Ecología en Bolivia
versión impresa ISSN 1605-2528versión On-line ISSN 2075-5023
Ecología en Bolivia v.47 n.1 La Paz abr. 2012
Artículo
Subandean and adjacent lowland palm communities in Bolivia
Comunidades de palmas de bosques subandinos y de las zonas bajas adyacentes en Bolivia
Henrik Balslev1*, Zamir Pérez Durán2, Dennis Pedersen1, Wolf L. Eiserhardt1, Adriana Sanjinés Asturizaga1,3 & Narel Paniagua-Zambrana1,3
1 Department of Biosciences, Ecoinformatics and Biodiversity, Aarhus University, Build. 1540, Ny Munkegade 114, DK-8000 Aarhus C., Denmark *
Autor para correspondencia Email: henrik.balslev@biology.au.dk
2Museo de Historia Natural Noel Kempff Mercado, Herbario del Oriente Boliviano, Av. Irala 565, Casilla 2489, Santa Cruz, Bolivia
3Herbario Nacional de Bolivia, Universidad Mayor de San Andrés, Casilla 10077 - Correo Central, La Paz, Bolivia
Abstract
Palm community diversity and abundance was studied in the subandean zone and adjacent lowlands in 65 transects (5 x 500 m) covering 16.25 ha distributed over an area of 700 x 125 km in north-central Bolivia. A total of 38 palm species were found. The most diverse palm community was in lowland evergreen terra firme forest (28 palm species/4080 palm ind./ha, and seven growth forms), followed by the lower montane forest below 800 m elevation (19 palm species/ 2873 ind./ ha, and four growth forms) and lowland evergreen floodplain forests (17 palm species/3400 palms ind./ha, and five growth forms). The montane evergreen forests above 800 meters elevation (16 palm species/2583 palms ind./ha and three growth forms) and lowland seasonal evergreen and semideciduous forests (seven palm species/1207 palm ind./ha and five different growth forms) were the least diverse communities. Small palms were the most common growth form being represented by 18 (47%) species. Of the 38 palm species altogether, 21 (55%) were solitary, 15 (40%) were cespitose, and two (5%) were colonial. The abundant and dominant palm species, both in the canopy and in the understory, are the same as in other Amazonian forests and shared among several forest types in this study. The high species richness of the lowland evergreen terra firme forest is mostly due to a number of rare species that are lacking in other palm communities. The most abundant palm species were in general species with wide distributions shared among several palm communities suggesting that they are ecological generalists. The montane forests have their own set of palm species, such as Dictyocaryum lamarckianum, Euterpe precatoria var. longevaginata, and Geonoma undata, that separate them from other forest types.
Key words: Palm abundance, Arecaceae, Community Structure, Growth form, Species richness
Resumen
Se estudió la diversidad de comunidades de palmeras en la zona subandina y en tierras bajas adyacentes en 65 transectos (5 x 500 m), que abarcan 16.25 hectáreas de bosque distribuidas en un área de 700 x 125 km en el centro-norte de Bolivia. Un total de 38 especies de palmeras fueron encontradas en nuestros transectos. La comunidad de palmeras más diversa fue la del bosque de hoja perenne de terra firme (28 especies, 4.080 individuos por hectárea) seguida por los bosques premontanos < 800 m, (19 sp. y 2.873 ind./ha) y los bosques inundable de tierras bajas de hoja perenne (17 especies, 3.400 ind./ha). Los menos diversos fueron los bosques montanos por encima de 800 m (2.583 ind./ha en 16 especies) y los bosques caducifolios de tierras bajas, (1.207 ind./ha y sólo siete especies). De las 38 especies, 21 (55%) fueron solitarias, 15 (40%) fueron cespitosas y dos (5%) fueron coloniales. Las especies abundantes y dominantes, tanto en el dosel como en el sotobosque, son las mismas que en otros bosques de la Amazonia y aparecen en varias formaciones forestales en este estudio. La elevada riqueza de especies del bosque de hoja perenne de terra firme de tierras bajas se debe principalmente a un conjunto de especies raras que no aparecen en otras comunidades de palmeras. Las especies más abundantes fueron, en general, especies con distribuciones amplias y que son compartidas por varias de las comunidades de palmeras, lo que sugiere que son especies generalistas. Los bosques montanos tienen su propio conjunto de especies (tales como Dictyocaryum lamarckianum, Euterpe precatoria var. longevaginata y Geonoma undata) que los define, además de las especies comunes que comparten con otras formaciones forestales.
Palabras clave: Abundancia de palmas, Arecaceae, Estructura de comunidad, Forma de crecimiento, Riqueza de especies.
Introduction
Palms are emblematic for tropical forests throughout the Americas, Africa, and Southeast Asia. Everywhere palms are conspicuous elements of the vegetation, standing out due to their particular growth form with single woody trunks and terminal crowns of large palmate or pinnate leaves. Palms are also conspicuous in many open vegetation types such as savannas, where they may form large stands of single species, sometimes bordering rivers, or abounding where the ground water comes close to the surface. Palms are ecologically important as so-called keystone species that provide food for humans and animals during prolonged drought periods (Paine 1995, Garibaldi & Turner 2004), and they are ecologically important as structural elements of the tropical forests where they are not only part of the uppermost canopy but also fill in the mid- and under-storey forest; even the forest floor may be covered with small, shrubby palms (Kahn & de Granville 1992, Henderson 2002). Finally, palms may also form climbing growth forms, especially in Southeast Asia, where hundreds of palm species have long, thin stems and depend on surrounding vegetation for support (Henderson 2009). To humans living in tropical forests, palms are possibly the economically most important plant family. Palm leaves are used for thatching roofs, palm stems are used as poles for house construction, palms provide fibres for weaving fishing nets, hammocks, brooms and much more, and many palm fruits are edible, either for their soft mesocarp or for their oily endosperm (Paniagua-Zambrana et al. 2007, Balslev et al. 2008, Macía et al. 2011). To understand the structure, richness, and composition of palm communities is, therefore, of direct interest to socio-economic stakeholders and to the political decision makers who regulate the use and extraction of products from tropical forests (Millennium Ecosystem Assessment 2006, Balslev 2011).
The Bolivian palm flora is well known from a taxonomic point of view. It is well described in the Flora de Palmeras de Bolivia (Moraes 2004), which provides keys to the 28 genera and 80 palm species known to occur in Bolivia. The book also has morphological descriptions of each species and notes on their ecology, including maps of their distribution in Bolivia. Another rich source of information about Bolivian palms is Colecciones de las Palmeras de Bolivia (Moreno & Moreno 2006), which is a richly illustrated account of all Bolivian palms with many notes on their morphology, ecology, and usefulness. In contrast, there are only a few studies of Bolivian palm communities. In an inventory of 75 plots of 0.1 ha (total 7.5 ha) covering an altitudinal range of 150-5,700 m elevation in the Madidi region there were 24 palm species with dbh ≥ 2.5 cm, and they were found in overall densities of 320-470 ind./ha but only up to 3,000 m elevation (Paniagua-Zambrana 2005). A palm community at Río Hondo, also in the Madidi National Park, had nine palm species with dbh ≥ 10 cm in 9.6 ha and a density of 180 ind./ha (Cabrera & Wallace 2007). Some information can be extracted from ethnobotanical or other inventories that were not focussed on palms. In Iturralde province, an ethnobotanical survey included six palm species with dbh > 10 cm and the two 1-ha plots had 207 and 63 individual palms, respectively (De Walt et al. 1999).
To contribute to a more detailed knowledge of Bolivian palm communities we established 65 transects in subandean forests and adjacent lowland forests in central Bolivia. We were particularly interested in answering the following questions: 1) How are the palm communities composed? 2) How rich are the palm communities in species? 3) How abundant are palms in different kinds of forests and what is the relative abundance of each species? 4) Which palm growth forms are present and how are they represented in the different palm communities? 5) How are the architectural types (solitary, cespitose, colonial) represented in the palm communities? And 6) how are different leaf types represented in the palm communities?
Study area
Our study area covers ca. 700 x 125 km (Fig. 1) in the subandean zone and adjacent lowlands in north-central Bolivia, stretching from the border with Peru to the so-called Codo de los Andes lying at the latitude of the city of Santa Cruz. Politically our study area crosses the departments of Beni, La Paz, Cochabamba, and Santa Cruz. Geographically, our study area is located on the southwest rim of the Amazon basin, where the rivers Mamoré and Beni leave the Andes to flow into the mighty plain.
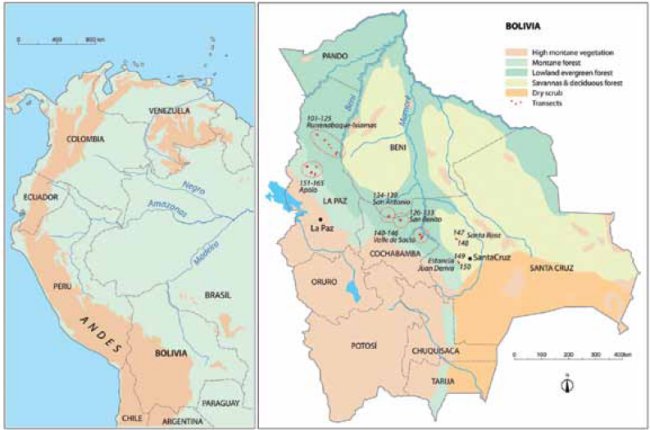 | |
| Figure 1. | Study area in the subandean zone and adjacent lowlands in north-central Bolivia superimposed on a simplified vegetation map (based on CIA vegetation map of Bolivia). The transects (red dots) were placed in seven areas indicated with dotted circles. Twenty-one transects were placed in lowland evergreen terra firme forests (104-106,110-115,122-125,134,137-140,142-143,146), 17 transects in lowland evergreen floodplain forests (126-133,135,141,143,145), 4 in lowland seasonal evergreen and semideciduous forests (147-150), 19 in lower montane forests (101-103,107-109,117-121), and 15 in montane forests (151-165). |
The climate in this area (www.climatediagrams.com) is rather wet to the north near the border with Peru, where the precipitation is close to 2,000 mm per year with a short dry season of 2-3 months in June-August during which the monthly precipitation is below 100 mm. In the north temperatures are rather stable at 24-26°C, with the warmest period coinciding with the wettest part of the year. To the south near the city of Santa Cruz the climate is dryer, with around 1,300 mm precipitation per year and with a 6-7 months dry season from April to August/September with less than 100 mm per month. Here the temperatures vary from 20-26°C, again with the warmest period coinciding with the wettest months. Minimum temperature in this area may be a little above freezing point in some winter days, restricting growth of many tropical palms. Along the Andean slope the temperature falls with elevation, usually 0.5°C per 100 meters elevation, so we encounter an average temperature of ca. 16°C at 2,000 m. The precipitation on the slopes varies tremendously depending on topography and winds.
Our study area borders with and partially overlaps the enormous Madidi National Park, which is known as one of the areas on the planet with the highest biodiversity (Jørgensen et al. 2005, Cornejo-Mejía et al. 2011) and is located within the great Andean biodiversity hotspot (Myers et al. 2000). The vegetation in the low lying parts of our study area covers a gradient from a species rich evergreen rainforest in the humid north to a less species rich seasonal evergreen and semideciduous forest in the seasonal south (Navarro & Ferreira 2004). Away from the rivers, where flooding never occurs, the upland forest is typical Amazonian terra firme, but on the alluvial plain along the rivers the forest is flooded every year in the wet season, which changes its structure to being more open and not as rich in species as the upland forest. Along the slopes of the Andes there are low ranges forming parallel low mountain chains that reach up to 800 m above sea level in the Andean foreland. These hills are covered with lower montane evergreen forest that is similar to adjacent lowland terra firme, but sometimes not as tall and also usually less species rich because the substrate is rocky and contains less water. Higher up on the slopes the forest changes to true montane forest with a low stature, a very different species composition, and often with many epiphytes covering the branches of trees. This forest reaches up to 3,000 m or more above sea level, but we only visited places up to ca. 2,000 m above sea level. According to this variation of the vegetation, we have divided our study sites into five categories: 1) lowland evergreen terra firme forest, 2) lowland evergreen floodplain forest, 3) lowland seasonal evergreen and semideciduous forest, 4) lower montane evergreen forest (<800 m), and 5) montane evergreen forest (800-2,000 m).
We accessed the forest at seven sites (Figure 1, Annex 2): 1) along the road between San Buenaventura and Ixiamas in Iturralde province in the department of La Paz, where we had access to lowland evergreen terra firme forest northeast of the road and to lower montane forests southwest of the road; 2) near Apolo in the department of La Paz, where we had access to several sites of montane forests at altitudes of 800-2,000 m; 3) near the village of San Antonio in the Beni department near the border with Cochabamba department, where we had access to both lowland evergreen terra firme and floodplain forest; 4) near the indigenous village of San Benito in the department of Cochabamba at the Isiboro river, where we had access to lowland evergreen floodplain forest; 5) along the Cochabamba-Santa Cruz highway near Valle de Sajta and the lateral road to Puerto Villarroel, where we had access to both lowland evergreen terra firme and floodplain forests; 6) near the village of Santa Rosa 90 km northwest of Santa Cruz, where we had access to lowland seasonal evergreen and semideciduous forest, and 7) in the Espejillos protected area 30 km west of Santa Cruz and Estancia Juan Deriva, where we had access to lowland seasonal evergreen and semideciduous forest.
Methods
We installed 65 transects measuring 5 x 500 m (0.25 ha) and collected our data according to a pre-established protocol (Balslev et al. 2010) that involves: 1) searching out a forest segment without or with only slight human impact; 2) marking a baseline 500 m long with stakes every five meters, and 3) counting and identifying all palm individuals including seedlings, juveniles, sub-adults and adults in 5 x 5 m subunits. Our protocol (Balslev et al. 2011) involves collecting additional ecological data, but that data is not used in this paper. We collected and photographed 172 herbarium vouchers to document species that were difficult to identify or otherwise interesting. These vouchers are deposited in the AAU, LPB, and UASC herbaria and can be seen with photographic images in the Aarhus University Herbarium database (www.aubot.dk, enter collector=Balslev; country=Bolivia; family=Arecaceae). In the field, the data were written on pre-printed data sheets and subsequently entered into a computerized database. Species occurrence from our data is available through the Global Biodiversity Information Facility portal (www.gbif.org).
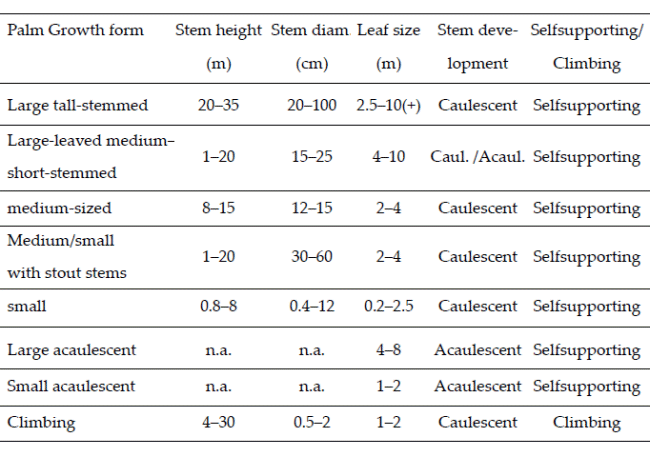
From our observations in the field and general knowledge of the palms, we divided all species into three architectural models (Dransfield et al. 2008) - cespitose, solitary, and colonial. Our category cespitose is in some other works called clustering, and our category colonial is sometimes called clustering by stolons or rhizomes. We classified all the species into growth forms in a system that considers overall stem size, leaf size, stem development (caulescent versus acaulescent), and whether the stem is self-supporting or climbing (Table 1, Balslev et al. 2011). Finally, we noted whether the leaves were pinnate or costapalmate - the only two leaf forms represented in our sample.
| Table 1. | Growth forms of American palms defined by the overall size of their stems and leaves, whether caulescent or acaulescent, and whether climbers or not (Balslev et al. 2011). |
 | |
One species was encountered with two varieties in our sample (Euterpe precatoria var. precatoria and var. longevaginata); for simplicity of descriptions and discussions, they are counted as if they were two species.
Results
Palm community composition
Lowland evergreen terra firme forest (Fig. 2)
 | |
| Figure 2. | Lowland evergreen terra firme forests and their palms. A-B. Under-storey and tall forest at San Antonio. C. Euterpe precatoria var. precatoria, Valle de Sajta. D. Geonoma macrostachys var. acaulis near San Benito. E. Oenocarpus bataua, Valle de Sajta. |
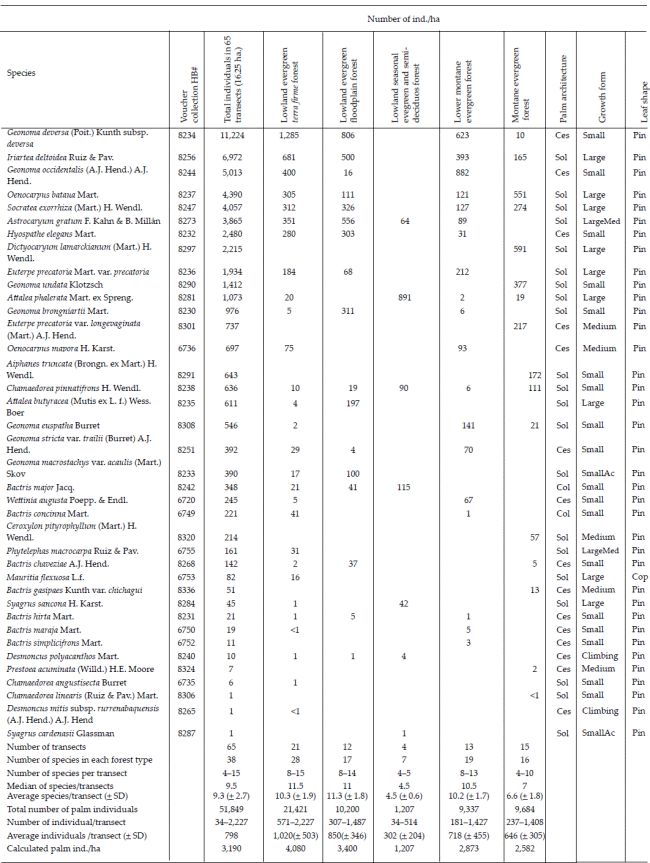
In the 21 transects (5.25 ha) located in lowland evergreen terra firme forest we encountered 28 species of palms and an average density of 4,080 palm ind./ha (Annex 1). Eight species were particularly abundant with over 200 ind./ ha; these included the small palms Geonoma deversa, G. occidentalis, and Hyospathe elegans, that are part of the understorey; a large-leaved medium- to short-stemmed palm, Astrocaryum gratum, which is part of the midstorey, and the large palms Iriartea deltoidea, Oenocarpus bataua, Socratea exorrhiza, and Euterpe precatoria var. precatoria, that form part of the canopy layer (Annex 1). All seven growth forms were represented, but the most common ones were small palms (14 species) and large palms (8 species), whereas the other growth forms were represented by only one or two species each. Of the 28 species 15 were solitary, 11 were cespitose, and two were colonial. Almost all of the species (27) had pinnate leaves; only Mauritia flexuosa had costapalmate leaves (Table 2). Mauritia flexuosa is a swamp palm and its occurrence in terra firme forest was in small patches of muddy soil.
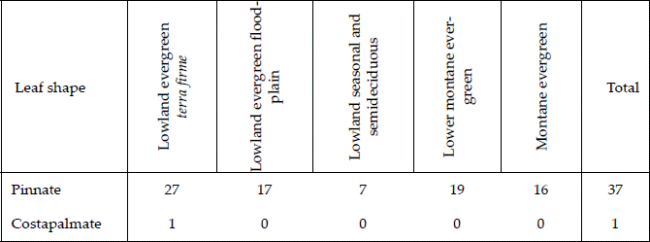
| Table 2. | Number of species with pinnate and costapalmate leaves in five forest types in the subandean zone and adjacent lowlands in Bolivia. Palm leaf shapes vary much, but in our sample only two leaf forms were present, one of them being the costapalmate leaf represented by only a single species Mauritia flexuosa. |
 | |
Lowland evergreen flood-plain forest (Fig. 3)
 | |
| Figure 3. | Lowland evergreen floodplain forests and their palms at San Benito. A. View of the under-storey with a large tree with buttresses and lianas. B. Inundated forest on the floodplain. C. Astrocaryum gratum, a mid-storey palm. D. Bactris major, a colonial under-storey palm. E. Geonoma deversa, the most abundant palm species in both floodplain and terra firme forests. |
This forest type was surveyed in 12 transects (3 ha) and we encountered 17 palm species with an average density of 3,400 palm ind./ ha The floodplain forest was dominated in the under-storey by the small palms Geonoma deversa, Hyospathe elegans, Geonoma brongniartii, and the acaulescent G. macrostachys var. acaulis; the mid-storey was dominated by Astrocaryum gratum, and the canopy by the large palms Iriartea deltoidea, Oenocarpus bataua, Socratea exorrhiza, and Attalea butyracea. The dominant floodplain species are to a large extent the same as in the terra firme, but G. occidentalis and Euterpe precatoria drop to very low densities and are replaced by G. brongniartii and Attalea butyracea (Annex 1). Of the seven growth forms only five were found in the floodplain forests and, as in the terra firme forest, small and large palms were the most common. Of the 17 species nine were solitary, seven were cespitose, and one was colonial. All the floodplain palm species had pinnate leaves (Table 2).
Lowland seasonal evergreen and semideciduous forest (Fig. 4)
 | |
| Figure 4. | Lowland seasonal evergreen and semideciduous forest at Reserva Estancia Juan Deriva. A-C. Gap, canopy, and understory. D. Syagrus sancona, common in the canopy of seasonal evergreen and semideciduous forests. E. Syagrus cardenasii, an endemic under-storey palm of Bolivian seasonal evergreen and semideciduous forests, Agua dulce, Monumento Natural Espejillos, F. Attalea phalerata, a widespread palm in seasonal evergreen and semideciduous forest. |
Located at the edge of the Amazon basin, in flat landscapes at 200-450 m elevation, this forest type had seven species and an average of 1207 ind./ha The four transects of 0.25 ha each in this forest type were established in places that do not suffer inundation. The seasonal evergreen and semideciduous forest understory included the small palms Bactris major, Chamaedorea pinnatifrons, and a single record of the small acaulescent Syagrus cardenasii. The seasonal evergreen and semideciduous forest mid-storey had substantial populations of Astrocaryum gratum, and the canopy had abundant Attalea phalerata and some Syagrus sancona. The abundance of Attalea phalerata is an indicator of the lowland seasonal and semideciduous forests. Finally, the climber Desmoncus polyacanthos had some rare occurrences (Annex 1). Although there were only seven palms species in the seasonal evergreen and semideciduous forest, they represented five different growth forms with the large and small palms being represented by two species each. There were five solitary species and one each of cespitose and colonial palms. All palm species in the seasonal evergreen and semideciduous forest had pinnate leaves (Table 2).
Lower montane evergreen forest (Fig. 5)
 | |
| Figure 5. | Lower montane evergreen forest. A. Panorama of the forest in Iturralde province along the San Buenaventura-Ixiamas road. B. Under-storey. C. River Beni near Rurrenabaque. D. Juvenile Socratea exorrhiza, E. Juvenile Iriartea deltoidea. F. Geonoma stricta var. traillii. |
The lower montane forests were studied in 13 transects (3.25 ha) located at elevations of 250-620 m above sea level on the low cordillera that runs parallel to the San Buenaventura-Ixiamas road. There we encountered 19 palm species and an average density of 2873 ind./ ha The understory was dominated by several small palm species including Geonoma deversa, G. occidentalis, and G. euspatha; the mid-storey had many individuals of Astrocaryum gratum and Oenocarpus mapora; and the canopy was dominated by Iriartea deltoidea, O. bataua, Socratea exorrhiza, and Euterpe precatoria var. precatoria. In addition, there were several less abundant species, particularly small palm species in the under-storey (Annex 1). Only four growthforms were represented among the lower montane forest palms; most were small (12 species) or large (five species), whereas large/medium and medium-sized palms were represented by one species each. The lower montane forest had a single colonial species, nine cespitose, and nine solitary species, respectively. All palms had pinnate leaves (Table 2).
Montane evergreen forest (Fig. 6)
 | |
| Figure 6. | Montane evergreen forests. A. View of forest near Apolo. B. Under-storey of montane evergreen forest near Apolo, C. Dictyocaryum lamarckianum, a large and abundant palm in the montane forest, Apolo. D. Geonoma undata, a small under-storey palm, Pucasucho, near Apolo. E. Ceroxylon pityrophyllum photographed in forest fragments at Pucasucho, near Apolo. F. Euterpe precatoria var. longevaginata, characteristic of montane forest, Apolo. |
The montane evergreen forest was studied in 15 transects (3.75 ha) in the hills around Apolo at elevations of 850-1,900 m above sea level. There we encountered 16 species and an average density of 2582 ind./ha. The understory was dominated by Geonoma undata, Chamaedorea pinnatifrons, and Aiphanes truncata; the mid-storey had large populations of Oenocarpus mapora and in some locations Ceroxylon pityrophyllum and Bactris gasipaes var. chichagui; whereas the most abundant canopy palms were Iriartea deltoidea, O. bataua, Socratea exorrhiza, Dictyocaryum lamarckianum, and in some places also Attalea phalerata. In addition to these, there were a number of rare, mostly small palm species (Annex 1). The palms in the montane forest belonged to three growth forms (large, medium, and small) that were almost equally represented. Most of the montane forest species were solitary (11 species), whereas five were cespitose and none were colonial. All palms in the montane forest had pinnate leaves (Table 2).
Growth forms
Of the eight growth forms described for tropical American palms (Table 1; Balslev et al. 2011), we encountered seven in our sample, large acaulescent palms being absent (Fig. 7). The growth form with most species was small palms, which included 18 (47%) of the 38 species encountered in our study. The second most common growth form was large palms, which included nine (24%) of the species, and the remaining 11 species belonged to the five remaining growth forms. The different forest types had different representation of the growth forms. Lowland evergreen terra firme forests had all seven growth forms represented among their species; floodplain and seasonal evergreen and semideciduous forest had five growth forms, and lower montane and montane forests had four and three growth forms, respectively (Fig. 7).
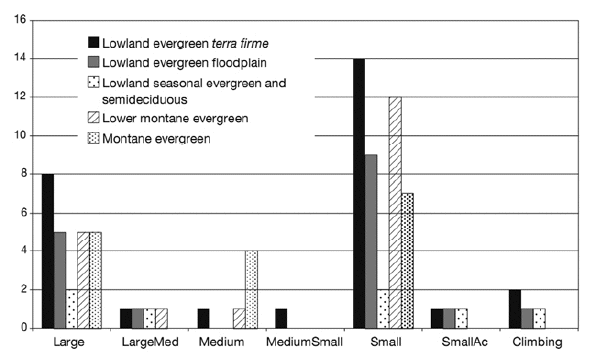 | |
| Figure 7. | Number of palm species in each growth form (see Table 1) in five forest types in the subandean and adjacent lowland forests in Bolivia. Of the eight growth forms defined for American palms (Balslev et al. 2011), only seven were encountered in our study area. |
Architecture
Of the three architectural types, solitary palms were represented by 21 species (55%), cespitose palms by 15 species (39%), and colonial palms by two species (5%). Colonial palms were absent in montane forests, but all three architectural types occurred in the remaining four forest types (Fig. 8).
 | |
| Figure 8. | The number of species of different palm architectural types in the five forest types surveyed in 65 transects (16.25 ha) in the subandean and adjacent lowland forests in Bolivia. |
Species richness
The total palm species richness was 38 species in 19 genera in the 65 transects (16.25 ha) distributed over our 700 x 125 km study area (Annex 1). The species rarefaction curves (Fig. 9) demonstrate that the lowland evergreen terra firme forest, with 28 species, is the richest among the five forest types and that the lowland seasonal evergreen and semideciduous forest with seven species is the poorest. At the local level we found a mean of 9.5 (range 4-15) palm species in transects (0.25 ha). The mean number of species varied from one forest type to another, lowland evergreen terra firme having the highest mean of 11.5 (8-15) species per transect, while in lowland seasonal evergreen and semideciduous forest the lowest mean was 4.5 (4-5) species per transect.
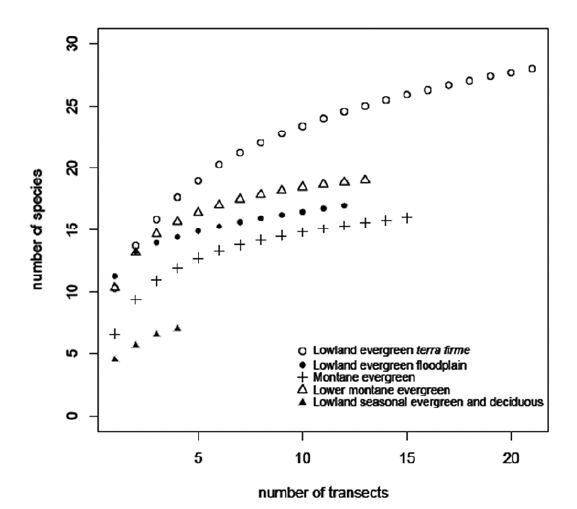 | |
| Figure 9. | Rarefaction curves showing the mean (expectation) of palm species richness if x transects randomly sampled from the total number of transects in each of the five forest types studied along the subandean and adjacent lowland forests in Bolivia. |
Species abundance
We counted 51,849 individual palms in the 65 transects (16.25 ha) giving an overall average density of 3190 ind./ha. The ranked abundances of the species (Annex 1, Fig. 10A) show that Geonoma deversa was superabundant in our sample, followed by a group of eight species (Iriartea deltoidea, Geonoma occidentalis, Oenocarpus bataua, Socratea exorrhiza, Astrocaryum gratum, Hyospathe elegans, Dictyocaryum lamarckianum, Euterpe precatoria) that all had >100 ind./ha on average, and finally the long tail of 29 species with <100 ind./ha including three species (Chamaedorea linearis, Desmoncus mitis var. rurrenabaquensis, Syagrus cardenasii) of which only one individual was encountered in the entire inventory. The most abundant species vary greatly in abundance between the five forest types (Fig. 10B).
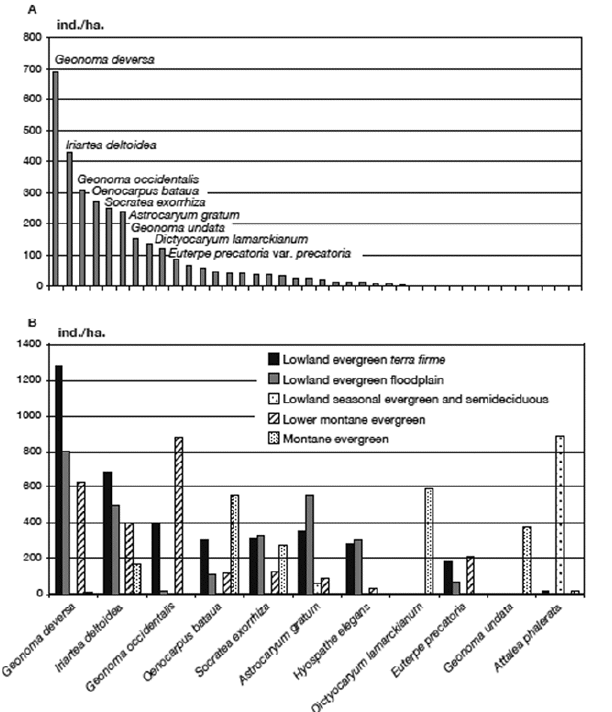 | |
| Figure 10. | A. Ranked abundance (ind./ha) of 38 palm species encountered in 65 transects (16.25 ha) in subandean and adjacent lowland forests in Bolivia. B. Abundances of the 11 most common palm species in the five forest types |
Leaf form
Of the 38 species encountered, all except one, Mauritia flexuosa, have pinnate leaves with costapalmate leaves being the only exception (Table 2).
Discussion
Palm community composition
Lowland evergreen terra firme forest
This forest type has the highest palm species richness in the study area, the highest density of palms, and most growth forms represented. In terms of local richness (species/transect), it is similar to floodplain and lower montane forests. The relatively high number of species in this forest type compared to the other forest types in our study area is largely due to a long tail of rare species with low abundances. This could suggest that the relatively high species richness could be an artefact of more intensive sampling of lowland terra firme forest compared to the other forest types in our study (21 transects versus 4, 12, 13, 15). A visual inspection of the rare-faction curve (Fig. 9), however, falsifies this hypothesis as there is a widening gap between the species numbers with increasing sampling effort. Compared to lowland evergreen terra firme forest elsewhere in tropical America, its diversity is within the range encountered in those forests (Balslev et al. 2011). When compared toother Amazonian terra firme forests (Kahn & Castro 1985, Vormisto et al. 2004, Cintra et al. 2005, Montufar & Pintaud 2006, Poulsen et al. 2006, Costa et al. 2009), these Bolivian evergreen terra firme forests are in the lower end of the range of species richness found elsewhere. In terms of composition, the dominant palm species are mostly the same as found elsewhere in the Amazon basin; exceptions to this being the under-storey palm Geonoma occidentalis, which has a south-western distribution in the Amazon basin (Henderson 2011) and Astrocaryum gratum, which belongs to a complex of allopatric species and, therefore, is lacking in most other parts of the Amazon basin (Kahn 2008). The dominant canopy palms include Iriartea deltoidea, Oenocarpus bataua, Socratea exorrhiza, and Euterpe precatoria, which belong to a group of common and widespread species appearing to underpin the theory of oligarchy in the western Amazon terra firme forests (Macía & Svenning 2005). Their high dominance in several different forest types investigated in this study agrees with the hypothesis that the most widespread Amazonian palms tend to be tall habitat generalists (Ruokolainen & Vormisto 2000).
Lowland evergreen flood plain forest
This forest type had fewer species than the terra firme forest, and although it was examined in fewer transects (12 vs. 21), the species rarefaction curve (Fig. 9) demonstrates that the floodplain forest is truly poorer in species. This pattern agrees well with the situation elsewhere in the Amazon basin, where floodplain forests in general are poorer in species than adjacent terra firme forests due to the fact that many species, including several palms, do not tolerate the annual flooding associated with that habitat (Balslev et al. 1987, Kahn & Mejía 1990). It is worth noting, however, that the average local species richness (species/transect) is slightly higher in the floodplain (11.3) than in terra firme (10.3), but the difference is not significant (P=0.083, Mann-Whitney test). The abundant species are the same in the two forest types and the high number of species in the terra firme habitat is due to several rare species that occur with low abundance there. The floodplain, in contrast, does not have this tail of rare species and actually no species was found in the floodplain forest that did not also occur in the terra firme forest, although some species, such as Geonoma brongniartii and Attalea butyracea, were much more abundant on the floodplains. In our sample, the floodplain forest had lower palm density (3,400 ind./ha) than the terra firme forest (4,080 ind./ha), but this seems not to be the rule since some floodplain forests in Bolivia have more individuals than adjacent upland forests (Paniagua-Zambrana 2005).
Lowland seasonal evergreen and semideciduous forest
Within our study area, the lowland seasonal evergreen and semideciduous forest is by far the poorest in terms of species richness having only seven of the 38 species we encountered. It also had much fewer palm ind./ha than the other forest types; it had less than half as many ind./ha (1207) as the second place montane forest (2,582 ind./ha), and less than one-third as many as the terra firme evergreen lowland forest (4,080 ind./ha). Our sampling inlowland seasonal evergreen and semideciduous forests was less intensive (only four transects), but the rarefaction curve (Fig. 9) suggests that species richness really is low compared to the other forest types sampled. The relative species abundances were quite different compared to the other forest types. One species (Syagrus cardenasii) was found only in the seasonal evergreen and semideciduous forest; four species (Attalea phalerata, Bactris major, Syagrus sancona, Desmoncus polyacanthos) were quite abundant in the seasonal evergreen and semideciduous forest but rare in other types, and the remaining two species (Astrocaryum gratum, Chamaedorea pinnatifrons) were more evenly distributed among the other forest types. This shows that the seasonal evergreen and semideciduous forest has a different species composition than the other forest types, especially when the abundance of the species is taken into account. Low species numbers and abundance of palms are also found in other tropical American seasonal evergreen and semideciduous forests. In the seasonal forest at Pinkaití Research Station in central Brazil (Para state), there are 10 species of palms with 167-800 adult ind./ha (Salm et al. 2007). On the Yucatan Peninsula, semideciduous and semi-evergreen seasonal forests with 1,100-1,300 mm annual precipitations have 3-4 palm species and 2,167-2,406 palm ind./ha, including all size-classes (Alvarado-Segura et al., unpublished data). In general, palm species richness is highest in aseasonal, wet, and warm areas (Bjorholm et al. 2005, Pintaud et al. 2008, Eiserhardt et al. 2011b). The reduction of palm species richness towards the dryer end and cooler areas is most likely a consequence of their structure, which is based on a single perennial bud with no reserve buds; if the apex is injured, the whole shoots dies (Richards 1996).
Lower montane evergreen forest
This forest type is very similar to the lowland evergreen terra firme forest in our study area (Annex 1). The two have similar species composition, abundance ranking of the species, and local diversity (10.2 palm species/transect versus the terr a firme forest 10.3 species/transect). The difference is that lower montane forests have lower palm abundance, i.e., 30% less ind./ ha and nine fewer species, mostly because several of the rare terra firme palms drop out on the low hills. On low hills along Río Ucayali in Peru, some 700 km to the northeast, we found a richer palm flora with 36 species (8 transects/ 2 ha) even though the palm abundance there (1,622 ind./ha) was relatively low (Balslev et al. 2010). In a lower montane forest in western Ecuador, there were 15 species in a 0.2 ha sample, with a very high density of 3,755 ind./ ha (Borchsenius 1997). The high variability among palm communities in lower montane forests suggests that even though they occupy a similar position in the landscape topography, these forests are not very uniform from one place to another and their palm communities are influenced by a variety of factors.
Montane evergreen forest
The species richness in the montane forests was low (16) overall and also locally (6.6± 1.8 sp./transect), although not as low as in the seasonal evergreen and semideciduous lowland forests. The montane forest share half of their species of palms with forest types at lower elevations (Geonoma deversa, Iriartea deltoidea, Oenocarpus bataua, Socratea exorrhiza, Attalea phalerata, Chamaedorea pinnatifrons, Geonoma euspatha, Bactris chaveziae), but the other half were only found at the high elevations (Dictyocaryum lamarckianum, Geonoma undata, Euterpe precatoria var. longevaginata, Aiphanes truncata, Ceroxylon pityrophyllum, Bactris gasipaes var. chichagui, Prestoea acuminata, Chamaedorea linearis), suggesting that they have a palm flora distinct from the palm floras of lowland forest types. The coherence of montane forest palm communities again is probably influenced by the peculiar structure of palms with large and single apical meristems that make them vulnerable to frost and other unfavourable conditions that may kill a whole plant by damaging only a single meristem. Some of the montane forest palms are apparently very well adapted to the habitat as witnessed by their high abundances. Dictyocaryum lamarckianum dominates the canopy and has an average of 591 ind./ha, while Geonoma undata dominates the under-storey and has as many as 377 ind./ha. It is also remarkable that the second most abundant palm in the montane forests, Oenocarpus bataua with 551 ind./ha, is also an abundant palm in the evergreen lowland forest types. In Panama, in an evergreen montane forest at 700-1,500 m elevation, there were 24 under-storey palm species in a 0.4 ha sample (Andersen et al. 2010); three of the species are shared with the Bolivian montane forests studied here. A Colombian evergreen montane forest in Risaralda located at 1,750-2,250 m above sea level had only 10 palm species in a 2.5-ha sample (Correa-Gómez & Vargas-Ríos 2009); nevertheless, four of the species are shared with our Bolivian sample. In south-eastern Ecuador there were 25 species of palms in 19 transects (4.75 ha) in an evergreen montane forest at 800-1,000 m (Byg et al. 2006), seven species of which are shared with the Bolivian sample presented here. As for the lower montane forest, the montane forest palm communities seem to vary considerably on a continental scale, although some species are widespread and turn up in samples taken as far apart as Panama, Colombia, Ecuador, and Bolivia. Our sample of 15 transects in montane evergreen forest was taken within a geographically limited area (max distance between transects 41 km versus 700 km for the entire study) which may have limited the species richness and variability encountered in this forest type.
Growth forms
The high diversity of small palms in all except the lowland seasonal evergreen and semideciduous forests is similar to palm communities throughout tropical America, which is also true for the second position large palms, and their proportions of the total species number (47% and 24%) fall well within the ranges reported elsewhere (Balslev et al. 2011), so this seems to be a general pattern which is corroborated by our results.
Architecture
The percentage of cespitose species in our sample (39%) is much below the average of 57% and even below the quartiles encountered in a series of tropical American palm communities (reviewed in Balslev etal. 2011). Between the five forest types studied here, there were similar but not equal proportions of cespitose versus solitary and colonial species (Figure 8). Although the cespitose life form could be seen as an adaptation to withstand unfavorable climatic conditions (cold, drought) because of the capacity to sprout from the base, that hypothesis is not supported by our data; instead, both seasonal evergreen and semideciduous and montane forests, where such adverse climatic conditions exist, have higher proportions of solitary palm species.
Species richness
In a continental context, the regional species richness of 38 species in 19 genera lies well within the range of 4-47 species encountered in 65 different inventories of tropical American palms, made in sample areas of 0.1-7.2 ha (reviewed by Balslev et al. 2011). In the context of the western Amazon basin, the species richness encountered here at the southern edge of the basin is on the lower side. For instance, the number of species registered here is substantially lower than the 56 species in 21 genera registered in Peru's upper Río Ucayali valley 700 km to the north, which was studied in a smaller area (275 x 60 km) and with only 35 transects (8.75 ha, Balslev et al. 2010). It is also much less than the 48 palm species found in 10 transects (2.5 ha) in the Iquitos-Pebas region in Peru (Vormisto et al. 2004) some 1,200 km to the north. The rarefaction curves (Fig. 9) suggest that more intensive studies in lowland evergreen terra firme forest and in lowland seasonal evergreen and semideciduous forest would reveal more species there than registered in the present study, but that the species numbers would still be lower than further north along the Andes in the western Amazon basin. This agrees with a general pattern of a latitudinal gradient with high palm species richness along the equatorial line and with decreasing richness further north and further south. This pattern has been demonstrated both with the use of transect derived data (Kristiansen et al. 2011) and range map derived data (Bjorholm et al. 2005). For local species richness the average of 9.3 species/transect in our 65 transects is lower than values found elsewhere in the western Amazon (Kristiansen et al. 2011). Overall, our results show that palm species richness in central Bolivia is in the lower end of what is found in the Amazon basin and in particular the western part of this region. Our data are derived from forests on the southern edge of the Amazon basin and a few seasonal evergreen and semideciduous forests bordering the Amazon basin that house forest types that are much less species rich, also when it comes to palms. Since the lower species richness is associated with lower precipitations and lower minimum temperature, this spatial pattern underpins climate as a very strong driver of the distribution of palm diversity, as has been shown for tropical American palms (Eiserhardt et al. 2011a).
Abundance
The abundances reported here include all individuals of palms, including seedlings, juveniles, sub-adults and adults. A separate analysis of abundances of different age classes in a future study may very well reveal interesting differences between the palm communities studied and the species they are made up of. The abundances of palms in Bolivian forests studied here fall well within the limits reported for palm communities throughout tropical America (reviewed in Balslev et al. 2011). Also here, as in almost all other communities, there is an uneven distribution of individuals among the species. The superabundant Geonoma deversa (Fig. 10A) had very high densities in the three forest types in the humid lowlands (terra firme, floodplain, lower montane), but disappeared with higher elevations (montane forests) and increasing seasonality (deciduous forests). This species is widely distributed in the Amazon basin (Henderson 2011) and its very high local abundance in some places has made it an attractive palm for thatch (Flores & Ashton 2000, Paniagua-Zambrana et al. 2007). The other abundant species (Fig. 10B) are mostly widespread in the Amazon basin (Henderson et al. 1995), but a few of them are abundant in more limited ecosystems. Geonoma occidentalis is distributed in the south-western Amazon basin (Henderson 2011), where it -as seen in this study- can be quite abundant. Morphologically, Geonoma deversa and G. occidentalis are very similar and they are closely related phylogenetically (Roncal et al. 2011), so it is interesting and maybe surprising that they co-occur in many transects. Dictyocaryum lamarckianum and Geonoma undata are also among the 10 most abundant species overall even if they are restricted to the montane forest, which reflects their highly dominant position there. Attalea phalerata occurs in four of the forest types investigated here and it is the most abundant species in the lowland seasonal evergreen and semideciduous forest where it is particularly abundant on humid soils in topographic depressions and in areas with a high freatic level, but only barely present in the other forest types. This species is distributed around the southern edge of the Amazon basin (Henderson 1995) and is obviously adapted to grow under seasonal and man made dry conditions even if it does enter less seasonal forest types in low numbers. In the other extreme, the ten least abundant species all have less than 50, and five of them have less than 10, ind./ha. These rare species are most characteristic of the lowland evergreen terra firme forest where six of them were found, but there are also rare species in the other forest types; two rare species were encountered in the floodplains, three in the lowland seasonal evergreen and semideciduous forest, three in the lower montane forest, and three in the montane forest.
Leaf form
The palm leaf has been considered the most complex determinate organ found in plants (Tomlinson 1979), and it is the distinguishing character of the family. Palm leaves are plicate and mostly dissected, they may be induplicate or reduplicate, pinnate, palmate, or costapalmate; the lamina, though mostly dissected, may in some cases be entire, and the pinnate lamina may in some cases be bi-pinnate (Dransfield et al. 2008). Considering this variation in palm leaf shapes, it is remarkable that almost all species in our study area have reduplicate pinnate leaves, the only exception being Mauritia flexuosa, which has reduplicate costapalmate leaves. Elsewhere in tropical America, induplicate palmate and costapalmate leaves are found in genera of the subfamily Coryphoideae. This subfamily occurs in South America (Sabal, Trithrinax, Cocoothrinax, Itaya, Chelyocarpus, Copernicia), but not in our study area. Reduplicate palmate and costapalmate leaves are characteristic of the subtribe Mauritiinae (Lepidocaryum, Mauritiella, Mauritia) of the subfamily Calamoideae, which is distributed in South America, but with only Mauritia represented in our study area. In Central America, where coryphoid palms are common, a much larger proportion of palm species (63%) and individuals are palmate (Balslev et al. 2011, Alvarado-Segura, unpublished data). This marked difference can be attributed to broad-scale biogeographic processes. The palmate-leaved coryphoid palms are thought to have reached tropical America through a boreotropic invasion during Paleocene/Eocene (Bjorholm et al. 2006).
Conclusions
The palm communities along the subandean zone and adjacent lowlands in Bolivia are rich in species and have high abundance of individual palms. The diversity of the communities falls well within the ranges reported for other tropical American palm communities, but they lie in the lower end of the diversity spectrum for the Amazon basin. Our data corroborate the latitudinal gradient in palm richness from the equatorial line towards the south with less rich communities further away from the equatorial line. Among the communities studied, the lowland evergreen terra firme forests were the richest and included a "tail" of rare species that was much less pronounced in the other forest types. The strong influence of climate on the diversity and richness of palm communities was corroborated within our study area, where species richness fell toward higher elevations with colder climates and towards drier and more seasonal areas with seasonal evergreen and semideciduous forests. Particularly the montane forests were qualitatively different with half of their species being restricted to them. Otherwise the dominant species in all communities were generally widespread and with broad ecological amplitudes.
Acknowledgements
We thank the European Commission Framework 7 programme for support to the PALMS project (contract no. 213126 to HB), and the Danish Natural Science Research Council for supporting our palm research (grant no 10-083348 to HB). Francis Kahn kindly reviewed and updated Table 1. We thank J.L. Luteyn for reading and commenting on the manuscript.
Literature
Andersen, K.M., B.L. Turner & J.W. Dalling. 2010. Soil-based habitat partitioning in understorey palms in lower montane tropical forests. Journal of Biogeography 37: 278-292.
Balslev, H. 2011. Palm harvest impacts in northwestern South America. The Botanical Review 77: 370-380.
Balslev, H., W. Eiserhardt, T. Kristiansen, D. Pedersen & C. Grández. 2010. Palms and palm communities in the upper Ucayali river valley - a little known region in the Amazon basin. Palms Journal of the International Palm Society 54(2): 57-72.
Balslev, H., C. Grández, N.Y. Paniagua-Zambrana, A.L. Møller & S. Lykke Hansen. 2008. Palmas (Arecaceae) útiles en los alrededores de Iquitos, Amazonía Peruana. Revista Peruana de Biología 15(1): 121-132.
Balslev, H., F. Kahn, B. Millan, J.-C., Svening., F. Borchsenius, T. Kristiansen, D. Pedersen & W. Eiserhardt. 2011. Species diversity and growth forms in tropical American palm communities. The Botanical Review 77: 381-425.
Balslev, H., J.L. Luteyn, B. Øllgaard & L.B. Holm-Nielsen. 1987. Composition and structure of adjacent unflooded and floodplain forest in Amazonian Ecuador. Opera Botanica 92: 37-57.
Bjorholm, S., J.-C. Svenning, W.J. Baker, F. Skov & H. Balslev. 2006. Historical legacies in the geographical diversity patterns of New World palm (Arecaceae) subfamilies. Botanical Journal of the Linnaean Society 151:113-125.
Bjorholm, S., J.-C. Svenning, F. Skov &H. Balslev. 2005. Environmental and spatial controls of palm (Arecaceae) species richness across the Americas. Global Ecology and Biogeography 14: 423-429.
Borchsenius, F. 1997. Palm communities in western Ecuador. Principes 41: 93-99.
Byg, A., J. Vormisto & H. Balslev. 2006. Using the useful: characteristics of used palms in south-eastern Ecuador. Environment, Development and Sustainability 8: 495-506.
Cabrera, W.H. & R. Wallace. 2007. Densidad y distribución de palmas arborescentes en un bosque preandino - amazónico de Bolivia. Ecología en Bolivia 42(2): 121-135.
Cintra, R., A. De Carvalho Ximenes, F.R. Gondim & M.S. Kropf. 2005. Forest spatial heterogeneity and palm richness, abundance and community composition in terra firme forest, central Amazon. Revista Brasileira de Botanica 28: 75-84.
Correa-Gomez, D.F. & O. Vargas-Ríos. 2009. Regeneration of palms in native forests and plantations at Otun-Quimbaya Fauna and Flora Sanctuary (Risaralda, Colombia). Caldasia 31:195-212.
Cornejo-Mejía, M., P.M. Jørgensen, M.J. Macía, I. Loza, A. Fuentes & L. Cayola (eds.). 2011. Memorias de los 10 años de investigación botánica realizada en la Región Madidi: "conociendo una de las regiones más biodiversas del mundo". Herbario Nacional de Bolivia, Missouri Botanical Garden,. La Paz. 30pp. [ Links ]
Costa, F.R.C., J.L. Guillaumet, A.P. Lima & O.S. Pereira. 2009. Gradients within gradients: The mesoscale distribution patterns of palms in a central Amazonian forest. Journal of Vegetation Science 20: 69-78.
De Walt, S., G. Bourdy, L.R. Chávez de Michel & C. Quenevo. 1999. Ethnobotany of the Tacana: Quantitative inventories of two permanent plots of northwestern Bolivia. Economic Botany 53: 237-260.
Dransfield, J., N.W. Uhl, C.B. Asmussen, W.J. Baker, M.M. Harley & C.E. Lewis. 2008. Genera palmarum: The evolution and classification of palms. Kew Publishing, London. 732 pp [ Links ]
Eiserhardt, W.L., S. Bjorholm, J.-C. Svenning, T.F. Rangel & H. Balslev. 2011a. Testing the water-energy theory on American palms (Arecaceae) using geographically weighted regression. PLoS ONE 6(11): e27027.
Eiserhardt, W.L., J.-C. Svenning, W.D. Kissling & H. Balslev. 2011b. Geographical ecology of palms (Arecaceae): determinants of diversity and distributions across spatial scales. Annals of Botany 108:1391-1416.
Flores, C.F. & P.M. Ashton. 2000. Harvesting impact and economic value of Geonoma deversa, Arecaceae, an understory palm used for roof thatching in the Peruvian Amazon. Economic Botany 54:267-277.
Garibaldi, A. & N. Turner. 2004. Cultural keystone species: Implications for ecological conservation and restoration. Ecology and Society 9(3): 1 (online) URL: http://www.ecologyandsociety.org/vol9/iss3/art1
Henderson, A. 1995. Palms of the Amazon. Oxford University Press, Oxford. 362 pp [ Links ]
Henderson, A. 2002. Evolution and ecology of palms. The New York Botanical Garden Press, Bonx. 259 pp.
Henderson, A. 2009. Palms of southern Asia. Princeton University Press, New Jersey. 199 pp.
Henderson, A. 2011. A revision of Geonoma (Arecaceae). Phytotaxa 17:1-271.
Henderson, A., G. Galeano & R. Bernal. 1995. Field guide to the palms of the Americas. Princeton University Press, New Jersey. 353 pp.
Jørgensen, P.M., M.J. Macía, A. Fuentes, S.G. Beck, M. Kessler, N. Paniagua, R. Seidel, C. Maldonado, A. Araujo-Murakami, L. Cayola, T. Consiglio, T.J. Killeen, W.H. Cabrera, F. Bascopé, D. De la Quintana, T. Miranda, F. Canqui & V. Cardona-Peña. 2005. Lista anotada de las plantas vasculares registradas en la región de Madidi. Ecología en Bolivia 40(3): 70-169.
Kahn, F. 2008. The genus Astrocaryum. Revista Peruana de Biologia 15(supl. 1): 31-48.
Kahn, F. & A. de Castro. 1985. The palm community in a forest of central Amazonia, Brazil. Biotropica 17:210-216.
Kahn, F. & J.-J. de Granville. 1992. Palms in Forest Ecosystems of Amazonia. Ecological Studies 95. Springer-Verlag, Berlin, Heidelberg, New York.
Kahn, F. & K. Mejía. 1990. Palm communities in wetland forest ecosystems in Peruvian Amazonia. Forest Ecology and Management 33/44:169-179.
Kristiansen, T., J.-C. Svenning, D. Pedersen, W.L. Eiserhardt, C. Grández & H. Balslev. 2011. Local and regional palm (Arecaceae) species richness patterns and their cross-scale determinants in the western Amazon. Journal of Ecology 99: 1001-1015.
Macía, M.J., P.J. Armesilla, R. Cámara-Leret, N. Paniagua-Zambrana, S. Villalba, H. Balslev & M. Pardo-de-Santayana. 2011 Palm uses in northwestern South America: A quantitative review. The Botanical Review 77:462-570.
Macía, M.J. & J.-C. Svenning. 2005. Oligarchic dominance in western Amazonian plant communities. Journal of Tropical Ecology 21: 613-626.
Millennium Ecosystem Assessment. 2006. Ecosystems and Human Well-being. Our Human Planet: Summary for Decision Makers. Island Press, Washington. 137 pp. [ Links ]
Montufar, R. & J.C. Pintaud. 2006. Variation in species composition, abundance and microhabitat preferences among western Amazonian terra firme palm communities. Botanical Journal of the Linnean Society 151:127-140.
Moraes Ramírez, M. 2004. Flora de palmeras de Bolivia. Herbario Nacional de Bolivia, Instituto de Ecología, Carrera de Biología, Universidad Mayor de San Andrés, Plural Editores, La Paz. 262 pp
Moreno Suárez, L.R. & O.I. Moreno Suarez. 2006. Colecciones de las palmas de Bolivia. Editorial FAN, Santa Cruz. 575 pp. [ Links ]
Myers, N., R.A. Mittermeier, C.G. Mittermeier, G.A.B. de Fonseca & J. Kent. 2000. Biodiversity hotspots for conservation priorities. Nature 403: 853-858.
Navarro, G. & W. Ferreira. 2004. Zonas de vegetación potencial de Bolivia: Una base para el análisis de vacíos de conservación. Revista Boliviana de Ecología y Conservación Ambiental 15: 1-40.
Paine, R.T. 1995. A conversation on refining the concept of keystone species. Conservation Biology 9(4): 962-964.
Paniagua-Zambrana, N.Y. 2005. Diversidad, densidad, distribución y uso de las palmas en la región del Madidi, noreste del departamento de La Paz (Bolivia). Ecología en Bolivia 40(3): 265-280.
Paniagua-Zambrana, N.Y., A. Byg, J.-C. Svenning, M. Moraes, C. Grández & H. Balslev. 2007. Diversity of palm uses in the western Amazon. Biodiversity and Conservation 16: 2771-2787.
Pintaud, J.-C., G. Galeano, H. Balslev, R. Bernal, F. Borchsenius, E. Ferreira, J.-J. de Granville, K. Mejía, B. Millán, M. Moraes, L. Noblick, F.W. Stauffer & F. Kahn. 2008. Las palmeras de América del Sur: diversidad, distribución e historia evolutiva. Revista Peruana de Biología 15(supl. 1): 7-29
Poulsen, A.D., H. Tuomisto & H. Balslev. 2006. Edaphic and floristic variation within a 1-ha plot of lowland Amazonian rain forest. Biotropica 38: 468-478.
Richards, P.W. 1996. The tropical rain forest. Cambridge University Press, Cambridge. 575 pp.
Roncal, J., A. Blach-Overgaard, F. Borchsenius, H. Balslev & J.-C. Svenning. 2011. A dated phylogeny complements macroecological analysis to explain diversity patterns in Geonoma(Arecaceae). Biotropica 43(3): 324-334.
Ruokolainen, K. & J. Vormisto. 2000. The most widespread Amazonian palms tend to be tall and habitat generalists. Basic and Applied Ecology 1: 97-108.
Salm, R., N. V. de Salles, W.J. Alonso & C. Schuck-Paim. 2007. Cross-scale determinants of palm species distributions. Acta Amazonica 37(1): 17-26.
Tomlinson, P.B. 1979. Systematics and ecology of the Palmae. Annual Review of E cology and Systematics 10: 85-107.
Vormisto, J., J.-C. Svenning, P. Hall &H. Balslev. 2004. Diversity and dominance in palm (Arecaceae) communities in terra firme forests in the western Amazon basin. Journal of Ecology 92: 577-588.
Artículo recibido en: Enero de 2012.
Manejado por: Ramiro López
Aceptado en: Marzo de 2012.
| Annex 2. | Locality information for 65 transects in the Andean foothills and adjacent lowlands in central Bolivia used to study palm communities. |
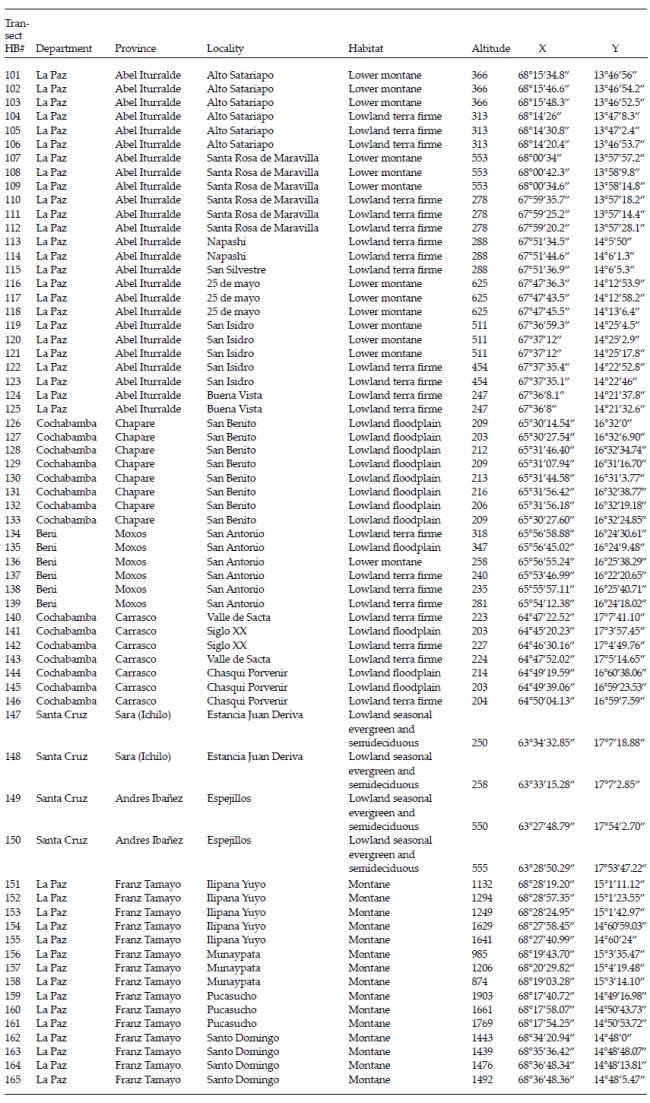 | |












 uBio
uBio