Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim vol.34 no.5 La Paz 2017
LOS COMENTARIOS Y / O ARTÍCULOS TEÓRICOS
Mechanistic theoretical proposals for: Alkenes by
claisen rearrangement of α allylthio carbenes;
Aza-cope rearrangement of 4- butenyliminium ions; 2-
substituted pyrrolidine derivatives; Synthesis of
perhydrogephyrotoxin, key step; Part v; The organic chemistry notebook, nº 13
Propuestas mecanicistas teóricas para alquenos por reordenamiento de claisen de
carbenos α-aliltio;
Reordenamiento aza-cope de iones 4-buteniliminio; Derivados pirrolidínicos substituidos en 2; Síntesis de perhidrogepirotoxina, paso clave; Parte v; el cuaderno de quimica organica, nº 13
José A. Bravo1,*, José L. Vila2
*Corresponding author: jabravo@umsa.bo
ABSTRACT
This is the thirteenth theoretical assay in the series: “The Organic Chemistry Notebook Series, a Didactical Approach”.
The aim of this series of studies is to help students to have a graphical view of organic synthesis reactions of diverse nature. We have taken a series of reactions compiled by W. Carruthers in ‘Some modern methods of organic synthesis’, and we have proposed didactical and mechanistic views for them. This theme is included in the chapter
“Formation of carbon-carbon double bonds” in the mentioned text.
-sulphured alkenescan be obtained by Claisen rearrangement of -allylthio carbenes. A theoretical reaction pathway is proposed here. A short mechanism is proposed for the aza-Cope rearrangement from 4-butenyliminium ions. From this, 2-substituted pyrrolidine derivatives are explained in their reaction step by step under a theoretical approach. With hydroxyl and amino groups as substituents, bicyclic pyrrolidine derivatives are reached. The synthesis of perhydrogephyrotoxin, a natural product, is explained mechanistically here by the using of the preceding reactions.
Keywords: Organic Chemistry, Alkenes, -Allylthio carbenes, 4-butenyliminium ions, 2- Substituted pyrrolidine derivatives, Perhydrogephyrotoxin, Claisen rearrangement, Aza-Cope rearrangement, Mechanisms of Reactions, W. Carruthers.
RESUMEN
Este es el décimo tercer ensayo teórico en la serie: “El cuaderno de química orgánica, un enfoque didáctico”.
El objetivo de esta serie de estudios es ayudar a los estudiantes a disponer de una visión gráfica de reacciones de síntesis orgánicas de diversa naturaleza. Hemos tomado una serie de reacciones compiladas por W. Carruthers en: ‘Some modern methods of organic synthesis’, para las cuales hemos propuesto vistas mecanicistas y didácticas. Este tema está incluido en el capítulo “Formation of carbon-carbon double bonds” del mencionado texto.
Los alquenos -sulfurados pueden obtenerse por reordenamiento de Claisen de carbaniones -aliltio o carbenos α aliltio. Aquí se propone una ruta de reacción teórica para el reordenamiento de carbenos α aliltio. Se propone un mecanismo breve para la transposición de aza-Cope a partir de iones 4-buteniliminio. De acá, los derivados de pirrolidina 2-sustituidos se explican en su reacción paso a paso bajo un enfoque teórico. Con grupos hidroxilo y amino como sustituyentes, se alcanzan derivados de pirrolidina bicíclicos. La síntesis de la perhidrogefirotoxina, un producto natural, se explica con enfoque mecánicista aquí mediante el uso de las reacciones precedentes.
INTRODUCTION
Master classes of organic chemistry showed us the difficulties experimented by students due to lack of knowledge of classical mechanisms. A mechanistic proposal is naturally mandatory if a rational explanation of products emerging from a synthesis is going to be formally accepted and understood. As academics we are committed with the didactics and we have designed a series of articles exposing mechanistic theoretical proposals, articles have a character of review, meaning thus the use of published works on varied themes on synthesis. The present contribution: Mechanistic theoretical proposals for: alkenes by Claisen rearrangement of -allylthio carbenes; aza-cope rearrangement of 4-butenyliminium ions; 2-substituted pyrrolidine derivatives; synthesis of perhydrogephyrotoxin, key step; part V; the organic chemistry notebook, Nº 13, is the thirteenth study in the series: “The Organic Chemistry Notebook Series, a Didactical Approach” [1-12].
REVIEW OF REACTIONS, MECHANISTIC THEORETICAL PROPOSALS, DISCUSSION
α allylthio carbenes [13]
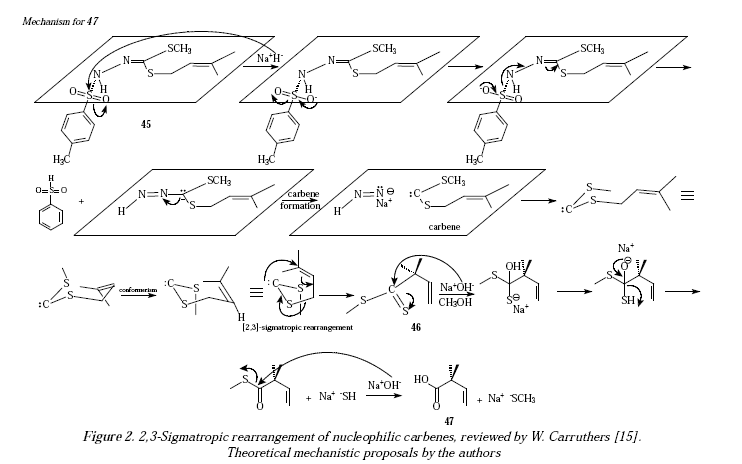
The rearrangement of -allylthio carbanions and -allylthio carbenes is feasible. For example, Β;y,-ene-carboxylic acids can be obtained from -allylthio carbenes, as shown schematically in Figure 1. The presence of six electrons to achieve the electrocyclic reaction is mandatory [13-15]. Figure 2 shows a theoretical mechanistic proposal for the reaction of Figure 1.
In Figure 2 the strong base H- provided by NaH performs a nucleophilic attack over sulphur in the tosylate moiety of compound 45. After resonance of the N=N bond finally situating the charge excess over carbon. Substrate is now free of tosylate, the protective group, and the breaking of the N=N-C bond generates the expected carbene. Carbene is responsible then of the Claisen rearrangement once the appropriate conformer forms to become intermediate 46. Under alkaline conditions intermediate 46 transforms into the carboxylic acid 47, under the form of carboxylate given the alkaline conditions of the reaction.
Aza-Cope rearrangement of 4-butenyliminium ions [13]
4-Butenyliminium ions (48) can suffer aza-Cope rearrangements [13]. This is a carbon-carbon formation reaction [13]. The reaction takes place easily, mean mild conditions, but its reversibility character gives it no much utility in synthesis [13]. However, hydroxyl or alkyl substituents can change such condition and drive the reaction toward products (49), depending on the substitution pattern, concretely C-3 substituted in the butenyl group [13]. According to Figure 3, 48 with R4=OH, the reaction can be addressed to products (49) with an alkyl or hydroxyl as substituent at the C-3 position of butenyl by capturing the iminium salt in an intramolecular Mannich reaction [13,16]. See Figure 3 for the reaction and its comparison with the oxy-Cope rearrangement. This is an excellent pathway for the synthesis of 2-substituted pyrrolidines [13,16].
In Figure 3, the transition state can be imagined as a chair-like conformer. The driving force to provoke the pericyclic reaction (substrate 48) is evidently the charge deficiency of nitrogen. The demand of the iminium ion for neutrality makes possible the electronic movement through p orbitals from the electron-rich double bond C1=C2 to polarize bonds until the rupture of the bond C-3-C-4 (48). Without an oxygen (an electron donor group) or an alkyl group (an electron donor group) as the R4 substituent, the reaction becomes reversible, since the driving force (the electron demanding iminium ion) works also for the reaction in the inverse sense, is worth to say, bond polarization occurs from the electron-rich double bond C2=C3 through p orbitals until the iminium ion. Once the electron donor group is introduced in the molecule as a substituent at C-3, the reaction can be driven toward products quantitatively, since once the rupture C-3-C-4 occurs, and taking into account the considerations done in a previous contribution by us [12], the center C-3 that supports the OH (or R) group becomes cationic (eventually). This cationic charge (+ or ) is thus stabilized by the electron donor: O (OH) or C (R). In absence of OH (R), it is logical to think that the C+-3 (formed in the direct reaction) also induces the reverse reaction.
Substituted pyrrolidine derivatives; synthesis of acetylnicotine 53 and a bicyclic derivative 54 [17]
In the synthesis of an acetylnicotine derivative [17], pyridine 3-aldehyde was mixed with N-methyl-2-hydroxybutenamine (50) under thermal conditions in a benzene medium, camphor sulfonic acid was added as catalyst [17]. The yield was 84%. The iminium salt (51) passes by an aza-Cope rearrangement to intermediate 52. Intermediate 52 is then driven to an intramolecular Mannich reaction to afford acetylnicotine 53 [17]. Figure 4 and Figure 5 show the succinct reaction scheme and the mechanistic proposal respectively.
In a first step a nucleophilic attack from the basic nitrogen of N-methyl-2-hydroxybutenamine (50) provokes the breaking of the double bond of carbonyl, placing thus the electron excess over the oxygen atom. This attack converts the secondary amine into a quaternary ammonium, which subsequently frees a proton that neutralizes the negative charge of oxygen of the alcoxy function. We count now a tertiary amine and a diol in the adduct. By acid catalysis (camphor sulfonic acid) the alcohol derived from the ketone becomes protonated. This activates the substrate for an intramolecular nucleophilic attack over the carbon of the protonated alcohol function. The nucleophile is the nitrogen of the tertiary amine which converts after the attack in an iminium ion by dehydration of the protonated alcohol (-H2O). The iminium ion adopts a chair-like transition state for an aza-Cope rearrangement (51). After the pericyclic reaction (aza-Cope rearrangement) we obtain compound 52, which is still an iminium ion giving the substrate reactivity enough to induce a Mannich intramolecular reaction. Thus, as discussed previously regarding the reactivity of iminium ions, the wish of neutrality of nitrogen makes possible the concerted electronic movement to neutralize the iminium ion. The result is the pyrrolidine derivative 53, after returning the H+ to the catalyst.
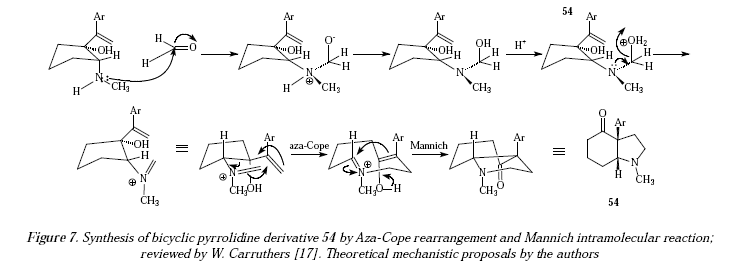
Another pyrrolidine derivative coming from iminium ion aza-Cope rearrangement is compound 54 in Figure 6 [17]. To form this bicyclic pyrrolidine derivative 54, the hydroxyl and amino must be neighboring substituents on a ring [17]. Due to this reaction the original ring is expanded by one carbon [17]. When OH and NHR are cis to each other, the product, the cis-fused bicyclic compound, is the result of high stereoselectivity [17]. There isnt much selectivity when the OH and NHR group are in a trans mutual position [17]. Figure 7 is the theoretical mechanism approach.
Comments
The synthetic route according to Figure 7 is the same to that described in Figure 5 for the synthesis of acetylnicotine and it corresponds to the same type of mechanistic comments there.
Synthesis of perhydrogephyrotoxin, key step
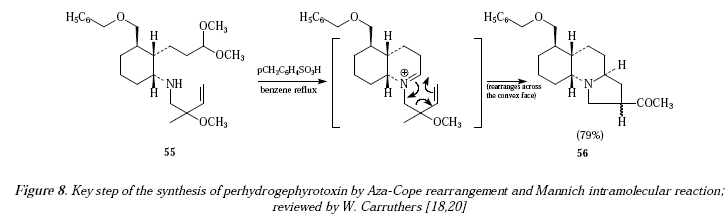
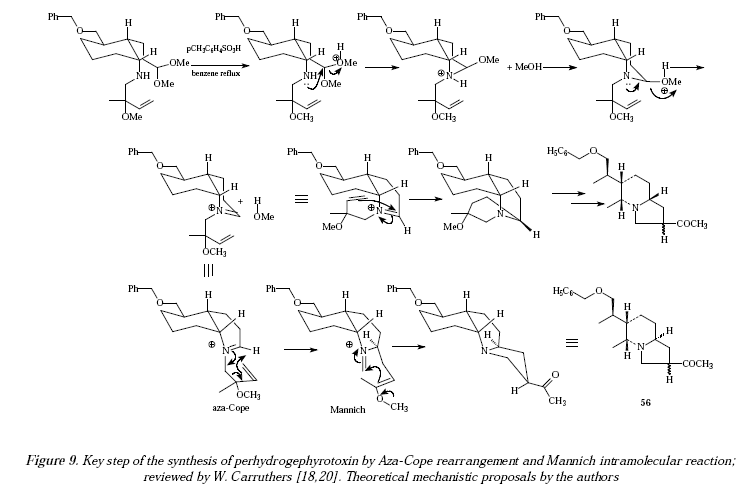
This synthetic sequence has found plenty of applications when synthesizing natural products [17]. For instance, during the synthesis of perhydrogephyrotoxin, the key step involves the using of butanylamine 55 which becomes the tricyclic 56 (79% yield) [17-19]. See the key step of this synthesis in Figure 8 as compiled by W. Carruthers and first investigated by Overman et al. [18,20]. The mechanistic proposals are shown in Figure 9.
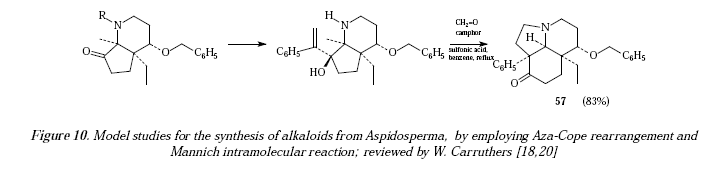
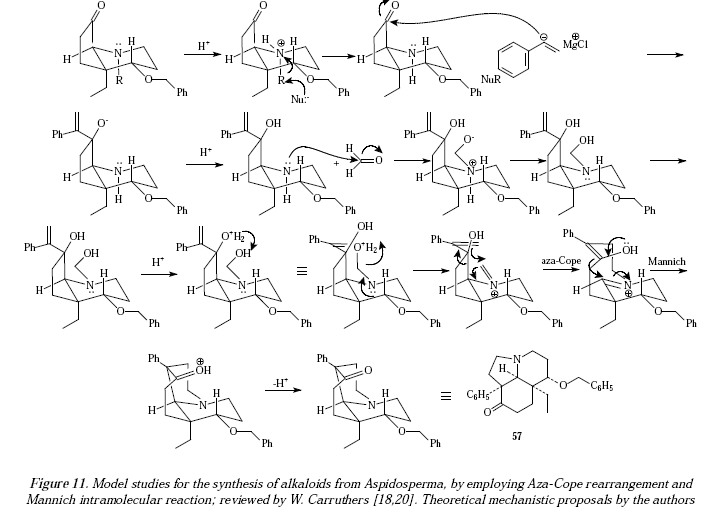
Also the sequence is employed in model studies for the synthesis of alkaloids from the genus Aspidosperma [17,18] where the tricyclic ketone 57 was obtained in 83% yield [20]. See Figure 10 for the reaction and Figure 11 for its mechanistic proposals.
ACKNOWLEDGEMENT
The authors express their gratitude to Prof. Eduardo Palenque, Department of Physics, Universidad Mayor de San Andrés, for his bibliographic support.
NOTAS
1Natural Product Laboratory, Phytochemistry, Chemical Sciences Department, School of Pure and Natural Sciences FCPN, Universidad Mayor de San Andres UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, phone +59122792238, La Paz, Bolivia, jabravo@umsa.bo, www.umsa.bo
1Natural Product Laboratory, Green Chemistry, Chemical Sciences Department, School of Pure and Natural Sciences FCPN, Universidad Mayor de San Andres UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, phone +59122772269, La Paz, Bolivia, jlvila@umsa.bo, www.umsa.bo
REFERENCES
1. Bravo, J. 2005, The organic chemistry notebook series, a didactical approach. Theoretical mechanistic approach to diasteroselective synthesis of cis-1,2-dialkenylcyclopropanols and subsequent oxy-Cope rearrangement by Jin Kun Cha et al, Rev. Bol. Quim., 23, 1-10. [ Links ]
2. Bravo, J.A., Mollinedo, P., Peñarrieta, J.M., Vila, J.L. 2013, Mechanistic views of intramolecular hydroxycyclopropanation of -vinyl carboxylic esters, Rev. Bol. Quim., 30 (1), 24-41.
3. Bravo, J.A., Vila, J.L. 2014, Mechanistic views of stereoselective synthesis of tri and tetra-substituted alkenes, part I; the organic chemistry notebook series, a didactical approach, nº 3. Rev. Bol. Quim., 31 (1), 61-67. [ Links ]
4. Bravo, J.A., Vila, J.L. 2015, Mechanistic views of stereoselective synthesis of tri and tetra-substituted alkenes, part II; the organic chemistry notebook series, a didactical approach, nº 4, Rev. Bol. Quim., 32 (1), 15-23. [ Links ]
5. Vila, J.L., Bravo, J.A. 2015, Synthesis of alkenes by fragmentation reactions; Mechanistic views; the organic chemistry notebook series, a didactical approach, nº 5, Rev. Bol. Quim., 32 (2), 37-44. [ Links ]
6. Bravo, J.A., Vila, J.L. 2015, Synthesis of alkenes by oxidative decarboxylation of carboxylic acids; Mechanistic views; the organic chemistry notebook series, a didactical approach, nº 6, Rev. Bol. Quim., 32 (3), 45-52. [ Links ]
7. Bravo, J.A., Vila, J.L. 2015, Synthesis of alkenes from ketones via arylsulphonyl-hydrazones; mechanistic views; the organic chemistry notebook series, a didactical approach, nº 7, Rev. Bol. Quim., 32 (4), 82-89. [ Links ]
8. Bravo, J.A., Vila, J.L. 2015, Stereospecific synthesis of alkenes from 1,2-diols; mechanistic views; the organic chemistry notebook series, a didactical approach, nº 8, Rev. Bol. Quim., 32 (5), 121-125. [ Links ]
9. Bravo, J.A., Vila, J.L. 2016, Synthesis of alkenes by Claisen rearrangement of allyl vinyl ethers, part I; mechanistic views; the organic chemistry notebook series, a didactical approach, nº 9, Rev. Bol. Quim., 33 (1), 27-33. [ Links ]
10. Bravo, J.A., Vila, J.L. 2016, Synthesis of alkenes by Claisen rearrangement of allyl vinyl ethers, part II; mechanistic views; the organic chemistry notebook series, a didactical approach, nº 10, Rev. Bol. Quim., 33 (2), 95-103. [ Links ]
11. Bravo, J.A., Vila, J.L. 2016, Synthesis of alkenes by Claisen rearrangement of allyl vinyl ethers, part III; mechanistic views; the organic chemistry notebook series, a didactical approach, nº 11, Rev. Bol. Quim., 33 (3), 127-133. [ Links ]
12. Bravo, J.A., Vila, J.L. 2017, Claisen rearrangement of allyl vinyl ethers to afford alkenes, part IV; mechanistic theoretical proposals; the organic chemistry notebook series, a didactical approach, nº 12, Rev. Bol. Quim., 34 (2), 40-49. [ Links ]
13. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 177-178. [ Links ]
14. Kreiser, W., Wurziger, H. 1975, Darstellung von olefinen durch [2,3]-sigmatrope umlagerung, Tetrahedron Lett., 16, 1669- 1672. [ Links ]
15. Baldwin, J.E., Walker, J.A. 1972, 2,3-Sigmatropic rearrangement of nucleophilic carbenes. A new approach to stereospecific synthesis of carbon–carbon bonds, J. Chem. Soc. Chem. Commun., 354-355.
16. Overman, L.E., Kakimoto, M., Okazaki, M.E., Meier, G.P. 1983, Synthesis applications of aza-Cope rearrangements. 11. Carbon-carbon bond formation under mild conditions via tandem cationic aza-Cope rearrangement-Mannich reactions. A convenient synthesis of polysubstituted pyrrolidines, J. Am. Chem. Soc., 105, 6622-6629. [ Links ]
17. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 178-179. [ Links ]
18. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 180. [ Links ]
19. Overman, L.E., Fukaya, C. 1980, Stereoselective total synthesis of (.+-.)-perhydrogephyrotoxin. Synthetic applications of directed 2-azonia-[3,3]-sigmatropic rearrangements, J. Am. Chem. Soc., 102, 1454-1456. [ Links ]
20. Overman, L.E., Freerks, R.L., Petty, C.B., Clizbe, L.A., Ono, R.K., Taylor, G.F., Jessup, P.J. 1981a, Diels-Alder reactions of 1- (acylamino)-1,3-dienes, J. Am. Chem. Soc., 103, 2816-2822. [ Links ]












 uBio
uBio