Services on Demand
Journal
Article
Indicators
Related links
Share
Ecología en Bolivia
Print version ISSN 1605-2528On-line version ISSN 2075-5023
Ecología en Bolivia vol.52 no.2 La Paz Sept. 2017
Artículo
The diets of cattle and guanaco in the relict Chacoan savannahs of Bolivia
Dietas de Ganado y guanacos en sabanas relictuales del Chaco de Bolivia
Erika Cuéllar Soto*, Paul J. Johnson & David W. Macdonald
University of Oxford, Dept Zoology, Wildlife Conservation Research Unit, Recanati Kaplan Centre, Tubney OX13 5QL, Oxon, United Kingdom, *Corresponding author: erika.cuellar71@gmail.com
Abstract
Overlap in resource use between cattle and wild herbivores have implications for conservation, particularly if food is limited. The aim of this paper is to investigate the diets of a wild and a domestic herbivore living in sympatry in plant communities degraded by woody encroachment. Dung samples from both species were subjected to microhistological analysis, making of this work the first study describing and comparing the diet of sympatric guanacos and cattle in the Chaco region. We found a relatively high richness of 53 plant species in the diets of both herbivores, and 4 species were exclusive to cattle. A clear separation between the diets of guanaco and cattle was observed at two levels using analysis of similarity (ANOSIM): a) species level (r=0.52, p=<0.0001) and b) at ecological group level (r= 0.3312, p=<0.001). Cattle appear to forage mostly on the colonial woody species mainly responsible for the reduction of open areas while guanaco diets include more of the native grass most characteristic of the shrinking Chacoan savannahs. Our study raises questions for further research that could form core elements of future conservation plans for the guanaco population in the study area and beyond.
Keywords: Gran Chaco, Herbivores, Savannah, South America, Woody encroachment.
Resumen
La superposición en el uso de recursos entre el ganado bovino y los herbívoros silvestres tiene implicaciones para la conservación, particularmente si el alimento es limitado. El objetivo de este trabajo es investigar las dietas de un herbívoro silvestre y uno doméstico que viven en simpatría en comunidades vegetales degradadas por la invasión leñosa. Las muestras de heces de ambas especies fueron sometidas a análisis microhistológicos, siendo este trabajo el primer estudio que describe y compara la dieta de guanacos simpátricos con el ganado en la región de Chaco. Encontramos una riqueza relativamente alta de 53 especies de plantas en las dietas de ambos herbívoros, donde 4 especies fueron exclusivas en la dieta del ganado. Se observó una separación clara entre las dietas de guanaco y ganado en dos niveles usando el análisis de similitud (ANOSIM): a) nivel de especie (r = 0.52, p = <0.0001) y b) a nivel de grupo ecológico (r = 0.3312, p = <0.001). El ganado parece forrajear sobre todo en las especies leñosas coloniales principalmente responsables de la reducción de áreas abiertas, mientras que la dieta del guanaco incluye más la gramínea nativa, cuya perdida está caracterizando la reducción de las sabanas chaqueñas. Nuestro estudio plantea preguntas para futuras investigaciones que podrían formar elementos básicos de futuros planes de conservación para esta población de guanacos y otras especies aún existentes en el área de estudio.
Palabras clave: Gran Chaco, herbívoros, sabana, América del Sur, arborización.
Introduction
With intensifying land use in many South American regions, conflict between cattle ranching and the conservation of habitat for wild, grazing herbivores prompt to a potential conflict (Veblen & Young 2010). Savannahs are suitable for livestock (Scholes & Walker 1993) and increasingly shared by wild and domestic mammalian herbivores (Hopkins 1954, Weaver & Bruner 1984). However, several factors are causing the conversion of semi-arid savannahs to shrublands or woodlands, with a progressive loss of savannah structure and species composition (see for example Sankaran et al. 2005, Van Auken 2009, Wigley et al. 2010).
The reduction of natural savannahs in the driest part of the Gran Chaco in eastern Bolivia is mainly caused by the intensification of cattle ranching and the land management practices which has consequences on the vegetation structure in the form of woody encroachment (Navarro 2002, Cuéllar 2011). Furthermore, this woody encroachment is an increasing pressure on the natural habitat for a large and endangered wild herbivore, the guanaco (Lama guanicoe). In the Gran Chaco biome, guanacos are distributed mainly in private ranches surrounding the southern corner of the Kaa-Iya National Park. Guanaco and livestock (especially cattle) are sympatric in this area of less than 60,000 ha of the fragmented savannahs (Cuéllar & Núñez 2009). Even though our study was not focused on confirming any kind of preference or competition between these two ungulates, on one hand our expectation was to find some indication of overlapping in terms of food given previous observation of distribution overlapping. On the other hand, we were interested to confirm the consumption or not of the colonial woody species, P. chacoense by cattle and or guanacos. Studies on guanaco diet where guanaco and livestock are sympatric have described dietary flexibility of guanacos related to the seasonal shift in availability of food (Puig et al. 2011) or consisting mainly of grasses and forbs, i.e. in central Chile (Simonetti & Fuentes 1985) and northern Peru (Franklin 1975), as well as in the Magellanic forest-steppe of Chile's southern Patagonia (Raedeke 1979, 1980). In contrast, Bahamonde et al. (1986) described guanacos living in the shrub-grasslands of northern Argentina as browsers. Although cattle are considered to be grazers, according to the morphological adaptations of their digestive systems (following Hofmann 1989), they can browse depending on food availability (Bahamonde et al. 1986).
The aim of this paper is to investigate the diets of a wild and a domestic herbivore living in sympatry given that there are no previous studies on the diet of sympatric cattle and guanaco in the Chaco region where the structure and composition of the plant communities of the tropical dry forest greatly differ from the above sites. With the purpose of given a better understanding on the vegetation structure in the study area we describe the composition of the different plant communities within the savannah, which is currently a complex mosaic of successional stages.
In this work we describe sympatric cattle and guanaco diet by subjecting dung samples from both species to microhistological analysis (Baumgartner & Martin 1939). Based on our results We suggest the potential implications of cattle and guanaco interactions in the Chaco region in the context of grassland reduction and land management, as part of a conservation plan for the fringe guanaco population surviving in eastern Bolivia. The results of this work could be used as the baseline for further experimental studies on potential competition for food between cattle and guanaco and others such as resource selection or specific preferences within each species and between them.
Methods
Study area
The study area lies within the alluvial plain of the Parapetí River and presents a unique formation of savannahs on sandy soils of palaeodunes. It extends from 19°45'-20°30'S and from 62°00'-63°00'W in the extreme south of the Santa Cruz department of Bolivia, and includes the southwest corner of the Kaa-Iya National Park. The climate is predominantly semi-arid (Peel et al. 2007), with annual rainfall ranging from 200 mm in the extremely arid west (Taber et al. 1997) to 750 mm. The average annual temperature is 25-26°C, with below-freezing temperatures in June-August, while +40°C in September-November (Navarro & Fuentes 1999). The vegetation consists of a mosaic of savannah, scrub secondary forest, and remnant primary forest on well-drained soils, taxonomically defined as the "Aridic Haplustoll" (Abril & Bucher 2001).
Vegetation communities
The vegetation is dominated by a vegetation mosaic composed mainly by four main communities with relative abundances as follow: grasslands (0.2%), scrubland (15.5%), shrubland and woody vegetation (72%) (Cuéllar 2011). The strata vary between 0.40 and 4 metres. Grassland was dominated by two of the 50 species recorded: with Aristida mendocina (Poaceae) making up the 35% and Lippia spp.(Verbenaceae)the56%oftherelative abundance with plants varying between 40 cm and 120 cm tall. Scrubland was dominated by Lippia spp. making up the 70% of the relative abundance, in contrast to the other 59 species recorded with plants up to approximately 140 cm tall. And shrubland, dominated by three species of the total 31 recorded: both Fabaceae (Mimosoideae) Pithecellobium chacoense 49%, Mimosa castanoclada 12%, and Bougainvillea sp. (Nyctaginaceae) 12%, with plants up to approximately four metres tall.
Dung collection
Guanaco and cattle dung samples were collected during one wet season from November 2008 to April 2009 at different sites in the study area, where both species were known to coexist. All transects were intensively surveyed on the same day, during the study period and therefore we assumed the faeces corresponded to different individuals. Only fresh dung (easily recognised by their moisture and dark colour) samples were collected. The samples were air-dried, preserved in alcohol (70%) and stored individually labelled plastic jars. Once in the lab (botanical laboratory, Gabriel Rene Moreno University, Bolivia) the dung samples were washed and oven-dried at 60°C, and ground to particles <1mm using a mill. Following standard fixing and staining procedures (Williams 1962) dung samples were analysed micro-histologically following Baumgartner and Martin (1939) for further species identification (Sparks & Malechek 1968).
Botanical composition of diet
Plant species in the dung samples were identified using a micro-histological plant database constructed by oven-drying, grinding and slide-mounting 79 plant species and then determining the patterns of their epidermal and cuticular structures (Williams 1962). These specimens were fixed on histological plates and photographed as part of the preparation of a visual key. Diet compositions for guanaco and cattle were determined from 29 and 25 dung samples, respectively. A total of 204 microscope slides, each bearing 20 observation fields, were observed under x 100 magnification (Baldi et al. 2004). The first 21 samples analysed were subdivided into five slides per sample, and then we reduced the number of slides to three on the following 33 samples. A total of 1,740 and 2,340 fields were observed for guanaco and cattle samples, respectively. In addition, we plotted cumulative number of plant species making up the diets of guanaco and cattle against the cumulative number of fields analysed. It showed that 340 fields appeared to be sufficient to obtain a reliable estimate of guanaco diet characteristics (the curve levels off around this value). In contrast, 1,340 fields were necessary to achieve an estimate for cattle samples. Relative frequency of plant species occurrence in the diets of guanacos and cattle was determined for each sample by dividing the number of microscopic fields in which a given species occurred by the sum total of frequencies for all species identified (Holechek & Gross 1982). Plant species were grouped in five categories: grasses, woody species, climbers, succulents, and forbs. We grouped the species found in the microhistological analysis in these five ecological groups to facilitate comparison with other studies.
Analysis of similarities (ANOSIM)
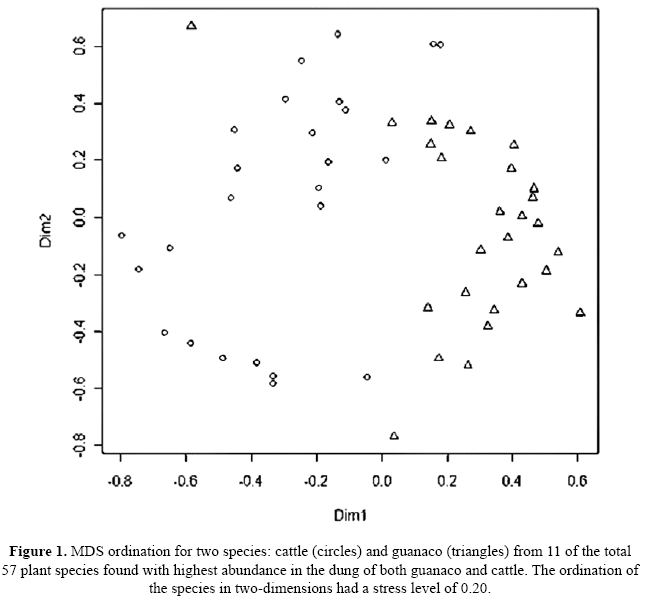
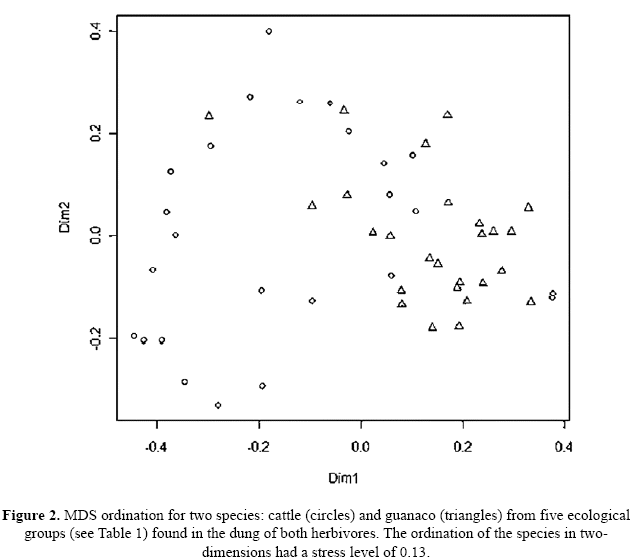
The Bray-Curtis similarity coefficient (unweighted group-average method) was used to construct a similarity matrix among dung samples. This matrix was subjected to Analysis of similarities (ANOSIM), to test whether there was a significant difference in the species composition consumed between guanaco and cattle using R software, Vegan package (R.Development.CoreTeam 2010). An analysis of similarities was performed for 11 (highlighted in Table 1) of the total 57 plant species found with highest abundance in the faeces of both guanaco (56%) and cattle (66%). The justification for using only 11 taxa is that the remainder were extremely scarce, from the total of the plant species present in both cattle and guanaco diets, only species with relative abundances above 4% were included in the analysis. In addition, we undertook the same analysis of similarity for the five ecological groups: grasses, succulents, forbs, climbers, and woody species (Table 1). To visualise the difference between cattle and guanaco diets, we applied non-metric multidimensional scaling (NMDS) to the similarity matrix using the isoMDS option in the R MASS package (Venables & Ripley 2002).
Results
A total of 57 plant species (from 31 families) were found in the dung analysis: 53 plant species were shared by both guanaco and cattle dung, and 4 species were exclusive to cattle. Of these, 45 plants were identified to the species level, seven to the genus level, and five to an ecological group level. For guanacos, the climber species Urvillea chacoensis (Sapindaceae) was present in 100% of the samples (n=29), followed by two woody species Ximenia americana (Olacaceae, 86%) and Celtis chichape (Olacaceae), a forb Angelphytum pseudosilphioides (Asteraceae) and a grass Aristida mendocina (each of the latter in 83% of the total samples from guanaco).
For cattle, the grass Chloris castilloniana (Poaceae) was present in 100% of the cattle samples (n=25), followed by the most successful woody pioneer species colonizing the open areas Pithecellobium chacoense, present in 96% of the cattle samples, and then by Agonandra sp. (Opiliaceae), Angelphytum pseudosilphioides, and Urvillea chacoensis (each of these present in 80% of the cattle samples).
In addition, we found that the total plant species recorded in the diet of both guanacos and cattle included 73.4% of plant species present in our database.
Diet characteristics of guanacos and cattle
There was a clear separation between the dung samples per species for guanaco and cattle (Fig. 1). The ANOSIM indicates that the main differences between guanaco and cattle were due to differential intake of Aristida mendocina, Pithecoctenium sp. (Bignoniaceae), Celtis chichape, Boungainvillea infesta (which are higher for guanaco) and Chloris castilloniana, Agonandra sp. and Pithecellobium chacoense (which are higher for cattle as compared to guanaco (r=0.52, p=<0.0001).
The main ecological groups in the diets of guanacos and cattle
The main differences between guanaco and cattle were due to differential intake of grasses (Aristida mendocina higher for guanaco and Chloris castilloniana higher for cattle), forbs and climbers (which are higher for guanaco) and woody species (which are higher for cattle as compared to guanaco). The ANOSIM performed for the five ecological groups derived from the plant species (Table 1) consumed by guanacos and cattle indicates the separation between the ecological categories of plants consumed by both species (r= 0.3312, p=<0.001). The ordination plot shows a separation of the ecological groups between guanaco and cattle (Fig. 2).
Discussion
We analyzed and compared the diet of sympatric guanacos and cattle in the Chacoan savannah, where woody encroachment is colonizing the open habitat. P. chacoense is the dominant species forming shrub aggregation in open areas. Our results showed that while a diverse range of plant species occurred in the diet of both cattle and guanacos, only a few dominated the diets of these herbivores (Table 1). Our expectation to find some indication for potential competition for food between cattle and guanaco was not confirmed, instead our results showed a clear separation between species and group of species consumed by these two herbivores.
This resource partitioning observed between cattle and guanaco is not completely consistent with the species-specific digestive traits given the morphological adaptations of their digestive systems (Hofmann 1989) and the fact that both species can be grazers or browsers depending on food availability (Bahamonde etal. 1986). Previous observations in the field suggested that both species were mainly browsers (Bahamonde et al. 1986). However, according to our results guanacos were mainly grazers and a possible explanation to what we observed when browsing woody species was in fact that guanacos were eating a climber species Urvillea chacoensis (Table 1) which appeared relatively frequent in its diet.
In terms of land management our results contradict what some ranchers affirmed regarding the non-consumption of the colonial woody species, P. chacoense by cattle. In fact, cattle ranchers used fire in the past to avoid the expansion of this colonial species (Navarro 2002). However, preliminary observations of the dominant shrub formations showed that almost 100% of the standing plants forming the shrub aggregation presented a burning signal in the base of the plant. Additionally, a preliminary experimental fire management showed that P. chacoense regenerated even after the aerial parts of plants had been completely burned (Cuéllar 2011). The fact that 96% of the cattle samples contained this colonial species, which is native and restricted to some Chaco areas in northern Argentina, northern Paraguay and southern Bolivia (Prado 1998), combined to the inadequate land management used in the region is a matter requiring further investigation. The introduction of livestock in the Chaco is considered to be one of the main factors leading to the progressive decline in numbers and the reduction of the geographical range of guanacos (Cuéllar 2011). Despite the evidence that wildlife - livestock interactions are to a large extent governed by exploitation competition (e.g. Coe et al. 2001, Stewart et al. 2002), it is possible that in the Chaco region cattle promote the expansion of a woody species and therefore the transformation of the structure and composition of the Chacoan savannahs, rather than inducing direct competition with guanacos for the same food resources. This is counter to the general assumption that where there are introduced herbivores sympatric with wild herbivores competition will result (Belovsky 1986, Rebollo et al. 1993, Mysterud 2000, Baldi et al. 2004). In contrast to cattle consumption of woody species, guanacos largely consumed the native grass A. mendocina (Poaceae), which has shrunk in distribution by 90% in this region over the last 40 years (Pinto 2005) and is gradually being replaced by an invasive forb, Lippia sp. (Navarro 2002). Therefore, in terms of strategies for promoting the conservation of the Chacoan savannah and the wildlife depending on it, experimental studies on the interaction between cattle and this woody species, which is colonizing the remaining open areas, should contribute to our understanding on the current approaches of land management. In addition, monitoring the effects of grazing by both guanaco and cattle on the current areas of grasslands is required to find out if the depletion of A. mendocina could favour the establishment of Lippia spp. and woody species. Similar situation has been previously observed in an arid region where the woody plant Terminalia sericea contributed to cattle diet, while at the same time constituting the major encroaching species in savannahs (Katjiua & Ward 2006). Finally, we emphasise the advantages of working with landowners in order to help to reduce woody expansion.
Acknowledgments
We are grateful to the Botanical Laboratory staff at the Gabriel Rene Moreno University in Bolivia and Mercy Lopez for her invaluable support. This is a chapter of a doctoral thesis of EC at Oxford University with the support of Wildlife Conservation Society, Gordon and Betty Moore Foundation within the Amazon-Andes Conservation Program (AACP) "Conserving Amazonian Landscapes". Other sponsors that supported the guanaco project were: The Shared Earth Foundation, Whitley Fund for Nature, The Rolex Award for Enterprise, Kaa-Iya Foundation.
References
Abril, A. & E.H. Bucher. 2001. Overgrazing and soil carbon dynamics in the western Chaco of Argentina. Applied Soil Ecology 16: 243-249.
Avila, J. 2002. Ecologia del fuego, Parque Nacional y ANMI Kaa-Iya del Gran Chaco. Edafologia y Forrajes. pp 122-138. En: Guerrero, J. (ed.), Ecología del fuego en el Parque Nacional y Área Natural de Manejo Integrado Kaa-Iya del Gran Chaco. Proyecto Kaa-Iya, Santa Cruz.
Bahamonde, N., S. Martin & A. Sbriller. 1986. Diet of guanaco and red deer in Neuquen Province, Argentina. Journal of Range Management 39: 22-24.
Baldi, R., A. Pelliza-Sbriller, D. Elston & S. Albon. 2004. High potential for competition between guanacos and sheep in Patagonia. Journal of Wildlife Management 68: 924-938.
Baumgartner, L.L. & A.C. Martin. 1939. Plant histology as an aid in squirrel food-habit studies. Journal of Wildlife Management 3: 266-268.
Belovsky, G.E. 1986. Generalist herbivore foraging and its role in competitive interactions. American Zoologists 26: 51-69.
Brower, J.E., J.H. Zar & C.N. von Ende. 1990. Field and laboratory methods for general ecology, 3rd ed. Wm. C. Brown, Dubuque. 288 p.
Bullock, J.M. 2006. Plants. pp. 186-213. In: Sutherland., W J. (ed.) Ecological Census Techniques. Cambridge University Press, Cambridge.
Coe, K.P., B.K. Johnson, J.W. Kern, S.L. Findholt, J.G. Kie & J.M. Wisdom. 2001. Responses of elk and mule deer to cattle in summer. Journal of Rangeland Management 54: 51-76.
Cuéllar, E. 2011. Ecology and conservation of the guanaco Lama guanicoe in the Bolivian Chaco: habitat selection within a vegetation succession. University of Oxford, Oxford. 178 p. [ Links ]
Cuéllar, E.& A. Núñez. 2009. Lama guanicoe (Müller, 1776 ): Artiodactyla-Camelidae. pp. 454-456. In: Aguirre, L.F., R. Aguayo, J. Balderrama, C. Cortéz, T. Tarifa (eds.). Libro Rojo de la Fauna Silvestre de Vertebrados de Bolivia. Ministerio de Medio Ambiente y Agua de Bolivia, La Paz.
Dearden,B.L.,R.E.Pegau&R.M.Hansen.1975. Precision of microhistological estimates of ruminant food habits. Journal of Wildlife Management 39:402-407.
Franklin, W., 1975. Guanacos in Peru. Oryx 13:191-202.
Fraser, M.D.& I.J. Gordon. 1997. Organic matter intake, diet digestibility and feeding behaviour of goats, red deer and South American camelids feeding on three contrasting Scottish vegetation communities. Journal of Applied Ecology 34: 687-698.
Hofmann, R.R. 1989. Evolutionary steps of ecophysiological adaptation and diversification of ruminants - a comparative view of their digestive-system. Oecologia 78:443-457.
Holechek, J. & B. Gross. 1982. Evaluation of different calculation procedures for microhistological analysis. Journal of Range Management 35: 721-723.
Hopkins, H.H. 1954. Effects of mulch upon certain factors of the grassland environment. Journal of Rangeland Management 7: 255-258.
Katjiua, M.L.J. & D. Ward. 2006. Cattle diet selection during the hot-dry season in a semi arid region of Namibia. African Journal of Range & Forage Science 23: 59-67.
M.L., Mcinnis. Vavra & W.C. Krueger. 1983. A comparison of four methods used to determine the diets of large herbivores. J. Range Manage. 36:302-306. [ Links ]
Mohammad, A.G., R.D. Pieper, J.D. Wallace, J.L. Holechek & L.W. Murray. 1995. Comparison of fecal analysis and rumen evacuation techniques for sampling diet botanical composition of grazing cattle. Journal of Range Management 48: 202-205.
Mysterud, A. 2000. Diet overlap among rumiants in Fennoscandia. Oecologia 124:130-137.
Navarro, G., 2002. Ecología estructural y dinámica de áreas quemadas en el PN-ANMI Kaa-Iya del Gran Chaco. pp 5-74. In: Guerrero, J. (ed.), Ecología del fuego en el Parque Nacional y Área Natural de Manejo Integrado Kaa-Iya del Gran Chaco. Proyecto Kaa-Iya, Santa Cruz.
Navarro, G.& A. Fuentes. 1999. Geobotánica y sistemas ecológicos de paisaje en el Gran Chaco de Bolivia. Revista Boliviana de Ecología y Conservación Ambiental 5: 25-50.
Peel, M.C., B.L. Finlayson & T.A. McMahon. 2007. Updated world map of the Koppen Geiger climate classification. Hydrology and Earth System Sciences 11:1633-1644.
Pinto, C. 2005. Análisis multitemporal de cambios de vegetación de la pampa chaqueña, en la zona de los arenales de Yanahigua, oeste del Parque Nacional Kaa-Iya. Technical report 87. Proyecto Kaa-Iya, Santa Cruz. 61 p.
Puig, S., M.I. Rosi, F. Videla & E. Mendez. 2011. Summer and winter diet of the guanaco and food availability for a High Andean migratory population (Mendoza, Argentina). Mammalian Biology 76: 727-734.
Prado, D. 1998. Chloroleucon chacoense. The IUCN Red List of Threatened Species 1998. http://dx.doi.org/10.2305/IUCN.UK.1998.RLTS.T36570A10006310.en.
R.Development.CoreTeam. 2010. R software, Vegan package in: Computing, R.F.f.S. (Ed.),Vienna,Austria,p. R:a languageand environment for statistical computing.
Raedeke, K. 1979. Population dynamics and socioecology of the guanaco (Lama guanicoe) of Magallanes, Chile. University of Washington, Seattle. 409 p. [ Links ]
Raedeke, K. 1980. Food habits of the guanaco (Lamaguanicoe) of Tierra del Fuego, Chile. Turrialba 30: 309-314.
Rebollo, S., L. Robles & A. Gomez-Sal. 1993. The influence of livestock management on land use competition between domestic and wild ungulates: sheep and chamois Rupicapra pyrenaica parva Cabrera in the Cantabrianrange. Pirineos(141)142:47-62.
San Martín, F.& F.C.Bryant. 1987. Nutrición de los camélidos sudamericanos: estado de nuestro conocimiento. Texas Tech University, Texas. 67 p.
Sankaran, M., N.P. Hanan, R.J. Scholes, J. Ratnam, D.J. Augustine, B.S. Cade, J. Gignoux, S.I. Higgins, X. Le Roux, F. Ludwig et al. 2005. Determinants of woody cover in African savannas. Nature 438: 846-849.
Scholes, R.J.& B.H.Walker. 1993. Cambridge studies in applied ecology and resource management: an African savanna. Synthesis of the Nylsvley study. Cambridge University Press; Cambridge University Press. 320 p.
Schwarm, A., S. Ortmann, C. Wolf, W.J. Streich & M. Clauss. 2008. Excretion patterns of fluid and different sized particle passage markers in banteng (Bos javanicus) and pygmy hippopotamus (Hexaprotodon liberiensis): Two functionally different foregut fermenters. Comparative Biochemistry and Physiology a-Molecular & Integrative Physiology 150: 32-39.
Simonetti, J.A.& E.R. Fuentes. 1985. Guanacos versus cabras y conejos como moduladores del matorral de Chile central. pp. 76-87. In: Venegas, C., Cunazza, C. (eds.), Actas de la IV Convención Internacional sobre Camélidos Sudamericanos. Punta Arenas.
Sparks, D.R.& J.C. Malechek. 1968. Estimating percentage dry weight in diets using a microscopic technique. Journal of Range Management 21: 264-265.
Stewart, K.M.,R.T. Bowyer, J.G. Kie,N.J. Cimon & B.K. Johnson. 2002. Temporospatial distributions of elk, mule deer, and cattle: resource partitioning and competitive displacement. Journal of Mammalogy 83: 229-244.
Tabachnik, B.G.& L.S. Fidell. 2012. Using multivariate statistics (6th edition). Pearson Education, Boston. 1024 p.
Taber, A., G. Navarro & M.A. Arribas. 1997. A new park in the Bolivian Gran Chaco - an advance in tropical dry forest conservation and community-based management. Oryx 31:189-198.
VanAuken,O.W.2009. Causes and consequences of woody plant encroachment into western North American grasslands. Journal of Environmental Management 90: 2931-2942.
Vavra, M.& J.L. Holechek. 1980. Factors influencing microhistological analysis of herbivore diet. Journal of Range Management 33: 371-374.
Veblen, K.E. & T.P. Young. 2010. Contrasting effects of cattle and wildlife on the vegetation development of a savanna landscape mosaic. Journal of Ecology 98: 993-1001.
Venables, W.N.& B.D. Ripley. 2002. Modern applied statistics with S. Fourth Edition, ed. Springer, New York. 498 p..
Weaver, J.E.& W.E. Bruner. 1984. Prairies and pastures of the dissected loess plains of central Nebraska. Ecological Monographs 18: 507-548.
Wigley, B.J.,W.J. Bond & M.T. Hoffman. 2010. Thicket expansion in a South African savanna under divergent land use: local vs. global drivers? Global Change Biology 16: 964-976.
Williams, O.B. 1962. A technique for studying microtine food habits. Journal of Mammalogy 43: 365-368.
Artículo recibido: 4 marzo 2017
Aceptado en: 25 mayo 2017
Manejado por: Daniel Larrea












 uBio
uBio