Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim v.34 n.1 La Paz abr. 2017
ARTÍCULOS ORIGINALES
HIGH CONTENTS OF OLEANOLIC ACID IN HIGHLAND BOLIVIAN PLANTS
ALTO CONTENIDO DE ACIDO OLEANOLICO EN PLANTAS BOLIVIANAS DE ALTITUD
Maribel Lozano, Yonny R. Flores, Giovanna R. Almanza*
Laboratorio de Química Bioorgánica, Instituto de Investigaciones Químicas IIQ, Ciencias Químicas, Facultad de Ciencias Puras y Naturales FCPN, Universidad Mayor de San Andrés UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, phone +59122795878, La Paz, Bolivia, gralmanza@umsa.bo, yrflores@umsa.bo
Received 01 30 2017 Accepted 04 17 2 017 Published 04 30 2017
ABSTRACT
Oleanolic acid is a pentacyclic triterpenoid widely distributed in the plant kingdom, it has several pharmacological properties and it has been marketed in China as hepatoprotective drug since decades. The plants are the main source of this interesting compound, in this sense; during this study we quantified this metabolite in several Bolivian highland plants (Tetraglochin cristatum, Lampaya castellani, Junellia seriphioides, Baccharis tola, Polylepis tomentella, P. hieronyni, P. besseri and Satureja boliviana) by a high-performance liquid chromatography (HPLC) method. The amount determined (2,87 to 22,69 mg/g) was higher than the concentration reported for the olive fruit (0,17-0,56 mg/g) and similar than the reported for olive leaf (31,0 mg/g) which is the main source of commercial oleanolic acid. So native species, such as Junellia seriphioides (22,69 mg/g), Lampaya castellani (13,87 mg/g), Baccharis tola (11,93 mg/g) and Polylepis besseri bark (11,06 mg/g) are an interesting potential natural source of oleanolic acid in the región.
Keywords: Oleanolic acid, HPLC quantification, Bolivian highland plants, Junellia seriphioides, Lampaya castellani, Baccharis tola, Polylepis besseri
*Correspond¡ng author: qiovyalmanza@qmail.com; qralmanza@umsa.bo
RESUMEN
Spanish title: Alto contenido de ácido oleanólico en plantas bolivianas de altitud. El ácido oleanólico es un triterpeno pentaciclico ampliamente distribuido en el reino vegetal, posee diversas propiedades farmacológicas y ha sido comercializado en China como droga hepatoprotectora desde hace décadas. Las plantas son la principal fuente de este interesante compuesto, en este sentido, durante este estudio cuantificamos este metabolito en varias plantas bolivianas de altura (Tetraglochin cristatum, Lampaya castellani, Junellia seriphioides, Baccharis tola, Polylepis tomentella, P. hieronyni, P. besseri y Satureja boliviana) por un método de cromatografía líquida de alta resolución (HPLC). La cantidad determinada (2,87 a 22,69 mg/g) fue mayor que la concentración reportada en frutos de oliva (0,17-0,56 mg/g) y similar a la reportada para hojas de oliva (31,00 mg/g) la fuente más importante de ácido oleanólico comercial. Por lo que especies nativas, como Junellia seriphioides (22,69 mg/g), Lampaya castellani (13,87 mg/g), Baccharis tola (11,93 mg/g) y Polylepis besseri corteza (11,06 mg/g) son una interesante potencial fuente natural de ácido oleanólico en la región.
INTRODUCTION
The natural product oleanolic acid (3p-hydroxyolean-12-en-28-oic acid) (Figure N° 1) is a bioactive pentacyclic triterpenoid compound that has been isolated from more than 1620 plant species, including many food and medicinal plants where it exists as free acid or as an aglycone of saponins [1, 2]
This metabolite is relatively non-toxic and possesses several promising pharmacological activities. Among them, the hepatoprotective effects were widely studied [1] and are the bases of a drug against liver disorders commercialized in China [3]. On the other hand, the antioxidant, anti-inflammatory, anti-HIV, and anticancer activities, among others, motivated a large number of studies, reviews and publications of this compound and its derivatives [1,2,3,4,5], giving as one of the main results, the possible commercialization of the first oleanolic acid-derived drug [5].
The compound is especially prevalent in the Oleaceae family, among which olive (Olea europaea) still serves as the main source of commercial oleanolic acid where it is found in a concentration from 0,17 (in fruits) to 31,0 mg/g (in leaves) [6,7,8]. Nevertheless, in our previous studies, we found oleanolic acid as one of the major secondary metabolites in several studied plants of Bolivian highlands (3500 to 4200 m.a.s.l.) [9]. Even plants that biosynthesize few secondary metabolites use part of their energy for the production of this compound, because it seems to play an important role during the periods of frost [9]. However, in our previous report, the measure amount of this metabolite was done by semi-quantitative classic methods, as thin layer chromatography (TLC), isolation by chromatography and recrystallization. In this study, we developed a protocol to quantify this important metabolite in plants by HPLC, because this technique is fast, reproducible, and reliable. Also, it only requires small amounts of sample, compared to classical techniques [10].
The plants are the main source of this metabolite [5,8,11]. So, for this study we selected eight plants from Bolivian highlands: Tetraglochin cristatum, Lampaya castellani, Junellia seriphioides, Baccharis tola, Polylepis tomentella, P. hieronyni, P. besseri and Satureja boliviana based on the analysis done by TLC in our previous report [9], where all of them showed a majority presence of this metabolite in the EtOH extract, in order to determine new potential natural sources of this interesting metabolite.
RESULTS AND DISCUSSION
In agreement of our previous studies [8], eight plants from Bolivian highlands (Tetraglochin cristatum, Lampaya castellani, Junellia seriphioides, Baccharis tola, Polylepis tomentella, P. hieronyni, P. besseri and Satureja boliviana) were considered for this study. The plant material was submitted to an EtOH extraction by maceration for 48 h and the content of oleanolic acid in the extracts was quantified by triplicate using the HPLC-DAD developed method.
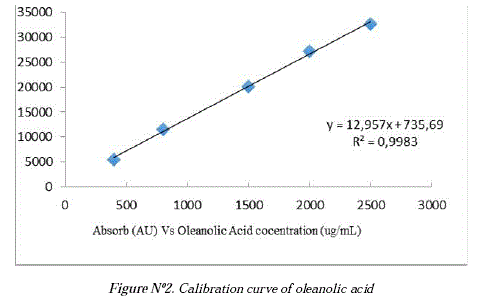
The HPLC developed method was based in three previously reported methods for triterpenic acids [12, 13, 14] and the calibration curve was done using the oleanolic acid isolated from Chenopodium quinoa residues after an acid hydrolysis of its saponins [13,15]. Figure N°l shows the HPLC-DAD chromatogram of oleanolic acid using a mixture of Acetonitrile/ 0,15 % Formic acid (93:7) as a mobile phase, an EclipsePlus C-18 column at a flow rate 1.0 ml/min, and detecting the analyte at 210 nm where the oleanolic acid appears at retention time ír= 8,2 min. Figure N°2 shows the calibration curve, which was obtained using the peak áreas of the standard whit different concentration. The área for every point was the average of three runs. The calibration curve was constructed in the range of 400-2500 ug/ml. The linear correlation equations and coefficient (r) was: 12,957x + 735,69 (r=0,9983.) showing a good linearity.
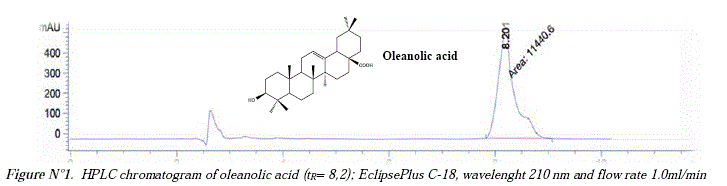
The limit of detection (LOD) was obtained as the sample concentration that caused a peak with a height three times the baseline noise level and the limit of quantification (LOQ) was calculated as 10 times the base noise level. Thus, the LOD and LOQ calculated were 138.96 μg/ml and 421,11 μg/ml respectively.
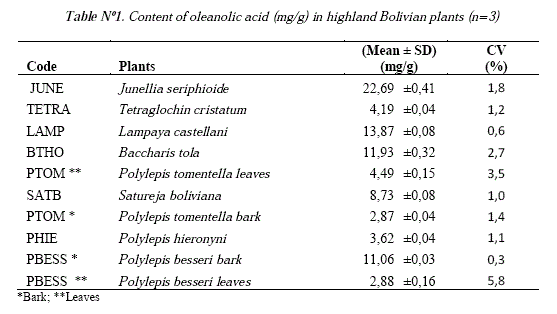
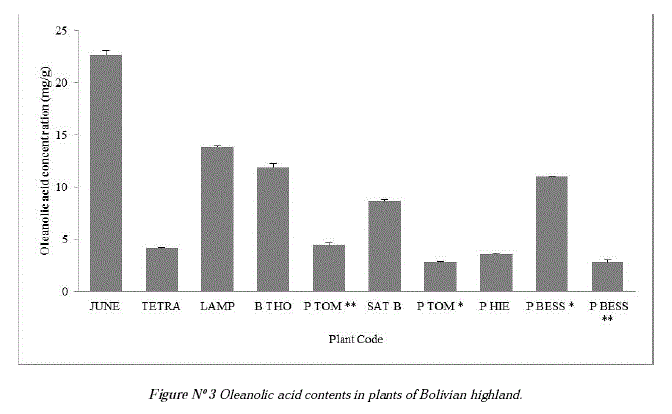
Then, the concentration of oleanolic acid was measured (Table N°l), showing that the species with high content of oleanolic acid are Junellia seriphioides, Lampaya castellani, Baccharis tola and Polylepis besseri bark. Nevertheless, it is important to note that all the plants evaluated showed a higher content of oleanolic acid than those found in the olive fruit (0,17-0,56 mg/g) and similar than the reported for olive leaf (31,0 mg/g) which is still used as main source of commercial oleanolic acid [7, 1]. In addition, one of these plants, Baccharis tola commonly known as "tola", is widely distributed in the Bolivian Highlands, so it could be used as new natural source of this compound. It is also interesting to note that the bark of Polylepis besseri has a good content of oleanolic acid, because the shredding bark of Polylepis is constantly peeling. Table N° 1 and Figure N° 3 show the high amounts of oleanolic acid in all the collected plants.
In conclusión, we developed a new HPLC-DAD protocol for the quantification of oleanolic acid in plant extracts. The method was used to quantify this compound in selected Bolivian highland plants determining high amounts in all of them, in particular in Junellia seriphioides, Lampaya castellani, Baccharis tola and Polylepis besseri bark which could be used as new natural source of this important metabolite.
EXPERIMENTAL
Plant material
Tetraglochin crístatum, Baccharis tola and Lampaya castellani were collected in Orinoca at Oruro department, Bolivia (3768 m.a.s.l); Junellia seriphioides was collected near to the Sajama mountain (3940 m.a.s.l) and finally Satureja boliviana and Polylepis species were collected at the Botanical Garden of the University of San Andrés (UMSA), located in Cota Cota town of La Paz City (3600 m.a.s.l.)- Voucher specimens of all plants where deposited in the Bolivian National Herbarium where were identified by the staff.
Oleanolic acid
The oleanolic acid, used as standard, was obtained of the industrial residue of Chenopodium quinoa Willd called "mojuelo" rich in saponins [15, 13]. That residue was extracted with EtOH/H2O (75/25) for 72 h at room temperature. The concentrated EtOH/H2O extract was submitted to an acid hydrolysis adding HCl 2N under reflux for 3 h, giving the sapogenin extract. The oleanolic acid was isolated out of that extract after a VLC chromatography on Silicagel G-60 and recrystallization, comparing the TLC and NMR data with those of the previous oleanolic acid isolated in our lab [13, 15].
Sample preparation
Aerial parts of Tetraglochin cristatum, Junellia seriphioides and Satureja boliviana; leaves of Lampaya castellani and Baccharis tola; bark and leaves of Polylepis tomentella, P. hieronyni, and P. besseri were dried in the bioorganic laboratory at room temperature for 2 or 3 days. 500 mg of the dried and ground plant material were extracted in 25 mi of EtOH 96 GL for 48 h. The extract was filtered through filter paper Whatman #4 and the residue was washed with 5 mi of EtOH, then the liquid extract was concentrated in vacuo. Before the analysis by HPLC the samples were dissolved and set, with MeOH (HPLC grade), to 5 mi volumetric flasks.
HPLC analysis
The HPLC data were recorded on Agilent Technologies HPLC 1100 series instrument consisting of a quaternary pump, continuous vacuum degasser, manual injection and column compartment coupled to a variable wavelength diode-array detector (DAD). The column was an Agilent Technologies EclipsePlus RP-C18 (4.6><250mmx5um) with a 0.5 uní pre-column filter (Agilent Technologies), and the column compartment was heated at 35 °C. The acetonitrile (HPLC grade, Sigma-Aldrich), formic acid >98% (Merck) and Ultrapure water at 18.3 MQ. resistance were used for
HPLC mobile phase. Water solvent was filtered through a 0.45 (im filtration system (Sartorius Stedim Biotech) before use.
Calibration curve
A series of oleanolic acid solutions were prepared in MeOH HPLC at five different concentrations (400, 800, 1500, 2000 and 2500 (xg/ml). The analyte was detected at 210 nm (DAD) and the área in the chromatogram at retention time 8,2 was recorder by triplicate. The analyte showed a linear relationship between 400 and 2500 (xg/ml yielded the following calibration curve y=12,957x+735,69 (R2= 0,9983).
Sample quantification
The samples were injected manually; the volume of standards and samples was 25 ul Chromatographic separation was done with an isocratic solvent of 93 % ACN as solvent D and 7% of 0.15 % (v) formic acid in water as a solvent C, at a flow rate of 1.0 ml/min. The solvent C was filtered with a Whatman 0.45 um PTFE/cellulose filter prior to use. Then, the área of the compound at retention time TR=8,2 min. was recorded by triplicate and the average was used for the calculation of the amount of oleanolic acid in the EtOH extract of every plant.
Statistics
The limit of quantification (LOQ) and the limit of detection (LOD) were calculated on the base of standard deviation of the response and the slope obtained from linear equation following equations: LOD = 3.3x(SD/slope "b") and LOQ = 10x(SD/ slope "b"); where SD is the standard deviation of the /-intercept.
ACKNOWLEDGEMENT
We are indebted to SIDA Swedish Agency for the financial support of the project "Biomolecules of industrial and medicinal interest. Anticancer". We also would like to thank to National Bolivian Herbarium for the identification of plant material.
REFERENCES
1. Pollier, J., Goossens A. 2012, Oleanolic acid, Phytochemistry, 77, 10-15. [ Links ]
2. Ovesná, Z., Vachalkova, A., Horvathova, K., Tothova, D. 2004, Pentacyclic triterpenoic acids: new chemoprotective compounds, Neoplasma, 51(5), 327-333. [ Links ]
3. Liu, J., 1995, Pharmacology of oleanolic acid and ursolic acid, J. Ethnopharmacol., 49, 57-68. [ Links ]
4. Liu, J., 2005, Oleanolic acid and ursolic acid: Research perspectives, J. Ethnopharmacol, 100, 92-94. [ Links ]
5. Sultana, N., Ata, A., 2008, Oleanolic acid and related derivatives as medicinally important compounds, Journal ofenzyme inhibition and medicinal chemistry, 23(6), 739-756. [ Links ]
6. Sporn, M. B., Liby, K. T., Yore, M. M., Fu L, Lopchuk J. M., Gribble G. W., 2011, New synthetic triterpenoids: Potent agents for prevention and treatment of tisuue injury caused by inflammatory and oxidative stress, Journal of Natural Products, 74, 537-545. [ Links ]
7. Romero, C, García, A., Medina, E., Ruiz-Mendez, M. V., De Castro, A., Brenes, M., 2010, Triterpenic acids in table olives, Food Chemistry, 118, 670-674. [ Links ]
8. Jager, S., Trijan, H., Kopp, T., Laszczyk, M.N., Scheffler, A., 2009, Pentacyclic triterpene distribution in various plants - Rich sources for a new group of multi-potent plant extracts, Molecules, 14, 2016-231. [ Links ]
9. Flores, Y., Sterner, O., Almanza, G. R., 2004, Oleanolic acid. Presence and importance in Highland Bolivian Plants, Revista Boliviana de Química., 21(1), 31 34. [ Links ]
10. Skoog, D.A., Holle,r F.J., Crouch, S.R., Principios de análisis instrumental, 2008 Cengage Learning, México. [ Links ]
11. Kowalski, R., 2007, Studies of selected plant raw materials as alternative sources of triterpenes of Oleanolic and Ursolic Acid types, J. Agruc. Food Chem,, 55, 656-662. [ Links ]
12. Zacchigna, M.., Cateni, F., Faudale, M., Sosa, S., Della Logia, R 2009, Rapid HPLC Analysis for Quantitative Determination of the Two Isomeric Triterpenic Acids, Oleanolic acid and Ursolic acid, in Plantago Major, Sel Pharm., 77, 79-86. [ Links ]
13. Lozano, M., Gonzales, E., Flores, Y., Almanza, GR, 2013, Effect in acute inflammation of sapogenin extract and Isolated sapogenins from quinoa waste (Chenopodium quinoa Willd), Revista Boliviana de Química, 30(2), 115-121. [ Links ]
14. Gbaguidi, F., Accrombessi, G, Moudachirou, M., Quetin-Leclercq, J., 2005, HPLC quantification of two isomeric triterpenic acids isolated from Mitracarpus scaber and antimicrobial activity on Dermatophilus congolensis, Journal of Pharmaceutical and Biomedical Analysis, 39, 990-995. [ Links ]
15. Flores, Y., Díaz, C, Garay, F., Colque, O., Sterner, O. and Almanza, G.R, 2005 "Oleanane-type triterpenes and derivatives from seed coat of Bolivian Chenopodium quinoa genotype salar", Revista Boliviana de Química, 22(1), 71-77. [ Links ]












 uBio
uBio 

