffractometer (automatic and computerized D5000 model), equipped with a scintillation detector and the DiffracPlus software, for X-ray diffraction (XRD) analysis.
2.4 Thermal and cycling tests of the SS-TES materials
Thermal stability was studied using a thermogravimetry equipment (TG) coupled with differential scanning calorimeter (DSC) (Mettler Toledo Model 1 1100 LF), from 100 to 350 °C, with a heating / cooling rate of 10 °C min-1, using around 10 mg of the sample and alumina crucible with lid. Thermal properties: like melting temperature (Tm) and latent heat (ΔH), and cyclic stability of the obtained SS-PCM were determined by a DSC 204 F1 Phoenix NETZSCH, under a pure nitrogen atmosphere with constant gas volumetric flow of 20 mL min-1. Cyclic stability was determined by the DSC 204 Fl Phoenix NETZSCH, using different temperature ranges and cycle numbers for each sample (Table 1), to establish the opportunities of the developed SS-TES materials for LHS. The same experiments, specifically the thermal dehydration processes of the hydrates, were used to analyze the TCS potentiality of the materials.
3 Results and Discussion
Previously, a sol-gel technique was developed to obtain SS-PCMs based on Li salts [23]. Now, in this work, the previously developed method was applied to obtain SS-TES materials employing firstly inorganic salts and then, wastes from the non-metallic mining industry, such as bischofite and carnallite. The obtained SS-TES materials were engaged not only for LHS, but also for their potentiality as TCS materials.
3.1 Development of SS-TES materials employing commercial inorganic compounds
3.1.1 SEM-EDX and XRD characterization of SS-TES materials
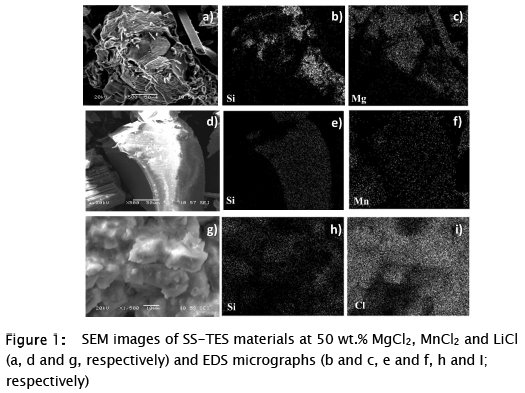
Firstly, 50 wt.% MgCl2·6H2O and 50 wt.% MnCl2 were prepared by the described method as two new SS-TES materials. In addition, a 50 wt. % LiCl based material was also synthetized as a reference material of the sol-gel procedure, according to a previous publication [23], and to compare with new magnesium and manganese based materials at the same salt content. The SEM images obtained for the MgCl2 based SS-TES material, showed separated phases: the SiO2 support material with the salt and segregated salt particles, which present an irregular morphology and a flat surface, respectively (Fig. 1 a, b and c). Smooth surface particles were obtained for the MnCl2 stabilized material, and the formation of detached salt particles from the stabilized material was also observed (Fig. 1 d, e and f). Conversely, the LiCl salt was found covering the entire surface of the polymer, as well as forming part of the same solid phase of these particles (Fig. 1 g, h and I) as was previously observed by Milian et al. [23]. The obtainment of separated phases by this sol-gel technique was associated with a high weight percentage (wt.%) of the TES material on the synthesis [23, 24].
3.1.2 Thermal and cycling performance of the SS-PCMs
The results of the thermal characterization of the synthesized compounds are summarized in Table 2. For LHS, it can be established that the LiCI SS-TES material melts and crystallizes at 607.8 and 600.9 °C, respectively; presenting a storage heat ΔHm of 188.7 kJ.kg-1 (Table 2), while its latent interval RL was close to 2 °C, comparable with previous work [23]. The manganese and magnesium chloride salts did not show a typical PCM behavior since no solidification occurred. MnCl2 SS-TES material melts and degrades at the same temperature, subsequently there was no solidification process that allows the reuse of the stored energy, while the SS-TES material of MgCl2.6H2O lost its water molecules, consequently, there was no fusion at low temperature or in the analyzed range (25 - 200 °C).
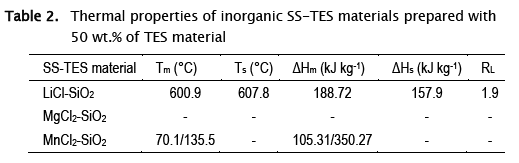
Based on the results, it was concluded that the method applied to Mg or Mn chlorides, under the experimental conditions of this work, did not developed SS-TES materials suitable for LHS, demonstrating that the success of the synthesis also depends on the nature of the PCM. Therefore, it was decided to carry out a deeper study with salts of industrial and mining interest: bischofite and carnallite as cases of study.
3.2 Application of the sol-gel method to wastes from non-metallic mining industry
The use of by-products obtained from mining industry as TES materials is of great interest due to its availability and continuous accumulation, mainly in northern Chile. Further benefits are that wastes from mining industries presented low costs and that their use decreases the environmental pollution caused by this sort of activity. The potential of bischofite and carnallite as TES materials has been evaluated by the "Thermal Energy Storage" research group of Center for Advanced Research in Lithium and Industrial Minerals (CELiMIN) [5,18].
Adittionally, the thermal process of the bischofite was presented according to the following decomposition reactions [10]:
Meanwhile, carnallite was decomposed according to the following global reaction, between 100 and 400 °C [26]:
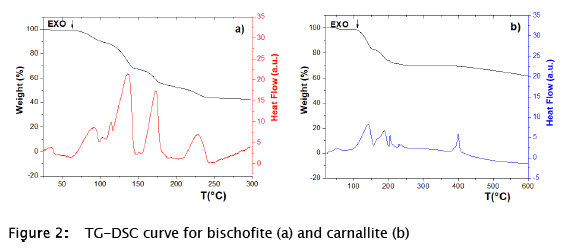
In our work, the carnallite and bischofite samples were analyzed by TG-DSC to have a reference of their thermal behaviour (Fig. 2) after their stabilization by SiO2. Both samples started decomposition processes at low temperatures close to 100 °C. Then, the sol-gel method was applied to obtain the SS-TES materials of these wastes in order to improve their thermal properties, mainly the thermal stability, in search of a possible application in TES systems.
3.2.1 SEM-EDX and XRD characterization of SS-TES materials
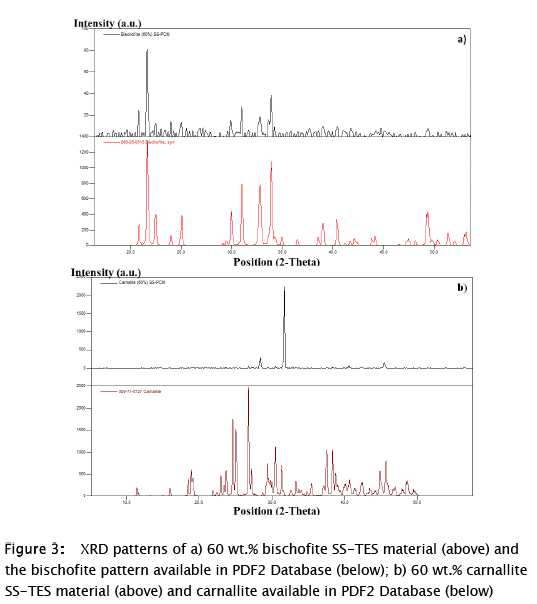
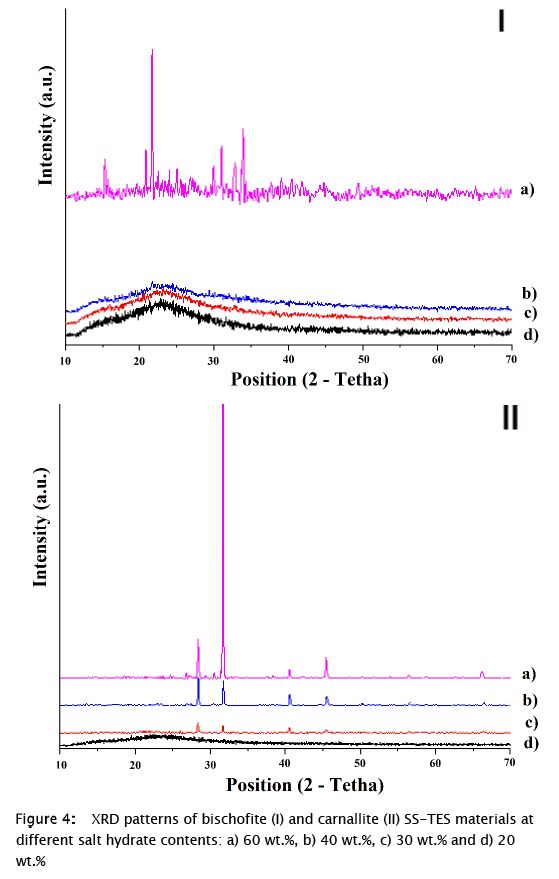
The XRD results for carnallite and bischofite SS-TES materials are presented in Fig. 3. The obtained patterns were compared with the XRD patterns of these materials available in the PDF2 database. The main peaks of bischofite were observed for 60 wt.% bischofite material (Fig. 3 a), therefore, these analyzes allowed to verify the recrystallization of this salt hydrate from synthesis solution. Although, no diffraction signals were obtained for samples at lower amounts of bischofite (Fig. 4 I b, c, d), due to the homogeneous dispersion of the salt hydrate into the silica SM, which is an amorphous material, as was previously reported [23,24]. Nevertheless, main peaks of pure carnallite (Fig. 3 b) were missing for carnallite SS-TES materials (Fig. 4 II). The absence of the main peaks of pure carnallite for carnallite SS-TES materials indicated the presence of a different solid phase agglomerated in the surface of the particles, which was corroborated later with SEM-EDS analysis.
On the one hand, it was confirmed that bischofite remained stable during the synthesis and in the final compound solidified as magnesium chloride hexahydrate. In addition, the appearance of the main peaks for the SS-TES material with 60 wt.% bischofite (21.64, 33.90, 30.98, 32.76, 20.83, 15.31, 29.92 and 49.41 °2θ, ordered by intensity) (Fig. 4 1A) indicates the formation of bischofite agglomerations during the synthesis at this weight percentage. On the other hand, the SS-TES materials of the bischofite at lower salt hydrate contents did not show signals in the diffraction patterns (Fig. 4 b, c, d), indicating that the bischofite was homogeneously distributed in the support material with no agglomerations.
On the other hand, peaks were obtained in the XRD pattern for all carnallite SS-TES materials (Fig. 4 II), except for the content of 20 wt.%. This SS-TES compound forms agglomerates at low percentages of the carnallite during synthesis, attributed to its lower solubility in the reaction medium, when compared with bischofite. However, the main peaks obtained for carnallite SS-TES materials (37.71, 28.38, 45.43 and 66.14 °2θ) do not correspond to the main signals corresponding to carnallite database pattern (26 % merit figure) (Fig. 3); indicating a possible dissociation, dehydration and / or non-agglomeration of an excess of the inorganic salt, after the condensation stage in the sol-gel process. The XRD pattern of 60 wt.% carnallite SS-TES material, obtained in this work, was compared with that obtained in a study of carnallite dehydration at 400 °C [5], both patterns showed the same signals (37.71, 28.38, 45.43 and 66.14 °2θ). The correspondence between these patterns was further evidence of the dehydration of the carnallite [5] when exposed to the sol-gel process.
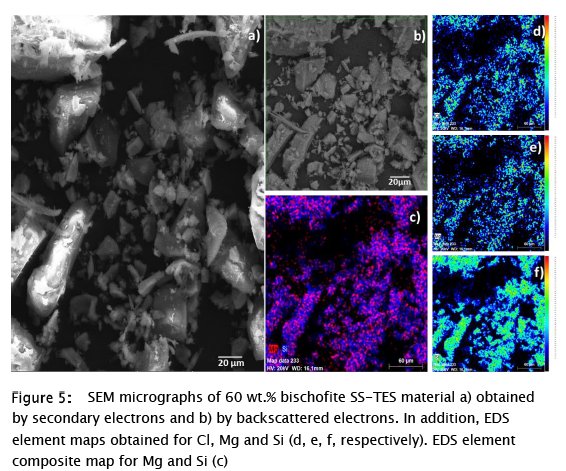
The samples were analyzed by SEM - EDS. In the case of the bischofite SS-TES materials, a fairly homogeneous distribution of the salt and the polymer was observed (Fig. 5), which was found not only in the elemental analyzes by EDS (Fig. 5 d, e, f), but it was also seen in the SEM image obtained with backscattered electrons (Fig. 5 b); where all the particles showed the same hue, that is, the same chemical composition. A combination of Mg and Si was observed, so it was concluded that the SiO2 polymer and the MgCl2 are in the same particles (Fig. 5 c).
SS-TES material of 60 wt.% carnallite (KMgCl3·6H2O) presented small particles adhered to the surface of the stabilized particles (Fig. 6 a). As can be observed, differences in composition appeared between these small particles and the stabilized ones (Fig. 6 b). The differences in composition were also confirmed thanks to the EDS maps, where Mg and K appear with greater intensity in different zones, indicating the segregation of carnallite into these cationic salts (Fig. 6 d and e). As well, the EDS mapping corroborated the presence of separated phases, since particles with bright yellow can be distinguished, which belong to K based material related to the mentioned small particles (Fig. 6 c). These results coincided with those obtained by XRD (Fig. 3 b), where the main peaks did not correspond to those present in the KMgCl3·6H2O pattern, but to the aggregated small particles on the surface of the material.

A deeper analysis by EDS allowed to identify the different phases appearing on the surface of the carnallite SS-TES material and the composition of the larger particles of this material. In this regard, different EDS points analyzes were performed (Fig. 7). Point 1 corresponds to adhered particles that, according to the images by backscattered electrons, have the same composition as the larger particle (point 3), while point 2 corresponded to particles with visibly different composition (Fig. 7 a). Elemental analyzes showed that points 1 and 3 are composed by the SS-TES material of KMgCl3·6H2O, while point 2 corresponds to a mix of KC1 and SiO2 mixture (Table 3). The partial dissociation of carnallite in potassium chloride (according to atomic percentage showed in Table 2 and XRD patterns (Fig. 4) was attributed to the dissolution of the salt during the sol-gel process and its subsequent separately recrystallization in its different components.

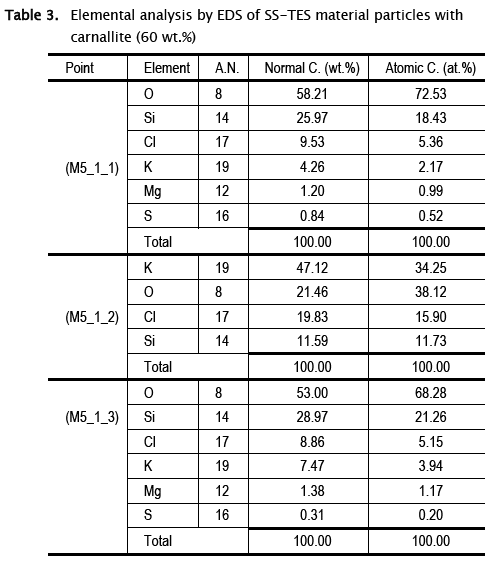
3.2.2 Thermal and cycling performance of the SS-PCMs
Once more, when thermal results were analyzed, the presence of the bischofite can be confirmed in the SS-TES materials by TG-DSC (Fig. 8 I). However, improvements in the thermal properties of bischofite-based materials were not achieved (Fig. 8) for LHS, since the process of heat store during phase change arose simultaneously with hydrate degradation (weight loss occurred). At the same time, the heat involved in the thermal dehydration process of these materials, increases with the increase in the amount of the hydrate added to the synthesis (Fig. 8 a, b, c).
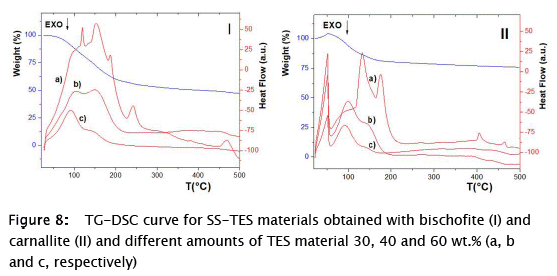
Cooling / heating studies were performed for these samples, but solidification process of the materials does not occur for any of the two salt hydrates. In addition, both SS-TES materials exhibit the same thermal degradation behavior as the pure salts, indicating that the used method did not influence or improve the heat storing process. Furthermore, bischofite does not undergo complete dehydration after the sol-gel synthesis process as in the case of previous work with Na2SO4.10H2O salt [24].
Finally, a single peak at 50 °C along with an increase in the mass of the material was observed for carnallite SS-TES material (Fig. 8 II), which may indicate water absorption by the carnallite, alike other reported works [9]. The peak at 50 °C in the carnallite samples was stabilized by the SiO2 particles (Fig. 8 II), being much higher than that of non-stabilized carnallite (Fig. 2 b). Water absorption process by carnallite could be favored when the salt was stabilized by the sol-gel method. In other words, the use of the applied sol-gel method results in the stabilization of carnallite, displaying a possible high potential for applications in storage of thermochemical energy (TCS), which most be further analyzed. Recently, similar works have been published which propose SS-TES materials for TCS and based on SiO2 as SMs. CaCl2 salt was stabilized in vermiculite, employing a laboratory scale reactor (127 cm3) with a scaled maximum power density around 150 kW m-3 [27]. Likewise, a new SS-TES material was developed by impregnation method for low-grade TCS; based on silica gel as SM and 30 wt.% of MgSC4·7H2O, Al2(SO4)3·18H2O and CuSO4·5H2O with high storage density values of 792.7, 580.5, and 712.6 J g-1, respectively [28]. As a result, further works could be directed to analyze the performance of the carnallite-SiO2 based SS-TES material and the operating settings, like partial pressure, temperature range, the chemical reversibility, dehydration and hydration isotherms and energy storage density, similar to the work published recently by Mamani and coworkers [29].
4 Conclusions
A one-step method based on a sol-gel process was applied to obtain SS-TES materials of commercial MgCl2·6H2O, MnCl2, LiCl and wastes from non-metallic mining industry: bischofite and carnallite. SEM analysis allowed to determine the morphology of samples and to analyze the achievement of shape stabilization of the TES materials. According to the TG-DSC performances, it was concluded that MgCl2 and MnCl2 based SS-TES materials did not crystalized, so, no heat storage occurs, under the experimental conditions; therefore, demonstrating that improvement of thermal properties by this sol-gel method for shape-stabilized materials also depends on the nature of the inorganic compound.
New stabilized materials based on bischofite and carnalite were obtained through the direct sol-gel process using TEOS as monomer and via acid hydrolysis. However, the cooling / heating studies allowed to prove that the solidification process of the SS-TES materials did not occur, which demonstrates the relevance of the chemical and thermal nature of the inorganic salt selected as TES material. Moreover, SS-TES materials exhibited the same thermal degradation behavior as the pure salts, which means that SiO2 as support material did not influence this thermal process. Therefore, the employed sol-gel technique did not improve the thermal properties of these materials for LHS using MgCl2, MnCl2, bischofite or carnallite, as occurs for LiCl. Conversely, the new material of carnallite / SiO2 showed a water absortion peak stabilized and enhanced by the presence of the SiO2 support material, which could indicate a high potential as a thermochemical storage medium. However, further studies related to the recovery of the stored heat by this carnallite silica TES materials should be performed in order to analyze it availability for TES applications, like the recycle of industrial waste heat or seasonal thermochemical storage applications.
Funding
The work was partially funded by the Chilean government ANID/FONDAP No 15110019, CONICYT/FONDECYT REGULAR N°1170675, CONICYT/PCI/REDES N° 170131, CONICYT/BECA DE DOCTORADO NACIONAL 2015 N°21150240, CORFO 16ENI2-71940 INGENIERIA2030 projects and ANID/FONDECYT DE POSTDOCTORADO 2020 N° 3200786.
References
[1] Alva G, Lin Y, Fang G (2018) An overview of thermal energy storage systems. Energy 144:341-378. [ Links ]
[2] Scapino L, De Servi C, Zondag HA, Diriken J, Rindt CC, Sciacovelli A. (2020). Techno-economic optimization of an energy system with sorption thermal energy storage in different energy markets. Appl Energy 258:114063. [ Links ]
[3] Mohamed SA, Al-Sulaiman FA, Ibrahim NI, Zahir MH, Al-Ahmed A, Saidur R, Sahin AZ (2017) A review on current status and challenges of inorganic phase change materials for thermal energy storage systems. Renewable Sustainable Energy Rev 70:1072-1089. [ Links ]
[4] Gutierrez A, Miró L, Gil A, Rodríguez-Aseguinolaza J, Barreneche C, Calvet N, Cabeza LF (2016) Advances in the valorization of waste and by-product materials as thermal energy storage (TES) materials. Renewable Sustainable Energy Rev 59:763-783. [ Links ]
[5] Gutierrez A, Ushak S, Mamani V, Vargas P, Barreneche C, Cabeza LF, Grágeda M (2017) Characterization of wastes based on inorganic double salt hydrates as potential thermal energy storage materials. Sol Energy Mater Sol Cells 170:149–59.
[6] Ushak S, Gutierrez A, Flores E, Galleguillos H, Grageda M (2014) Development of thermal energy storage materials from waste-process salts. Energy Procedia 57(0):627-632. doi: 10.1016/j.egypro.2014.10.217. [ Links ]
[7] Miró L, Navarro ME, Suresh P, Gil A, Fernández A I, Cabeza LF (2014) Experimental characterization of a solid industrial by-product as material for high temperature sensible thermal energy storage (TES). Appl Energy 113:1261-1268. [ Links ]
[8] Gasia J, Gutierrez A, Peiró G, Miró L, Grageda M, Ushak S, Cabeza LF (2015) Thermal performance evaluation of bischofite at pilot plant scale. Appl Energy 155:826-833. [ Links ]
[9] Gutierrez A, Ushak S, Linder M (2018) High Carnallite-Bearing Material for Thermochemical Energy Storage: Thermophysical Characterization. ACS Sustainable Chem Eng 6(5):6135-6145. [ Links ]
[10] Mamani V, Gutiérrez A, Ushak S (2018) Development of low-cost inorganic salt hydrate as a thermochemical energy storage material. Sol Energy Mater Sol Cells 176:346-356. [ Links ]
[11] Mamani V, Gutiérrez A, Fernández AI, Ushak S (2020) Industrial carnallite-waste for thermochemical energy storage application. Appl Energy 265:114738. [ Links ]
[12] Ushak S, Gutierrez A, Galleguillos H, Fernandez AG, Cabeza LF, Grágeda M (2015) Solar Energy Materials & Solar Cells Thermophysical characterization of a by-product from the non-metallic industry as inorganic PCM. Sol Energy Mater Sol Cells 132:385–91.
[13] Galazutdinova Y, Al‐Hallaj S, Grágeda M, Ushak S (2020) Development of the inorganic composite phase change materials for passive thermal management of Li‐ion batteries: material characterization. Int J Energy Res 44(3):2011-2022. [ Links ]
[14] Milián YE, Gutiérrez A, Grágeda M, Ushak S (2017) A review on encapsulation techniques for inorganic phase change materials and the influence on their thermophysical properties. Renew Sustain Energy Rev 73:983–99.
[15] Giro-paloma J, Martínez M, Cabeza LF, Fernández AI (2016) Types, methods, techniques, and applications for microencapsulated phase change materials (MPCM): A review. Renew Sustain Energy Rev 53:1059–75.
[16] Safari A, Saidur R, Sulaiman FA, Xu Y, Dong J (2017) A review on supercooling of Phase Change Materials in thermal energy storage systems. Renew Sustain Energy Rev 70:905–19.
[17] Galazutdinova Y, Grágeda M, Cabeza LF, Ushak S (2017) Novel inorganic binary mixture for low‐temperature heat storage applications. Int J Energy Res 41(14):2356-2364. [ Links ]
[18] Ushak S, Cruz MJ, Cabeza LF, Grágeda M (2016) Preparation and characterization of inorganic PCM microcapsules by fluidized bed method. Materials 9(1):24. https://doi.org/10.3390/ma9010024. [ Links ]
[19] Ling Z, Liu J, Wang Q, Lin W, Fang X, Zhang Z (2017) MgCl2·6H2O-Mg(NO3)2·6H2O eutectic/SiO2composite phase change material with improved thermal reliability and enhanced thermal conductivity. Sol Energy Mater Sol Cells 172:195–201.
[20] Fan S, Gao H, Dong W, Tang J, Wang J, Yang M, Wang G (2017) Shape‐stabilized phase change materials based on stearic acid and mesoporous hollow SiO2 microspheres (SA/SiO2) for thermal energy storage. Eur J Inorg Chem 14:2138-2143. [ Links ]
[21] Wang CL, Yeh KL, Chen CW, Lee Y, Lee HL, Lee T (2017) A quick-fix design of phase change material by particle blending and spherical agglomeration. Appl Energy 191:239–50.
[22] Wu Y, Wang T (2014) Preparation and characterization of hydrated salts / silica composite as shape-stabilized phase change material via sol – gel process. Thermochim Acta 2014;591:10–5.
[23] Milián YE, Reinaga N, Grágeda M, Ushak S (2020) Development of new inorganic shape stabilized phase change materials with LiNO3 and LiCl salts by sol-gel method. J Sol-Gel Sci Technol 94:22–33.
[24] Milian YE, Ushak S (2020) Design of synthesis route for inorganic shape-stabilized phase change materials. Direct sol–gel process versus vacuum impregnation method. J Sol-Gel Sci Technol 94:67–79.
[25] Xu Z, Yuan Z, Zhang D, Chen W, Huang Y, Zhang T, Sun Z (2018) Highly mesoporous activated carbon synthesized by pyrolysis of waste polyester textiles and MgCl2: Physiochemical characteristics and pore-forming mechanism. J Cleaner Prod 192:453-461. [ Links ]
[26] Ashboren D (1973) Carnallite decomposition into magnesia, hydrochloric acid and potassium chloride: A thermal analysis study; formation of pure periclase. J Appl Chem Biotechnol 23(1):77-86. [ Links ]
[27] Walsh S, Reynolds J, Abbas B, Woods R, Searle J, Jewell E, Elvins J (2020) Assessing the Dynamic Performance of Thermochemical Storage Materials. Energies 13(9):2202. https://doi.org/10.3390/en13092202. [ Links ]
[28] Ousaleh HA, Said S, Zaki A, Faik A, El Bouari A (2020) Silica gel/inorganic salts composites for thermochemical heat storage: Improvement of energy storage density and assessment of cycling stability. Materials Today: Proceedings. https://doi.org/10.1016/j.matpr.2020.04.354. [ Links ]
[29] Mamani V, Gutiérrez A, Fernández AI, Ushak S (2020) Industrial carnallite-waste for thermochemical energy storage application. Appl Energy 265:114738. https://doi.org/10.1016/j.apenergy.2020.114738. [ Links ]