Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
Compartir
Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim vol.32 no.5 La Paz dic. 2015
ARTÍCULOS ORIGINALES
Stereospecific synthesis of alkenes from 1,2-diols; mechanistic views; the organic chemistry notebook series, a didactical approach, N ° 8
Síntesis estereoespecífica de alquenos a partir de 1,2-dioles; vistas mecanicísticas; de la serie: El cuaderno de notas de química orgánica, un enfoque didáctico, N°8.
José A. Bravo1,*, José L. Vila2
1 Department of Chemistry, Laboratorio de Fitoquímica, Instituto de Investigaciones en Productos Naturales IIPN, Universidad Mayor de San Andrés UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, Phone 59122792238, La Paz, Bolivia, jabravo@umsa.bo
2Department of Chemistry, Laboratorio de Síntesis y Hemisíntesis, Instituto de Investigaciones en Productos Naturales IIPN, Universidad Mayor de San Andrés UMSA, P.O. Box 303, Calle Andrés Bello s/n, Ciudad Universitaria Cota Cota, Phone 59122795878, La Paz, Bolivia, joselu62@hotmail.com
*Corresponding author: joseabravo@outlook.com
Abstract
This is the eighth chapter in the series: "The Organic Chemistry Notebook Series, a Didactical Approach".
The aim of this series of studies is to help students to have a graphical view of organic synthesis reactions of diverse nature. Here we discuss, from a mechanistic stand point, some methods for the stereospecific synthesis of alkenes from 1,2-diols. One of the best ones utilizes as precursors, the cyclic thionocarbonates obtained from the diol with thiophosgene. We describe by mechanisms, the use of 1,3-dimethyl-2-phenyl-1,3,2-diazophospholidine as an alternative for the decomposition of thionocarbonates into alkenes. We discuss by mechanisms, a general and unambiguous method for the interconversion of Z- and E-alkenes. Thus, the conversion of the (Z)-cyclooctene into the E isomer is detailed here. The mechanism of the conversion of meso-1,4-diphenylbutan-2,3-diol into (Z)-1,4-diphenyl-2-butene is here described. We have used various series of reactions reviewed by W. Carruthers in 'Some modern methods of organic synthesis', and we have proposed didactical and mechanistic views for them. This theme is included in the chapter "Formation of carbon-carbon double bonds" in the text mentioned above.
Keywords: Organic Chemistry, Alkenes, 1,2-diols, Stereospecific synthesis, Mechanisms of Reactions, W. Carruthers.
INTRODUCTION
During master classes of organic chemistry we noticed that students are confronted with a lack of knowledge with regard to mechanisms. For instance, oxidation-reduction reactions which are among the most commonly employed constitute a kind of black box for the student's mind. A mechanistic approach about any kind of reaction enhances the capacity of facing new reactions with respect to an understanding of all processes involved in them, and also develops synthetic creativity. As academics we feel concerned with the didactical importance of covering these needs in debutant students in organic synthesis. This, the stereospecific synthesis of alkenes from 1,2-diols, is the eighth study in: "The Organic Chemistry Notebook Series, a Didactical Approach" [1-7].
REACTIONS AND THEIR MECHANISTIC PROPOSALS, DISCUSSION
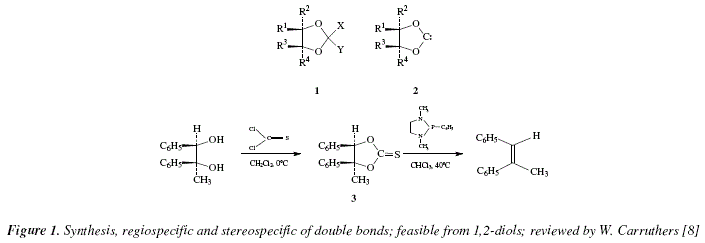
The synthesis, regiospecific and stereospecific of double bonds, is feasible from 1,2-diols [8]. All procedures include the formation of an intermediate of the type 1 (Fig. 1), which decomposes into products [8]. One of the best of the various methods available uses the cyclic thionocarbonates 3, (Fig. 1) [8]. These are obtained from reaction of the diol with thiophosgene, Fig. 1 [8]. The fragmentation started with the thionocarbonates transformed into alkenes by heating with triethylphosphate; more recently, the conversion is done only at 25-40 °C by using thionocarbonate with 1,3-dimethy-2-phenyl-1,3,2-diazophospholidine [8,9]. It is believed a concerted process or a process that includes an intermediate carbene (2) [8].
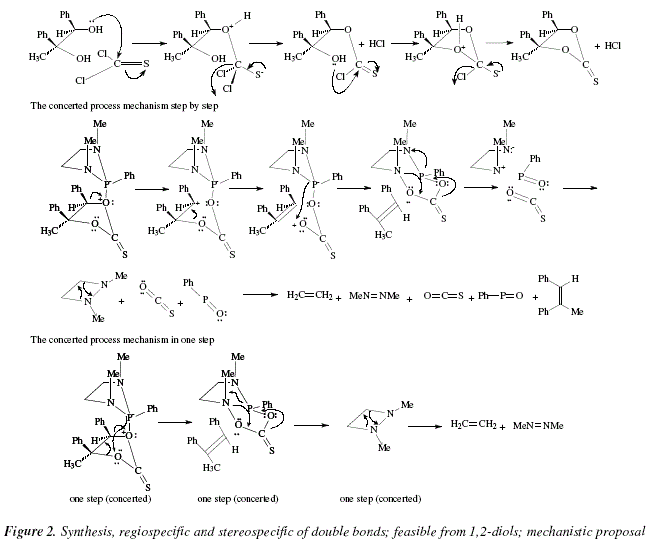
The mechanistic approach to the reactions appearing in Fig. 1, are exposed in Fig. 2.
Comments
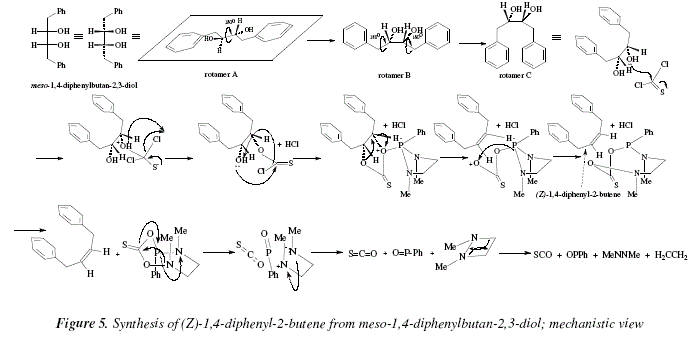
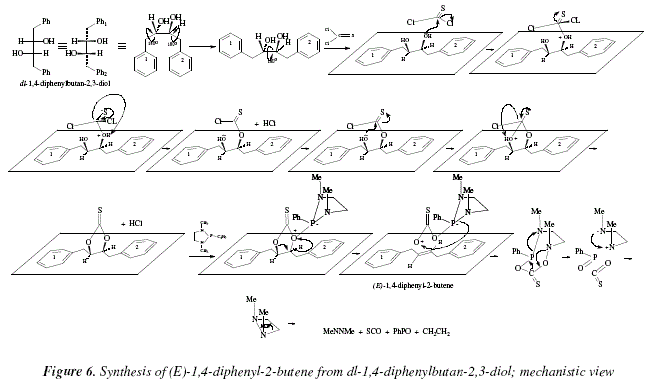
In Figure 2, a nucleophilic attack from the oxygen of the diol over the carbon of the analogue-of-carbonyl C=S, is shown in the first step. The protonated ether formed eliminates a HCl to afford an analogue of a thioester, the S=C-O (with the bond 'pi' shared between O and S [S-C=O<->S=C-O]). The carbon of C=S (analogue-of-carbonyl) suffers a new nucleophilic attack, this time from the other hydroxylic oxygen to replace another chloride that correlates with the liberated H+ from the just formed ether linkage (second HCl). Now, both oxygens are activated by the thionocarbonate toward elimination. However the process takes place only due to the catalysis-type action of the 1,3-dimethy-2-phenyl-1,3,2-diazophospholidine. The term "concerted mechanism" implies an only step when all excision-formation of bonds occur simultaneously. It is also implied the absence of ions, however with didactical purposes we explain the concerted mechanism (an only step) in three derived steps that include precisely ions. Besides the alkene, the residue is an eight-membered polyheterocycle. Also, we propose a disintegration of this cycle that occurs in one concerted step, in three didactical steps. The resulting four-membered diazocycle, disintegrate in a concerted way. The final products are: the alkene pursued, ethylene, azomethane [10], carbonyl sulfide [11], and PhP=O.
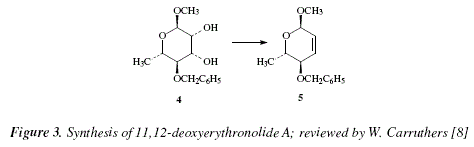
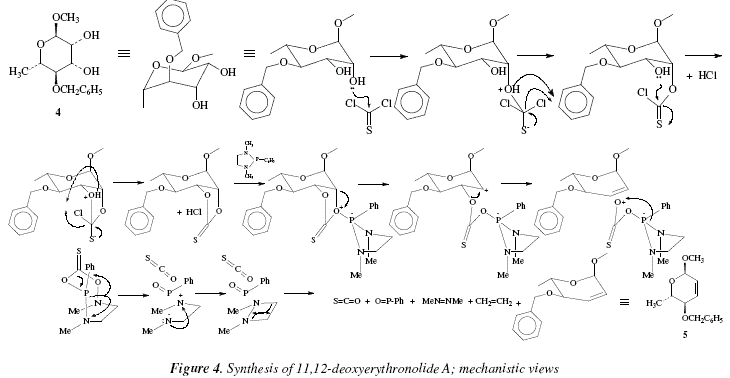
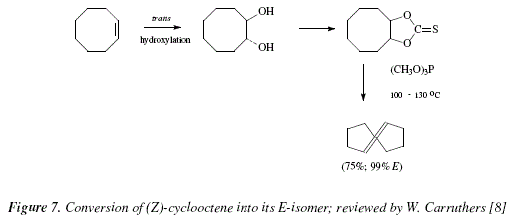
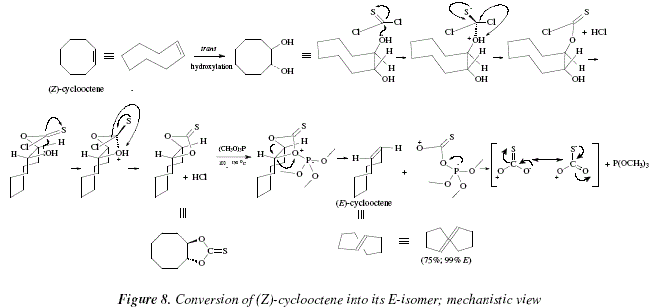
Given the mild conditions, the reaction can be used in the synthesis of complex molecules and sensitive due to the presence of many functional groups [8]. As an example, the 3,5-acetonide of the complex macrolide named as erythronolide A, can be transformed into the 11,12-deoxy derivative; hence, the diol 4 gives 5, with a yield of 75 % [8]. The reactions proceed by keeping stereospecificity following a syn elimination route [8]. This is an excellent method to produce cycloalkenes suffering strain due to 1,2-substituents mutually hindered [8]; see Fig. 3. This method and the trans perhydroxylation [12] constitute the virtuous cycle for the isomerization between Z- and E-alkenes [8]. Figure 4 shows the corresponding mechanisms.
Comments
The same remarks done for Fig. 2 are applicable for Fig. 4.
Other examples are the conversion of meso-1,4-diphenylbutan-2,3-diol into (Z)-1,4-diphenyl-2-butene [8], and the synthesis of its E-isomer starting from the dl-1,4-diphenylbutan-2,3-diol [8]. The mechanistic development of these transformations is pictured in Fig. 5 and 6 respectively.
Figure 7 represents the isomerization Z>E of (Z)-cyclooctene as reviewed by W. Carruthers [8].
Figure 8 shows mechanistically how (Z)-cyclooctene can be converted in its E-isomer [8], following a similar protocol to the one used previously in this discussion. Alternatively, another derivative of 1,2-diol, the benzylidene, can be treated with butyl-lithium [8]. This method is however of limited application, given the high susceptibility of many functional groups toward the butyl lithium [8]. The method is suitable with lightly substituted alkenes only [8,13]. Other methods with stereospecificity include acetals derived from the diol with ethyl-orthoformate [8,14], or N,N-dimethylformamide dimethyl acetal [8,15].
ACKNOWLEDGEMENT
The authors express their gratitude to Prof. Eduardo Palenque from the Department of Physics, Universidad Mayor de San Andrés, for his bibliographic support.
REFERENCES
1. Bravo, J. 2005, Rev. Bol. Quim., 23, 1. [ Links ]
2. Bravo, J.A., Mollinedo, P., Peñarrieta, J.M., Vila, J.L. 2013, Rev. Bol. Quim., 30, 24. [ Links ]
3. Bravo, J.A., Vila, J.L. 2014, Rev. Bol. Quim., 31, 61. [ Links ]
4. Bravo, J.A., Vila, J.L. 2015, Rev. Bol. Quim., 32, 15. [ Links ]
5. Vila, J.L., Bravo, J.A. 2015, Rev. Bol. Quim., 32, 37. [ Links ]
6. Bravo, J.A., Vila, J.L. 2015, Rev. Bol. Quim., 32, 45. [ Links ]
7. Bravo, J.A., Vila, J.L. 2015, Rev. Bol. Quim., 32, 82. [ Links ]
8. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 162-165. [ Links ]
9. Corey, E.J., Hopkins, P.B. 1982, Tetrahedron Lett., 23, 1979. [ Links ]
10. Rebbert, R.E. , Ausloos, P. 1962, J. Phys. Chem., 66, 2253. [ Links ]
11. Protoschill-Krebs, G., Wilhelm, C., Kesselmeier, J. 1996. Atmospheric Environment, 30, 3151. [ Links ]
12. Carruthers, W. Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 364. [ Links ]
13. Hines, J.N., Peagram, M.J., Thomas, E.J., Whitham, G.H. 1973, J. Chem. Soc. Perkin I, 2332. [ Links ]
14. Hiyama, T., Nozaki, H., 1973, Bull. Chem. Soc. Japan., 46, 2248. [ Links ]
15. Hanessian, S., Bargiotti, A., La Rue, M. 1978, Tetrahedron Lett., 737. [ Links ]












 uBio
uBio