Ecología en Bolivia
versión impresa ISSN 1605-2528versión On-line ISSN 2075-5023
Ecología en Bolivia v.43 n.1 La Paz 2008
An albino Gray-bellied Flowerpiercer (Diglossa carbonaria)
in the high Andes of Bolivia
Una diglosa carbonosa albina (Diglossa carbonaria)
en los altos Andes de Bolivia
Jennifer R. , A. Cahill
Centro de Biodiversidad y Genética, Universidad Mayor de San simón, Casilla 538, Cochabamba,
Bolivia, jcahill@fcyt.umss.edu.bo
Evolutionary Ecology Group, Department of Biology, University of Antwerp, B-2610 Antwerp, Belgium
In February 2003 I observed a bird that appeared to be a completely white flowerpiercer (Diglossa sp.) foraging on the shrub Brachiotum microdon (Melastomataceae) along the edge of a 34.9-ha Polylepis forest fragment in Sacha Loma, Cochabamba department (Bolivia). The bird moved normally within the fragment, mostly through the central part containing a 10-ha boulder field. When flying across this area it became camouflaged against the background of grey boulders. I caught the albino, after 4 hrs of effort, with a 12-m mist net placed transversally in the boulder field.
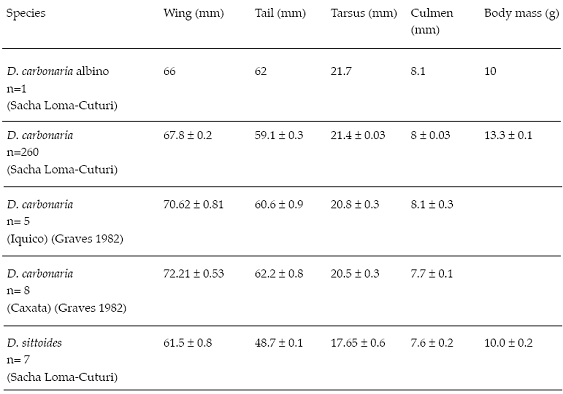
On the basis of culmen morphology (mainly the hooked maxilla) it was apparent that the bird was a Diglossa flowerpiercer. However, its albinism condition was total (complete absence of melanin) according to albino reports in the literature (Sage 1962, Dyck 1985, Møller & Mousseau 2001). The skin and feathers lacked pigmentation, the eyes were pinkish red and the culmen was pink, probably presenting this color just from the blood in capillaries as it lacked pigmentation. The legs and feet also were pinkish with no pigmentation. Analyzing the birds plumage attributes (Ralph et al. 1993), no signs of juvenal plumage were found. A skull inspection indicated complete ossification, and I concluded that it was an adult. Morphological measurements are given in Table 1.
Over the course of five years as part of a ringing program (following procedures of Ralph et al. 1993) to study forest specialists in 12 Polylepis forest fragments (2 – 47 ha) (3,500 – 3,900 m altitude) in Sacha Loma (17º 44´ S, 65º 34´ W) and Cuturi (17 º 42´ S, 65º 33´ W) near the city of Cochabamba, we captured 260 Gray-bellied Flowerpiercers (D. carbonaria), the morphometric data of which are given in Table 1. A previous study (Graves 1982) of the same species in Iquico and Caxata in Bolivias depto. La Paz presented similar results (Table 1). Measurements of the albino flowerpiercer were very similar to those of the Gray-bellied Flowerpiercer (Table 1) and fell well inside the range of values for wing and tarsus length of that species (Fig. 1). The only exception was body mass, which was about 3 g lighter than the average for normally pigmented individuals of the Gray-bellied Flowerpiercer (Table 1). Only 10% of this species captured individuals had a body mass of 10–11 g.
Only one other Diglossa species was captured with a very low frequency in Sacha Loma and Cuturi over the 5-year period. Seven individuals of the Rusty Flowerpiercer (D. sittoides) captured exclusively during the rainy season (Nov–Mar) had considerably smaller (and mostly nonoverlapping) measurements than the Gray-bellied Flowerpiercer and the albino captured in Sacha Loma (Table 1, Fig. 1). Given those notably smaller measurements of the Rusty Flowerpiercer and, based on capture rates, the higher probability of the albino being a Gray-bellied Flowerpiercer (97%
Table 1: Selected measurements of a complete albino Gray-bellied Flowerpiercer (D. carbonaria) from Sacha-Loma-Cuturi and normally pigmented from three localities in the Bolivian Andes. Values are means ± SE.
of all flowerpiercer captures), I concluded that the albino was assignable to the latter species. Furthermore, the assignation of the albino to other species of flowerpiercers present above 3000 m in Cochabamba (D. mystacalis and D. cyanea) also was discarded as these species of humid Yungas forest were never caught or reported in the Sacha Loma – Cuturi area.
Partial or incomplete albinism has been observed in many species of birds (Gross 1965), including a Giant Conebill (Oreomanes fraseri) in Polylepis forest in Peru (Servat 2002). However, total or true albinos (Bruckner 1941) such as those registered in Sharp-tailed Sparrow (Passerherbulus caudacutus) (Wayne 1921), Mallard (Anas platyrhynchos) (Sprunt 1931),
Red-winged Blackbird (Agelaius phoeniceus) (Nero 1954), Northern Bobwhite (Colinus virginianus) (Menzel 1962) and Herring gull (Larus argentatus) (Gross 1964) are rare in wild birds (7% of 1847 cases of some type of albinism according to Gross 1965). There are apparently no previous records of total or partial albinism for the Gray-bellied Flowerpiercer or other species in the genus Diglossa, although population and speciation studies have examined the plumage colors of many Diglossa museum specimens (e.g., Graves 1982, Vuilleumier 1984). In fact, reports of cases of albinism in Neotropical birds seem very scarce (Hosner & Lebbin 2006). According to van Grouw (2006), the reason that adult albinos
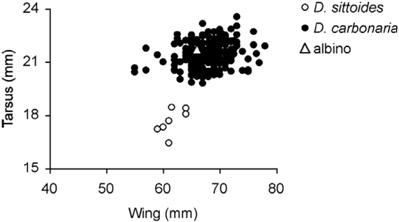
Fig. 1: Wing versus tarsus length of one complete albino Gray-bellied Flowerpiercers (Diglossa carbonaria); and seven Rusty Flowerpiercers (D. sittoides) captured in Polylepis forest in Sacha Loma-Cuturi.
are so rarely observed in the wild is that they have bad eyesight, imperfect flight and are therefore more susceptible to predation early on in life. Because partial albinism may be due to germline mutations (Møler & Mousseau 2001), diet, senility, shock, disease or injury (Sage 1962), and because complete albinism is most likely a result of inherited genetic factors which condition lack of tyrosinase and, consequently, melanin (Bruckner 1941, Sage 1962, Bensch et al. 2000), it is most probable that the albino flowerpiercer in Sacha Loma was homozygous for this condition. With 76% of Gray-bellied Flowerpiercer recaptures in different months and years recorded in the same fragments, it is tempting to propose a link between this unusual plumage condition and inbreeding as a consequence of small isolated Polylepis fragments and low dispersal.
It is evident that in very isolated populations a recessive gene may gain a much higher frequency of expression than in a population where gene flow is maintained over a large area (Sage 1962, Gross 1965). From the nectarivorous feeding habits of the Gray-bellied Flowerpiercer(Ridgely & Tudor 1989, Fjeldså & Krabbe 1990) it could be assumed that the species presents long distance dispersal in response to local and regional flowering seasons. However, Herzog et al. (2003) indicated that it is a year-round resident in Sacha Loma Polylepis forest fragments, probably shifting to a more insectivorous diet during the dry season when flower nectar is scarce. Naoki (1998) further suggested that morphological and behavioral adaptations of flowerpiercers allow them to use almost any flowers and to be resident on the same territory throughout the year. Therefore, seasonal shifts in feeding habits combined with restricted movements that are largely confined to isolated and patchily distributed Polylepis fragments could bring about reduced gene flow and increased homozygosity in the population.
Furthermore, the Gray-bellied Flowerpiercer is a restricted-range species (Stattersfield et al. 1998, Hennessey et al. 2003) not widely distributed in the high Andes. It is probably endemic to the Andes of Bolivia (Ridgely & Tudor 1989, Fjeldså & Krabbe 1990, Stotz et al. 1996), although it also may occur in northwest
Argentina (Moschione & San Cristobal 1993). Such a small geographic distribution could also be explained by a tendency for short distance dispersal. In a study on the zoogeography of Andean birds Vuilleumier (1984) concluded that the Gray-bellied Flowerpiercer presents partially continuous and partially disjunct populations in Bolivia.
This anecdotal record calls attention to the implications of fragmentation and isolation effects on birds dependent on Polylepis forests in the high Andes.
Acknowledgments
The albino was found during a bird ringing field trip, funded by the Flemish Interuniversity Cooperation (VLIR-IUC, Belgium) and the Centro de Biodiversidad y Genética, Universidad Mayor de San Simon. I thank J.V. Remsen and S.K. Herzog for comments on the manuscript.
References
1.- Bensch, S., Hansson, B., Hasselquist, D. & B. Nielsen. 2000. Partial albinism in a semi-isolated population of Great Reed Warblers. Hereditas 133: 167-170. [ Links ]
2.- Bruckner, J. H. 1941. Inheritance of white plumage in Phasianus. Auk 58: 536-542. [ Links ]
3.- Dyck, J. 1985. Plumage abnormal. Pp. 472 – 474 in Campbell, B. & E. Lack (eds. ). A Dictionary of Birds. Poyser, Calton. [ Links ]
4.- Fjeldså, J. & N. Krabbe. 1990. Birds of the high Andes. Apollo Books, Zoological Museum, University of Copenhagen & Svendborg, Copenhagen. 876 p. [ Links ]
5.- Graves, G. R. 1982. Speciation in the Carbonated Flower-piercer (Diglossa carbonaria) complex of the Andes. Condor: 84: 1-14. [ Links ]
6.- Gross, A. O. 1964. Albinism in the Herring Gull. Auk 81: 551-552. [ Links ]
7.- Gross, A. O. 1965. The incidence of albinism in North American birds. Bird Banding 36: 67-71. [ Links ]
8.- van Grouw, H. 2006. Not every white bird is an albino: sense and nonsense about colour aberrations in birds. Dutch Birding 28: 79-89. [ Links ]
9.- Hennessey, A.B., S.K. Herzog & F. Sagot. 2003. Lista anotada de las aves de Bolivia. Asociación Armonía/BirdLife International, Santa Cruz de la Sierra, Bolivia. [ Links ]
10.- Herzog, S. K., Soria, R. & E. Matthysen. 2003. Seasonal variation in avian community composition in a High- Andean Polylepis (Rosaceae) forest fragment. Wilson Bulletin 115: 438-447. [ Links ]
11.- Hosner, P. A. & D. J. Lebbin. 2006. Observations of plumage pigment aberrations of birds in Ecuador, including Ramphastidae. Boletín SAO 16: 01. [ Links ]
12.- Menzel K. E. 1962. Total albinism in a Nebraska Bobwhite. The Wilson Bulletin, 74: 99. [ Links ]
13.- Møller, A. P. & T. A. Mousseau. 2001. Albinism and phenotype of barn swallows (Hirundo rustica) from Chernobyl. Evolution 55: 2097-2104. [ Links ]
14.- Moschione, F. N. & J. San Cristobal. 1993. Registro del Payador Negro Diglossa carbonaria para la Argentina. Hornero 13: 307. [ Links ]
15.- Naoki, K. 1998. Seasonal change of fower use by the Slaty Flowerpiercer (Diglossa plumbea). The Wilson Bulletin, 110: 393- 397. [ Links ]
16.- Ralph, C. J., Geupel, G. R., Pyle, P., Martin, T. E. & D. F. DeSante. 1993. Handbook of feld methods for monitoring landbirds. Gen. Tech. Rep. PSW-GTR-144. Albany, CA: Pacifc Southwest Research Station, Forest Service, U.S. Department of Agriculture, USA. [ Links ]
17.- Ridgely R. S. & G. Tudor. 1989. The birds of South America, Volume I. The Oscine Passerines. Oxford University Press, Oxford, UK, 516 pp. [ Links ]
18.- Sage, B. 1962. Albinism and melanism in birds. British Birds 55: 201-225.
18.- Servat, G. P. 2002. A case of partial albinism in Giant Conebill, Oreomanes fraseri. Cotinga 18: 80.
19.- Stattersfeld, A.J., M.J. Crosby, A.J. Long & D.C. Wege. 1998. Endemic bird areas of the world: priorities for bird conservation. BirdLife International, Cambridge. [ Links ]
20.- Stotz, D. F, J. W. Fitzpatrick, T. Parker III, & D. K. Moskovits. 1996. Neotropical birds. The University of Chicago Press. Chicago, USA. Sprunt J. R. A. 1931. Total albinism in the Mallard
(Anas plathyrhynchos).Auk 48: 414.
21.- Vuilleumier, F. 1984. Zoogeography of Andean Birds: Two major barriers; and speciation and taxonomy of the Diglossa carbonaria superspecies. National Geographic Society Research Reports 16: 713-731. [ Links ]
Nota recibida en: Octubre de 2007
Manejada por: Sebastian Herzog
Aceptada en: Febrero de 2008












 uBio
uBio 

