Ecología en Bolivia
versión impresa ISSN 1605-2528versión On-line ISSN 2075-5023
Ecología en Bolivia vol.54 no.2 La Paz set. 2019
Nota
New records of the leaffish Monocirrhus polyacanthus (Perciformes, Polycentridae) in the upper Madeira River basin, Bolivia
Nuevos registros del pez hoja Monocirrhus polyacanthus (Perciformes, Polycentridae) en la cuenca alta del rio Madera, Bolivia
Gustavo Alvarez1*, Jaime Sarmiento2 & Guido Miranda-Chumacero1
1Wildlife Conservation Society - Bolivia, Calle Gabino Villanueva #340, entre las calles 24 y 25 de Calacoto, La Paz, Bolivia. *Corresponding author: Gustavo Alvarez, galvarez@wcs.org
2Museo Nacional de Historia Natural, Coleccion Boliviana de Fauna. Calle 27, Cota Cota, Campus Universitario, Casilla 8706, La Paz, Bolivia.
Recibido: 15.05.19, Aceptado: 20.07.19.
The Amazon leaffish, Monocirrhus polyacanthus Heckel 1840, belong to the family Polycentridae. South American polycentrids are small fishes reaching about 60-80 mm SL. The dorsal fin has 16-18 spines and 7-13 rays, the anal fin 12-13 spines and 7-14. There is no lateral line in the side (Britz & Kullander 2003). The family comprise three species: Polycentrus schomburgkii Müller & Troschel 1849 from coastal drainages of northeastern South America, P. jundia Coutinho & Wosiacki 2014 from the Rio Negro basin and Monocirrhus polyacanthus widely distributed throughout the Amazon and the Orinoco.
Monocirrhus polyacanthus is a small fish reaching approximately 80 mm of standard length (LS), with a remarkably laterally compressed body and long dorsal and anal fins. The mouth and head are characteristically large with a strongly protractile upper jaw and a mentonian filament resembling the petiole of a leaf (Eigenmann & Allen 1921, Britz & Kullander 2003, Nelson 2006). The color varies from orange-yellow to brown with remarkable mimicry through several markings that give the appearance of dry leaves (Fig. 1). The coloration can adapt to the environment and accentuates during reproductive periods.
The species prefer oxbow lakes and streams of black water rivers, with low pH (<5.0), low conductivity (<25 μS/cm), low sediment load, low concentration of nutrients, low current speed and high transparency. These streams are typically dark due to the decomposition of plants (Barros & Higuchi 2007). Adults of the species inhabit shallow waters, near fallen leaves on the surface, taking advantage of the leaf shape to feed on prey (Barros & Higuchi 2007).
The diet is dominated by fish (Characiformes, Perciformes) and lower proportions of invertebrates including crustaceans (Decapoda) and insects (Coleoptera, Hymenoptera, Ephemeroptera, Odonata) (Catarino & Zuanon 2010). Reproduction is unknown in the wild; the only available information comes from specimens observed in aquariums. M. polyacanthus has an aggressive courtship, with parental care of eggs and larvae by the male, while the female is responsible for driving away intruders (Barlow 1967).
The species is present throughout the Amazon basin and exploited by the ornamental fish trade (Pitman 2004). The presence of this species was usually reported in well-preserved forest streams (Oliveira et al. 2009). However, it is not as abundant as other species commonly found in the same habitat (Gutierrez 2003). Low abundance results in its rarity in fish collections and may explain the scarcity of information about its natural history and taxonomy. The group has never been thoroughly revised, and it is possible that there are more than one species in the genus (Britz & Kullander 2003).
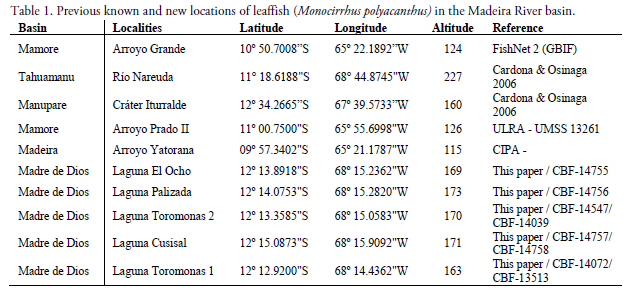
Monocirrhus polyacanthus was previously known from three localities in Bolivia (Cardona & Osinaga 2006). R.M. Bailey collected this species for the first time in Bolivia at the beginning of the 60 in the northeast of the country, 4 km from the border with Brazil (Cardona & Osinaga 2006). Only half a century later, the species was registered again, this time in the western part of the country, close to 200 km from the previous point (Cardona & Osinaga 2006) (Table 1, Fig. 2). According to these authors, a third specimen was later reported from the Nereuda river basin, in the extreme north of the country (Table 1, Fig. 2).
Here, we present new occurrence locations of Monocirrhus polyacanthus in the Madre de Dios River watershed in northern Bolivia. In addition, we conducted a review of the currently known distribution in South America, including the habitats in which leaf fish were captured.
Methods
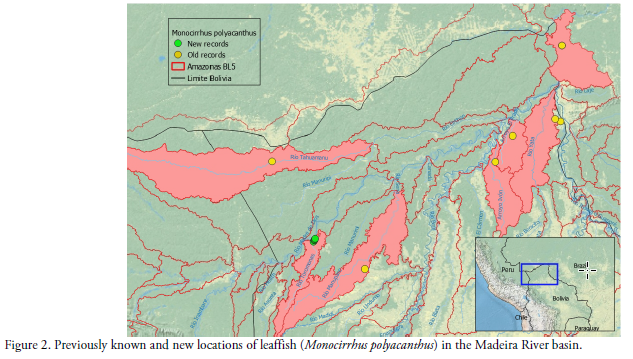
Specimens of the Amazon leaf fish were captured in oxbow lakes of the right margin of the Toromonas stream, a small tributary of the right margin of the Madre de Dios River (Table 1, Fig. 3). All localities are less than 200 m of elevation. The vegetation is a high pluvial forest more than 30 m high, with many types of swamp forest near the river or lagoons. (Fig. 4). Tall evergreen forest surrounds the lagoons that do not exceed three hectares, while depths reach a maximum of 4 m. The substrate is mostly muddy with leaves and submerged log (Fig. 3). Fieldwork was carried out between June-2011 and April-2012. The fish were caught with trawls of 2 and 5 meters in length, in different water bodies (lagoons) in the Toromonas stream that flow into the Madre de Dios River (Fig. 2).
Captured specimens were photographed alive in the field and preserved in 10% formaldehyde, and then deposited at the Coleccion Boliviana de Fauna under the collection codes: CBF-14755, CBF-14756, CBF-14547, CBF-14039, CBF-14757, CBF-14758, CBF-14072 and CBF-13513. For its later identification, cataloguing and final preservation in 75% alcohol. We collected the voucher specimens under scientific permits of the Direccion General de Biodiversidad - Bolivia (MMAyA-VMCC-DGBAP-UVSAP N° 463/09).
For taxonomic determination of the specimens we used the original description of the species (Heckel 1840), as well as Eigenmann & Allen (1921), in which the species Monocirrhus mimophyllus, currently considered a junior synonym of M. polyacanthus, is described. Monocirrhus polyacanthus is readily distinguished by the presence of a mental filament; ventral border of lacrimal and preopercular without serrations; marbled color pattern without horizontal series of points; 23-24 anal fin pterygiophores; % of caudal fin scaled; and posterior lateral line occasionally present in Monocirrhus (Coutinho & Wosiacki 2014). A strongly compressed body, very large protractile gape, and a color pattern mimicking dead leaves also characterize the species.
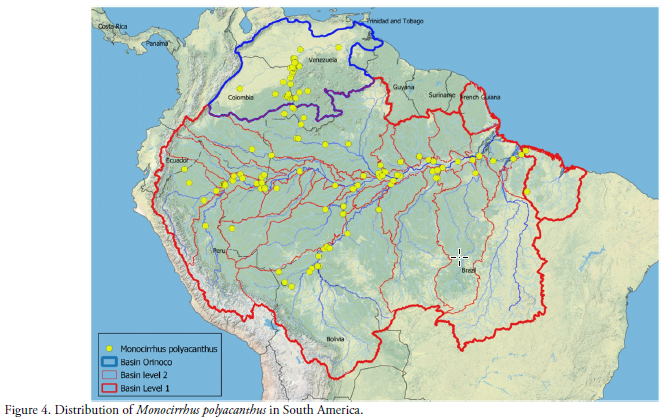
In order to complete the information on the known distribution in South America, we made a compilation of all reported localities that include the geographic location (latitude and longitude) in the Fishnet2 and GBIFdatabases (Fishnet2 2016, GBIF 2016), together with data from local publications and collections. Using the databases available in FishNet and GBIF as a baseline, together the records reported here, we produce an updated distribution map for the species, including 165 localities (Fig. 4).
Results
Fourteen water bodies were sampled, and twelve specimens of Monocirhus polyacanthus were found in five previously undocumented localities of the Madre de Dios River basin in northern Bolivia (Table 1, Figs. 1-2). The specimens of Monocirrhus from the upper Madeira River in Bolivia were found in small (< 3 ha) and shallow (< 4 m) black water lakes.
New records. Bolivia, La Paz, Laguna el ocho (12° 13.8918" S, 68° 15.2362" W), G. Alvarez coll., 07/07/2011, CBF-14755 (1). Bolivia, La Paz, Laguna Palizada (12° 14.0753" S, 68° 15.282" W), G. Alvarez coll., 08/07/2011, (CBF-14755 (1). Bolivia, La Paz, Laguna Toromonas 2 (12° 13.3585" S, 68° 15.0583" W), G.Alvarez coll., 05/07/2011, CBF-14547 (3). Bolivia, La Paz, Laguna Toromonas 2 (12° 13.3585" S, 68° 15.0583" W), G. Alvarez coll., 26/04/2012, CBF-14039 (1). Bolivia, La Paz, Laguna Cusisal (12° 15.0873" S, 68° 15.9092" W), G.Alvarez coll., 08/07/2011, CBF-14757 (4). Bolivia, La Paz, Laguna Cusisal (12° 15.0873" S, 68° 15.9092" W), G.Alvarez coll., 28/10/2011, CBF-14758 (1). Bolivia, La Paz, Laguna Toromonas 1 (12° 12.920" S, 68°14. 43.62" W), G. Alvarez coll., 27/04/2011, CBF-14O72(1).
Two further records of Monocirrhus polyacanthus were added for Bolivia: 1) the Yatorana stream in the Bruno Racua Wildlife Reserve of the Madeira River basin, from research carried out by Centro de Investigation para la Preservation de la Amazonia of the University of Pando (CIPA, UAP, H. Calderón, no collection number) and 2) in the Prado II stream of the Mamoré basin, based on studies by the Unidad de Limnología y Recursos Acuaticos of University of Cochabamba (ULRA, UMSS, M. Maldonado, collection number: UMSS 13261) (Table 1).
Discussion
The current distribution map of Monocirrhus polyacanthus in South America, allows us to identify three large "distribution areas" of the species. The most obvious corresponds to the Orinoco basin in Colombia and Venezuela. Secondly, the main stem of the Amazon from upper Peru to the east coast of Brazil (in which two secondary areas can be differentiated upstream and downstream). Thirdly the most marginal area that includes the southern isolated localities of the upper Madeira River in Bolivia (Figs. 2 & 4). The Monocirrhus polyacanthus record in the Toromonas stream basin is the southernmost of the species for the Madeira River basin (Table 1).
Britz and Kullander (2003) estimated that the family Polycentridae possibly includes more species than the currently accepted. However a recent revision of the genus Polycentrus, the other genus in Polycentridae, identified the presence of at least four three new species (Coutinho 2010).
Keeping in mind the wide extension of Monocirrhus, the presence of physical barriers that could favor the processes of speciation and isolation (Losos & Schluter 2000, Dias et al. 2013), the idea currently accepted that fish (and other groups) speciation in the Amazon could be an event as recent as Late Miocene (Hoorn et al. 2010, Lundberg et al. 2010); and lifting of Fitzcarrald Arch that has probably controlled the speciation of aquatic populations such as freshwater fish that began to diverge 4 My ago (Espurt et al. 2010, Hubert et al. 2007), it is possible to expect the presence of a hidden diversity in Monocirrhus.
The species would correspond, therefore, to the so-called forest species, for which the great white water rivers, characteristic of the upper Madeira River basin, represent insurmountable barriers, because the size of rivers and by the chemical nature of the waters (Weitzman & Weitzman 1982, Sistrom et al. 2009, Duncan & Fernandes 2010, Piggot et al. 2011).
A bibliographic review collected distributional information in the Madeira River basin and beyond, thereby detailing the known altitudinal gradient for the species, which runs from the Amazon estuary at sea level, to the highest point at 231 m elevation in the Venezuelan Orinoco. The highest records in the Amazon basin are presented in this study (170 and 229 m elevation).
The Monocirrhus polyacanthus record in the Toromonas stream basin is the southernmost of the species for the Madeira River basin (Table 1). This does not allow us to think that it could be a new genetic identity for Monocirrhus. Therefore, we suggest expanding biogeographic, genetics and classical morphology studies to determine the possibility of the existence of more than one species of M. polyacanthus in South America.
Acknowledgments
The authors would like to acknowledge the Tacana Paiche Association of Tacana II Indigenous Territory at Madre de Dios River, the Consejo Indígena del Pueblo Tacana (CIPTA). To Hailin Calderon of Centro de Investigation y Protection de la Amazonia, Universidad Autonoma de Pando, and Mabel Maldonado of Unidad de Limnologia y Recursos Acuaticos, Universidad Mayor de San Simon, for providing the data of the specimens collected in the Abuna and Manupare rivers. To Jansen Zuanon, Celio Magalhães and Renildo Ribeiro de Oliveira who allowed us to review the INPA's leaf fish collections. To Julio Pinto and Rubén Marín of Unidad de Limnología, Instituto de Ecología, Universidad Mayor de San Andrés for their constant support to field and lab work. To Soraya Barrera for support in identifying and cataloguing specimens. To Rob Wallace and Nuria Bernal for his review and help in translating the manuscript and Daniel Sanjines for the edition of the latest version of the maps. Finally, to the Gordon and Betty Moore Foundation for support to carry out the research efforts.
References
Banguera-Hinestroza, E., H. Cardenas, M. Ruiz-García, M. Marmontel, E. Gaitá, R. Vasquez & F. García-Vallejo. 2010. Molecular identification of evolutionarily significant units in the Amazon River Dolphin Inia sp. (Cetacea: Iniidae). The Journal of Heredity 93: 312–322.
Barros, B. & H. Higuchi. 2007. Notes on morphological characters in early-developed Amazonian leaffish, Monocirrhus polyacanthus (Polycentridae, Perciformes). Kempffiana 3(2): 18-22.
Barlow, G.W. 1967. Social behavior of a South American leaf fish, Polycentrus schomburgkii, with an account of recurring pseudofemale behavior. The American Midland Naturalist 78(1): 215-234. DOI: 10.2307/2423382 [ Links ]
Britz R. & S.O. Kullander. 2003. Family Polycentridae. Leaffishes. pp. 603-604. En: Reis, R.E., S.O. Kullander & C.J. Ferraris Jr. (Org.) Check list of the freshwater fishes of South and Central America. EDIPUCRS, Porto Alegre.
Catarino, M.F. & J.Zuanon.2010. Feeding ecology of the leaf fish Monocirrhus polyacanthus (Perciformes: Polycentridae) in a terra firme stream in the Brazilian Amazon. Neotropical Icthyology 8(1): 183-186. doi.org/10.1590/S1679-62252010000100022
Cardona, J.A. & K. Osinaga. 2006. Nuevo dato de la distribución de Monocirrhus polyacanthus Heckel, 1840 (Polycentridae, Perciformes) en Bolivia. Kempffiana 2(1):
57-59.
Coutinho, D.P. 2010. Revisão Taxonômica do Gênero Polycentrus Müller & Troschel, 1848 (Perciformes; Polycentridae) com a descrição de três novas espécies. Tesis de maestría, Universidade Federal de Pará / Museu Paraense Emílio Goelldi. Belém. 57 p. [ Links ]
Coutinho, D.P. & W.B Wosiacki. 2014. A new species of leaffish Polycentrus Muller & Troschel, 1849 (Percomorpha: Polycentridae) from the Rio Negro, Brasil. Neotropical Ichthyology 12(4):747-753. doi.org/10.1590/1982-0224-20140046
De Oliveira, R.R., M.S. Rocha, M.B. dos Anjos, J. Zuanon & L.H. Rapp Py-Daniel. 2009. Fish fauna of small stream of the Cauta Ipixuna Extractive Reserve, State of Amazonas, Brasil. Check List 5(2): 154-172.
Dias, M.S., J.F. Cornu, T. Oberdorff, C.A. Lasso & PA Tedesco. 2013. Natural fragmentation in river networks as a driver of speciation for freshwater fishes. Ecography
36: 683–689. doi.org/10.1111/j.1600-0587.2012.07724.x
Duncan, W.P. & M.N. Fernandes. 2010. Physicochemical characterization of the white, black and clearwater rivers of the Amazon Basin and its implications on the distribution of freshwater stingrays (Chondrichthyes, Potamotrygonidae). Pan-American Journal of Aquatic Sciences 5(3): 454-464.
Eigenmann, C.H. & W.R. Allen. 1921. A leaf mimicking fish. Biological Bulletin 41(5): 301–305.
Espurt, N., P. Baby, S. Brusset, M. Roddaz, W. Hermoza & J. Barbarand. 2010. The Nazca Ridge and uplift of the Fitzcarrald Arch: implications for regional geology in 167 northern South America. pp. 89-100. En: Hoorn C, & F.P. Wesselingh (Eds) Amazonia: Landscape and Species Evolution: A Look into Past. Wiley-Blackwell, Chichester, West Sussex. DOI: 10.1002/9781444306408.ch6
Farias, I.P., J.P. Torrico, C. García-Dávila, M.C.F. Santos, T. Hrbek & J-F. Renno. 2010. Are rapids a barrier for floodplain fishes of the Amazon Basin? A demographic study of the keystone floodplain species Colossoma macropomum (Teleostei: Characiformes). Molecular Phylogenetics and Evolution 56: 1129–1135.
Fishnet2. 2016. Fish specimen data used in this study obtained from the Academy of Natural Sciences at Philadelphia, California Academy of Sciences, Illinois Natural History Survey, UNELLEZ Museo de Zoología, MCZ-Harvard University, Swedish Museum of Natural History, Texas A&M University, Florida Museum of History, University
of Michigan Museum of Zoology, National Museum of Natural History, Smithsonian Institution. Accessed through the Fishnet2 Portal, www.fishnet2.org, 2016-10-10.
GBIF.org. 2016. GBIF Occurrence Download of Monocirrhus polyacanthus. Disponible en: http://doi.org/10.15468/dl.xnf85j. Accessed on: 2016-11-29. [ Links ]
Goulding, M., R. Barthem & E. Ferreira. 2003. The Smithsonian Atlas of the Amazon. Smithsonian Books, Washington and London, 253 p.
Gutiérrez, A.L. 2003. Análisis de algunos aspectos tróficos y reproductivos de la comunidad de peces de un caño de aguas negras amazónicas en cercanías de Leticia (Amazonas, Colombia). Unpublished Monograph, Universidad Nacional de Colombia, Bogotá, 132 pp.
Heckel, J.J. 1840. Johann Natterer´s neus Flussfische Brasilien´s nach den Beobachtungen und Mittheilungen des Entdeckers beschrieben (Erste Abtheilung, Die Labroiden). Annalen des Wiener Museums der Naturgeschichte 2: 325-471, Pls. 29-30. [ Links ]
Hoorn, C., F.P. Wesselingh, H. ter Steege, M.A. Bermudez, A. Mora, J. Sevink, I. Sanmartín, A. Sanchez-Meseguer, C.L. Anderson, J.P. Figueiredo, C. Jaramillo, D. Riff, F.R. Negri, H. Hooghiemstra, J. Lundberg, T. Staedler, T. Särkinen & A. Antonelli. 2010. Amazonia through time: Andean uplift, climate change, landscape evolution, and biodiversity. Science 330: 927–931. DOI: 10.1126/science.1194585
Hubert, N., F. Duponchelle, J. Nuñez, C. Garcia-Da Vila, D. Paugy & J-F. Renno. 2007. Phylogeography of the piranha genera Serrasalmus and Pygocentrus: implications for the diversification of the Neotropical ichthyofauna. Molecular Ecology 16(10): 2115-2136. doi: 10.1111/j.1365-294X.2007.03267.x.
Losos, J.B. & D. Schluter. 2000. Analysis of an evolutionary species–area relationship. Nature 408: 847–850. DOI: 10.1038/35048558
Lundberg, J.G., M.H. Sabaj Pérez, W.M. Dahdul & O.A. Aguilera. 2010. The Amazon Neogene fish fauna. pp. 281-301. En: Hoorn C. & F.P. Wesselingh (Eds) Amazonia: Landscape and Species Evolution: A Look into Past. Wiley-Blackwell, Chichester, West Sussex.
Nelson, J.S. 2006. Fishes of the World 4th ed. John Wiley and Sons, New York, 601 p.
Pearse, D.E., A.D. Arndt, N. Valenzuela, B.A. Miller, V. Cantarelli & J.W. Sites Jr. 2006. Estimating population structure under nonequilibrium conditions in a conservation context: continent-wide population genetics of the giant Amazon river turtle, Podocnemis expansa (Chelonia: Podocnemididae). Molecular Ecology 15: 985–1006.
Piggott, M.P., N.L. Chao & L.P. Beheregaray. 2011. Three fishes in one: cryptic species in an Amazonian floodplain forest specialist. Biological Journal of the Linnean Society, 102:391–403.
Pitman, N., R.C. Smith, C. Vriesendorp, D. Moskovits, R. Piana, G. Knell & T. Watcher (Eds) 2004. Perú: Ampiyacu, Apayacu, Yagus, Medio Putumayo. Rapid Biological Inventories Report 12. Chicago, Illinois: The Field Museum.
Sistrom, M.J., N.L. Chao & L.B. Beheregaray. 2009. Population history of the Amazonian one-lined pencilfish based on intron DNA data. Journal of Zoology 278: 287–298.
Torrente-Vilara, G., J. Zuanon, F. Leprieur, T. Oberdorff & P. Tedesco. 2011. Effects of natural rapids and waterfalls on fish assemblage structure in the Madeira River (Amazon Basin). Ecology of Freshwater Fish. 20: 588-597. DOI: 10.1111/j.1600-0633.2011.00508.x
Weitzman, S.H. & M.J. Weitzman. 1982. Biogeography and evolutionary diversification in Neotropical freshwater fishes with comments on the refuge theory. pp. 403-422. En: Prance, G.T. (Ed.) Biological Diversification in the Tropics. Columbia University Press, Nueva York.












 uBio
uBio