Services on Demand
Journal
Article
Indicators
Related links
Share
Revista Boliviana de Química
On-line version ISSN 0250-5460
Rev. Bol. Quim vol.31 no.1 La Paz June 2014
ARTICULO ORIGINAL
SECONDARY METABOLITES FROM PODOCARPUSPARLATOREI PILGER
Marcelo Dávila![]() l; Olov Stenier
l; Olov Stenier![]() ; Nefson Hinojosa
; Nefson Hinojosa![]()
aCenter for Analysis and Synthesis, Lund University, P.O. Box 124, SE-221 00, Lund, Sweden; bCentro de Tecnología Agroindustrial, Facultad de Ciencias y Tecnología, Universidad Mayor de San Simón, Cochabamba, Bolivia.
*CorrespondÍng author: hinoiosanelfgivahoo.com.ar
ABSTRACT
Phytochemical investigation on Podocarpus parlatorei Pilger led to the isolation of three biflavones together with three diterpenes. Their chemical structures were elucidated by spectroscopic analyses, primarily 1 and 2D NMR spectroscopy as well as high-resolution mass spectrometry. The isolated compounds are reported from this plant for the first time.
Keywords: Biflavone, diterpene, Podocarpus parlatorei
RESUMEN
La investigación fitoquímica en Podocarpus parlatorei Pilger condujo al aislamiento de tres biflavonoides junto con tres diterpenos. Sus estructuras químicas fueron determinadas por análisis espectroscópicos, principalmente por NMR 1 y 2D, así como por espectrometría de masas de alta resolución. Los compuestos aislados se reportan en esta planta por primera vez.
INTRODUCTION
The genus Podocarpus compromises at least 107 species of shrubs and trees of the family Podocarpaceae, which are mainly distributed in the southern hemisphere [1]. These species have proven to be valuable plants for the discovery of natural producís like terpenoids, nor-diterpenoid dilaciones, flavonoids and biflavonoids [2].
Podocarpus parlatorei is a perennial native tree, which occurs naturally in Bolivia and Argentina [3]. It is distributed in the sub-Andean Amazonian Forest regions of Bolivia and is an important timber source in its native áreas [4]. As part of our interest in phytochemical investigations of Bolivian plants, extracts of the leaves of Podocarpus parlatorei were fractionated and six natural producís were isolated and characterized. All the identified compounds are know from previous reports, but were obtained from this plant for the first time.
RESULTS AND DISCUSSION
The ethanolic extract of the aerial parts of P. parlatorei was divided using liquid-liquid partition to afford a chloroform and a water-methanol fraction. The chloroform fraction was subsequently purified using a combination of vacuum liquid chromatography, column chromatography and size exclusión chromatography (Sephadex LH-20), to afford three biflavonoids (1-3) and three diterpenoids (4-6) (Figure 1). The structural elucidation of compounds was achieved on the basis of :H and 13C NMR spectra, 2D COSY, NOESY, HMQC and HMBC experiments, and HR-ESI-MS.
The elemental composition of compound (1) was determined to be C33H24O10, based on the ID NMR spectra as well as HR-ESI-MS data (molecular ion peak at m/z 581.1453 [M+H]+), indicating twenty-two degrees of unsaturation. The :H NMR spectrum (see Table 1) displays signáis corresponding to twenty-four protons: three methoxy groups, two singlets at 13.05 and 12.91, which are characteristic of hydrogen-bonded hydroxyl groups (5-OH and 5"-OH), a broad hydroxy signal at 5 10.88 (1H, br), the remaining twelve signáis appeared between 5 6.00 and 8.50 ppm.
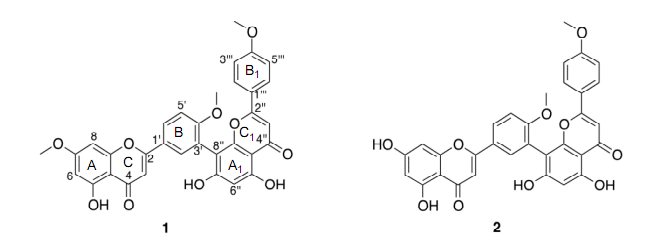

Moreover, the COSY and HMBC spectra suggest that the following protón systems are part of the structure: a tetra-substituted aromatic ring (A-ring) with a pair of /weto-coupled protons at 5 6.36 (1H, d, J= 2.2; 6-H) and 6.79 (1H, d, J= 2.2; 8-H), a 1,3,4 tri-substituted ring (B-ring) at 5 7.37 (1H, d, J= 8.9), 8.08 (1H, d, J= 2.4) and 8.23 (1H, dd, J= 2.4, 8.9), a singlet at 8 7.00 (1H, s; 3-H), characteristic for a protón at C-3 in flavonoids, a penta-substituted aromatic ring (Aj-ring) with a single protón at 5 6.42 (1H, s; 6"-H), an AA'BB' aromatic spin system at 8 7.60 (2H, d, J= 9.0) and 6.93 (2H, d, J= 9.0) (Bj-ring), and a signal at 8 6.90 (1H, s) was attributed to 3"-H. Analysis of 13C NMR spectrum and a combination of COSY, HMQC and HMBC experiments suggested that 1 is a biflavonoid linked between C-3' and C-8", which was confirmed by HMBC correlations between 2'-H and C-8". Therefore, the structure of (1) was determined to be 5,5",7"-trihydroxy-4',4'",7-trimethoxy-3',8"-biflavone (sciadopitysin), previously isolated from Taxus cuspidata [5].
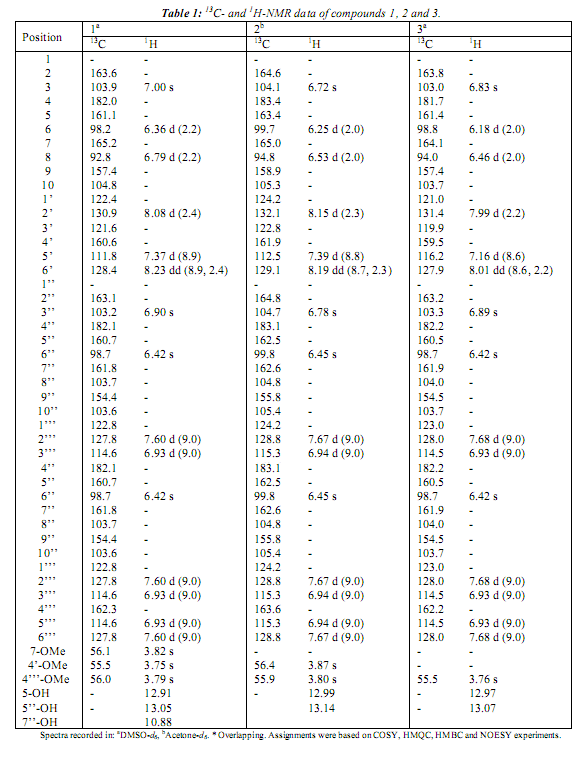
Compound (2) (C32H2201o) and (3) (C3iH2o010) are biflavonoids with molecular mass ionpeak at m/z 567.1290. [M+H]+ and m/z 553.1161 [M+H]+, respectively. The NMR features of (2) and (3) showed some similarities to those of 1 (Table 1). The analysis of :H NMR and HR-ESI-MS data showed that (2) has one methoxy group less than (1), while (3) revealed the absence of two methoxy groups. Carbons and protons were assigned on the basis of 2D NMR data (COSY, HMQC and HMBC). Based on their data the structures were established as 5,5",7,7"-tetrahydroxy-4',4'"-dimethoxy-3',8"-biflavone (2) (isoginkgetin) and 4',5,5",7,7"-pentahydroxy-4'"-methoxy-3',8"-biflavone (3) (podocarpusflavone A). Compound (2) has previously been isolated from Ginkgo biloba [6], while (3) has been reported from Podocalyx loranthoides [7].
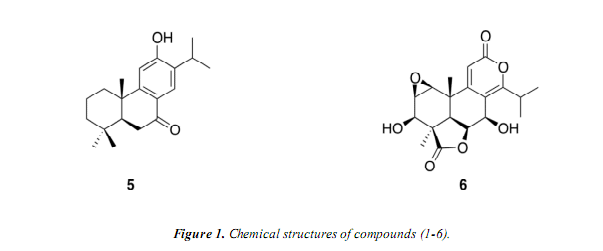
The molecular formula of compound (4) (C2oH2803) was established by 13C NMR data (Table 2) and HR-ESI-MS (m/z 317.2130 [M+H]+), which indicates seven degrees of unsaturation. The 13C NMR spectrum suggest the presence of one carboxylic carbón and six carbón signáis assigned to an aromatic ring, of which one signal was downshifted to 5 154.8 and should be an oxygenated aromatic carbón. These facts indicated that (4) is tricyclic. Its ATR-IR spectrum displayed bands for a hydroxy group at 3387, a carbonyl group at 1693 and aromatic double bonds at 1589 an"1. The :H NMR and 2D COSY spectrum showed that the following protón systems are present in the molecule: A
methine hydrogen of an isopropyl group at 8 3.24 (15-H) and two non-equivalent methyl doublets at 5 1.31 (16-H3) and 1.32 (17-H3). The aromatic región exhibited two or/Ao-coupled doublets at 5 6.52 (12-H) and 6.92 (11-H), which indicated the presence of a 1,2,3,4-tetrasubstituted aromatic ring. Three methylene groups assigned to ring A at 5 2.22/1.27 (1-H2), 2.02/1.55 (2-H2) and 2.21/1.08 (3-H2) were identified, as well as a spin system consisting of a methine protón at 8 1.44 (5-H), a methylene group at 8 2.23/1.94 (6-H2) and an additional methylene group at 8 2.92/2.61 (7-H2). Besides the methyl groups of the isopropyl group, two methyl singlets resonating at 5 1.28 and 8 1.12 were assigned to C-18 and C-20, respectively. Onthe basis of HMBC spectrum, the methyl protons of isopropyl 16-H3 and 17-H3 exhibited correlations to C-14; and the hydrogen 12-H showed correlations to C-13, C-14 and C-9, which indicated that the isopropyl group was at carbón C-14 and the hydroxy group at C-13. Another further correlations were observed from 11-H to C-10 as well as to the aromatic carbons C-8 and C-13. On the ring A, the methyl at 8 1.28 (18-H3) was located at C-4 via HMBC correlations to C-3, C-4, C-5 and the carboxylic carbón C-19. Furthermore, long-range correlations between the hydrogens at 3-H2, 5-H, I8-H3 and the carboxylic carbón C-19 showed that the carboxyl group was present at C-4. The methine protón 5-H exhibited correlations to C-4, C-6, C-7, C-9, CIO, C-18 and C-20. Finally, the protons 20-H3 showed correlations to C-l, C-5, C-9 and C-10. The relative stereochemistry of (4) was determined by the correlations present in a NOESY spectrum, and suggested that 5-Hoc, I-Ha (5 1.25), 3-Ha (8 1.08), 7-Ha (5 2.61) and 18-H3 are co-facial, while 20-H3,2-Hp (5 2.02) and 6-Hp (8 1.94) are on the opposite face. Thus, the structure of (4) was concluded to be identical to 4/?-carboxy-19-nor-totarol, previously reported from Podocarpus nagi [8].
The elemental composition of (5), C20H28O2, was determined by HR-ESI-MS (m/z 301.2179 [M+H]+). Its ATR-IR spectrum displayed bands for a hydroxy group at 3237, a carbonyl at 1648 and aromatic double bonds at 1592 an"1. As with (4), compound (5) is a tricyclic natural product with seven degrees of unsaturation, including one aromatic ring and one carbonyl group, which is conjugated with the aromatic ring. The :H NMR spectrum (Table 2)
showed the presence of three methyl groups at 8 0.95 (19-H3), 1.03 (18-H3) and 1.23 (20-H3), one isopropyl group consisting of a methine protón at 5 3.21 (15-H) and two methyl doublets at 5 1.20 (16-H3) and 1.21 (17-H3). Furthermore, two aromatic singlets at 8 6.76 (11-H) and 7.79 (14-H), which apparently are situated in positions 1 and 4 in the aromatic ring. In addition to this, three methylene groups at 8 2.28/1.52 (1-H2), 1.83/1.68 (2-H2) and 1.54/1.31 (3-H2) and a second order systemat 8 2.61/2.59 (6-H2) and 1.84 (5-H) can be observed. Fromthe HMBC data, the position of the carbonyl function was placed at C-7 by correlations between 14-H as well as 6-H2 and C-7. The isopropyl and hydroxy groups are located at C-13 and C-12, respectively, by the correlations observed from 14-H to C-15, C-9, C-12 and from 11-H to C-8, C-13 and C-12. All these results indicated that the structure of (5) was identicalto 12-hydroxyabieta-8,ll,13-triene-7-one (sugiol), previously reported fromPodocarpus nagi [8].
The molecular formula of compound (6) was established to be C19H22O7 from the [M+H]+ ion peak at m/z 363.1450 determined by HR-ESI-MS and the ID NMR data (Table 2), which indicate nine degrees of unsaturation. The ATR-IR spectrum displayed bands at 3444, 1770 and 1714 cm"1, which are indicative of hydroxy, y-lactone and 5-lactone moieties, respectively. The :H NMR spectrum showed the presence of two methyl singlets at 8 1.31 and 1.33, they were assigned to C-18 and C-20, respectively; one singlet olefinic protón at 8 6.27 (11-H). Furthermore, two protons at 8 3.56 (1-H) and 3.32 (2-H), correlated in the HMQC spectrum with carbons at 8 56.9 and 50.6, respectively, which allowed the establishment of the presence of an epoxy ring located at C-l/C-2 based on the HMBC correlations from 1-H to C-2, C-3, C-9 and C-10, and correlations from 2-H to C-l, C-3, and C-4. Three spin systems were evident in the COSY spectrum: correlations between two methyl doublets at 8 1.20 (16-H) and 1.17 (17-H), attributed to an isopropyl group, and the isopropyl methine protón at 8 3.27 (15-H), which appeared as a septet; the protón signal at 8 3.32 (2-H) correlated with 3.56 (1-H) and 4.23 (3-H), which coupled with the hydroxy protón at 8 5.29 (3-OH); the protón signal at 8 4.88 (6-H) coupled with 2.05 (5-H) and 5.21 (7-H), which correlated with the hydroxy protón at 8 5.78 (7-OH). HMBC correlations: from 11-H to C-8, C-12, C-14; as well as 7-H to C-9 and C-14; and from 15-H to C-8 and C-14 confirmed the assignment of S-lactone ring carbons. The location of the y-lactone between C-19 and C-6 were by correlations observed in the HMBC spectrum between 18-H3 and C-19, and between 7-H, 5-H and C-6. The NOESY spectrum exhibited correlations between 5-H, 6-H, 7-H and 18-H3, suggesting the (3-orientation of the y-lactone and the hydroxy group at 8 5.78 (7-OH). NOESY correlations between 1-H, 2-H, 3-H and 18-H3 suggest that the hydroxy group at 8 5.29 (3-OH) was P-oriented, as well as the epoxy group. Henee, compound (6) was concluded to be a nor-diterpene dilactone type-A, known as nagilactone C [9, 10]. Compound (6) has previously been isolated from Podocarpus nagi [9]
EXPERIMENTAL
General experimental procedures
The optical rotation was measured with a Perkin-Elmer 341 polarimeter at 20 °C. HR-ESI-MS spectra were recorded with a Waters Q-TOF Micro system spectrometer, using H3PO4 for calibration and as internal standard. ATR-IR spectrum was obtained with a Bruker-Alpha-P spectrometer. :H NMR (400 MHz) and 13C NMR (100 MHz) were measured with a Bruker DRX spectrometer; the spectra were recorder in DMSO-4? (solvent residual signáis![]() (solvent residual signáis
(solvent residual signáis![]() (solvent residual signáis at 5H 3.31 and 8C 49.00). The chemical shift
(solvent residual signáis at 5H 3.31 and 8C 49.00). The chemical shift![]() are given in ppm, and coupling constants (J) in Hz. Vacuum liquid chromatography (VLC) was carried out using TLC grade silica gel (Merck), while column chromatography (CC) were run on silica gel 60 (230-400 mesh, Merck) and Sephadex LH-20. TLC analyses were carried out on silica gel GF254 precoated plates (Merck); chromatograms were visualized under a UV lamp and by spraying with vanillin (6%)-sulfuric acid (1.5%)-ethanol solution, followed by heating.
are given in ppm, and coupling constants (J) in Hz. Vacuum liquid chromatography (VLC) was carried out using TLC grade silica gel (Merck), while column chromatography (CC) were run on silica gel 60 (230-400 mesh, Merck) and Sephadex LH-20. TLC analyses were carried out on silica gel GF254 precoated plates (Merck); chromatograms were visualized under a UV lamp and by spraying with vanillin (6%)-sulfuric acid (1.5%)-ethanol solution, followed by heating.
Plant material
The aerial parts of P. parlatorei Pilger were collected from south of Cochabamba-Bolivia at 2900 meters above sea level in April 2008. A voucher specimen (MZ-3007) was deposited at the National Herbarium "Herbario Nacional Martín Cárdenas" at Cochabamba-Bolivia
Extraction and isolation
The dried and pulverized whole plant material of P. parlatorei (300 g) was extracted with ethanol. The solvent was recovered in vacuo to yield a crade extract (10 g). A volume of methanol-water (5:1) was added to the crade extract and the mixture was agitated thoroughly to form a suspensión, which was extracted with chloroform three times (A, B and C). After evaporation, the fraction A was subject to VLC eluted with heptane-ethyl acétate gradient to give seven fractions (A1-A7). The compound (5) (5 mg) was purified from fraction A3 by CC using a mixture of heptane-ethyl acétate (80:20) as a solvent. Fraction A4 was applied to repeated silica gel VLC (chloroform-methanol (80:20) and chloroform) and CC on Sephadex LH-20 (chloroform-methanol (50:50)) to yield compound (4) (5 mg). Fraction C was subject to VLC on silica gel eluted with chloroform-methanol to give two main fractions Cl and C2. Fraction Cl (1.4 g) was subject to CC on Sephadex LH-20 eluted with chloroform-methanol (50:50) to give eight fractions (1-8). Fraction 1 (140 mg) was precipitated with methanol to give compound (6) (5 mg). Fraction 2 (200 mg) was precipitated with methanol to give compound (3) (3 mg). Fraction 3 (380 mg) was precipitated with methanol to give compound (1) (7 mg). Finally, fraction 8 was washed with cold acetone and filtered to yield compound (2) (1.5 mg).
5,5",7"-trihydroxy-4',4'",7-trimethoxy-3',8"-biflavone (sciadopitysin) (1). Yellow powder. mp 285-288 °C. :H NMR (DMSO-4 400 MHz) and 13C NMR (DMSO-4 100 MHz), see Table 1. HRMS-ESI m/z 581.1453 [M+H]+. Calculated for C33H24O10, 581.1448.
5,5",7,7"-tetrahydroxy-4',4"'-dimethoxy-3',8"-biflavone (isoginkgetin) (2). Yellow powder. mp 202-204 °C. :H NMR (acetone-^ 400 MHz) and 13C NMR (acetone-^ 100 MHz), see Table 1. HRMS-ESI m/z 567.1290 [M+H]+. Calculated for C32H22O10, 567.1291.
4',5,5'",7,7"'-pentahydroxy-4'"-methoxy-3',8"-biflavone (podocarpusflavone A) (3). Yellow powder. mp 240-242 °C. *H NMR (DMSO-4 400 MHz) and 13C NMR (DMSO-4 100 MHz), see Table 1. HRMS-ESI m/z 553.1161 [M+H]+. Calculated for C3iH20O10, 553.1135.
4P-carboxy-19-nor-totarol (4). Yellow oil. [a]D20 + 119 (c 0.38, MeOH). ATR-IR vmax 3387, 2954, 2872, 1693, 1589, 1448, 1357, 1274, 1183 cm1. lH NMR (methanol-^ 400 MHz) and 13C NMR (methanol-d4100 MHz), see Table 2. HRMS-ESI m/z 317.2130 [M+H]+. Calculated for C2oH2803, 317.2117.
12-Hydroxyabieta-8,ll,13-triene-7-one (sugiol) (5). White, amorphous solid. mp 290-292 °C. [a]D20 + 20 (c 0.06, MeOH). ATR-IR vmax 3237, 2955, 2929, 2863, 1648, 1592, 1303, 1268 cm"1. *H NMR (methanol-^ 400 MHz) and 13C NMR (methanol-^ 100 MHz), see Table 2. HRMS-ESI m/z 301.2179 [M+H]+. Calculated for C20H28O2, 301.2168.
Nagilactone C (6). White, amorphous powder. mp 300-305 °C. [a]D20 + 5 (c 0.88, DMSO). ATR-IR vmax 3444, 3274, 2249, 2122, 1770, 1714, 166, 1050, 1007, 820, 758 cm"1. lH NMR (DMSO-d6 400 MHz) and 13C NMR (DMSO-4100 MHz), see Table 2. HRMS-ESI m/z 363.1450 [M+H]+. Calculated for C19H22O7, 363.1444.
ACKNOWLEDGEMENTS
The authors gratefully acknowledge financial support from Swedish International Development Cooperation Agency (SIDA). We thank Lie. Modesto Zarate from the National Herbarium "Herbario Nacional Martín Cárdenas", for the taxonomic determination of the plant material.
REFERENCES
[1] BARKER, N. P, MULLER, E. M., MILL, R. R. South African Journal of Science, 2004, 100, 629. [ Links ]
[2] ABDILLAHI, H. S., STAFFORD, G. I., FINNIE, J. F., VAN STADEN, J. South African Journal of Botany, 2010, 76, 1. [ Links ]
[3] <http://www.tropicos.org> TROPICOS.ORG. Tropicos.org. Missouri Botanical Garden, (20 Sep 2012). [ Links ]
[4] WOOD, J. R. I. La guia "Darwin" de las Flores de Los Valles Bolivianos. 2005, Darwin Initiative, London. [ Links ]
[5] CHOI, S.-K, OH, H.-M, LEE, S.-K., JEONG, D. G, RYU, S. E, SON, K.-H., HAN, D. C, SUNG, N.-D, BAEK, N.-I, KWON, B.-M. Natural Product Research, 2006, 20, 341. [ Links ]
[6] HANRAHAN, J. R, CHEBIB, M, DAVUCHERON, N. L. M, HALL, B. J, JOHNSTON, G. A. R. Biorg. Med. Chem. Lett, 2003, 13, 2281. [ Links ]
[7] SUÁREZ, A, I., DÍAZ M, B, DELLE MONACHE, F, COMPAGNONE, R. S. Fitoterapia, 2003, 74, 473. [ Links ]
[8] YING, B.-P, KUBO, I. Phytochemistry, 1991, 30, 1951. [ Links ]
[9] HAYASHI, Y, TAKAHASHI, S., ONA, H, SAKAN, T. Tetrahedron Lett., 1968, 9, 2071. [ Links ]
[10] ITÓ, S, MITSUAKI, K. Heterocycles, 1976, 4, 595. [ Links ]












 uBio
uBio