Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim vol.30 no.2 La Paz 2013
ARTICULO ORIGINAL
FLAVONOIDS FROM BACCHARIS POLYCEPHALA WEDDELL
Marcelo Dávilaa,b; Olov Sternera; Nelson Hinojosab, *
aCentre for Analysis and Synthesis, Lund University, P.O. Box 124, SE-221 00, Lund, Sweden; bCentro de Tecnología Agroindustrial, Facultad de Ciencias y Tecnología, Universidad Mayor de San Simón, Cochabamba, Bolivia.
*Corresponding author: hinojosanel@yahoo.com.ar
Received 12 10 2013
Keywords: Flavonoid, Baccharispolycephala Weddell, 5,7,4'-trihydroxyflavanone, 5,7-dihydroxy-4'-methoxyflavanone, 5-hydroxy-4', 7-dimethoxyflavanone, 5,7,4'-trihydroxyflavonol, 5-7-dihydroxy-4'-methoxyflavonol, 5,7-dihydroxy-4 '-methoxyflavone.
ABSTRACT
Six flavonoids were obtained in a phytochemical investigation of the aerial parts of Baccharis polycephala. The structures of the natural products were established by NMR, ATR-IR spectroscopy and high resolution mass spectroscopy as 5,7,4'-trihydroxyflavanone (1), 5,7-dihydroxy-4'-methoxyflavanone (2), 5-hydroxy-4',7-dimethoxyflavanone (3), 5,7,4'-trihydroxyflavonol (4), 5,7-dihydroxy-4'-methoxyflavonol (5) and 5,7-dihydroxy-4'-methoxyflavone (6). The presence of the isolated compounds in Baccharis polycephala is reported for the first time.
RESUMEN
En el presente estudio fitoquímico seis flavonoides fueron aislados de las partes aéreas de Baccharis polycephala. Sus estructuras químicas fueron determinadas por métodos espectroscópicos de NMR, ATR-IR y HRMS. Los compuestos aislados son: 5,7,4'-trihidroxiflavanona (1), 5,7-dihidroxi-4'-metoxiflavanona (2), 5-hidroxi-4',7-dimetoxiflavanona (3), 5,7,4'-trihidroxiflavonol (4), 5,7-dihidroxi-4'-metoxiflavonol (5) y 5,7-dihidroxi-4'-metoxiflavonoide (6), estos son reportados en Baccharis polycephala por primera vez.
INTRODUCTION
Flavonoids are important compounds isolated from a wide range of plants. Diets with high content in flavonoids are associated with positive health effects and the prevention for several diseases. Many flavonoids have been shown to possess potentially useful biological activities, such as antibacterial, antifungal, and antiviral activity [1]. Additionally, pharmacological studies have demonstrated the anti-inflammatory effects [2] and antioxidant capacity [3] of several flavonoids, and some have been demonstrated to possess cytotoxic properties and anticancer activity [4, 5]. The American genus Baccharis comprises more than 500 species of which many have been investigated and shown to be rich sources of various flavonoids [6]. Baccharis species are widely used in traditional medicine, all over America, and the list of the reported traditional uses is long [6-8]. For example, Baccharis articulata (Lam.) Pers. (Carquejilla) is traditionally used as a diuretic and digestive in Brazil, Uruguay and Argentina, as well as is used the Baccharis conferta H.B.K. (Quauhizquiztli) in the region of Veracruz, Mexico. Baccharis obtusifolia HBK, Baccharis latifolia (R. et P.) Pers., Baccharis pentlandii D.C. and Baccharis subulata Wedd. are used to treat rheumatism, liver diseases, wounds and ulcers. In the course of our continuous studies of Bolivian plants, a phytochemical investigation of Baccharis polycephala was performed. No previous investigation regarding the contents of secondary metabolites of this species has been reported in the literature. B. polycephala is a shrub growing in the mountainous region of Bolivia and Argentina [9], and the fractionation of an extract led to the isolation and characterization of six flavonoids.
RESULTS AND DISCUSSION
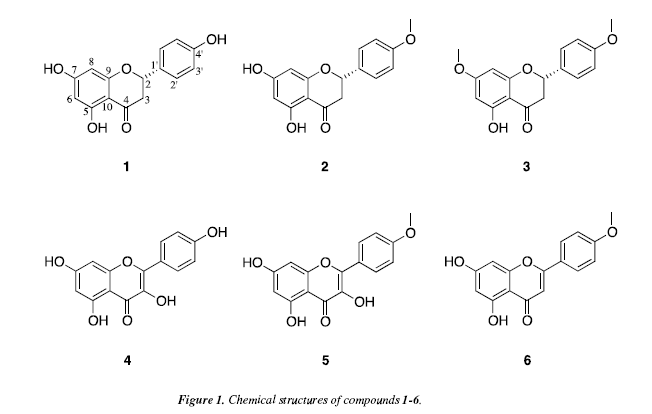
The dried aerial parts of Baccharis polycephala were ground and extracted with first hexane, then chloroform and finally ethanol, and TLC analyses suggested that the secondary metabolites are present in the chloroform extract. This was fractionated by vacuum liquid chromatography, column chromatography and centrifugal preparative chromatography, as described in the experimental part, eventually affording the six pure compounds shown in Figure 1. The structures of compounds 1-6 (Figure 1) were established by a combination of 1H and 13C NMR spectroscopy, both 1D and 2D experiments, ATR-IR spectroscopy, and high resolution ESI mass spectrometry. Compound 1 was obtained as yellow oil. Its molecular formula C15H12O5 was established on the basis of the ESI-HRMS data, and as the 13C NMR spectrum only displayed 13 signals it was assumed that 1 contains two pairs of equivalent carbons. 1 consequently has ten degrees of unsaturation. The ATR-IR spectrum showed bands at 1637, 1518 and 3306 cm-1 indicating the presence of a conjugated carbonyl group, an aromatic ring and hydroxyl group, respectively. The 1H NMR spectrum (recorded in CD3OD) revealed the presence of three-proton spin system at 5 5.34 (1H, dd, J=13, ![]() which are typical proton signals of a flavanone [10]. The aryl substituent at C-2 is obviously para-substituted, and this was indicated by the second order pattern of protons
which are typical proton signals of a flavanone [10]. The aryl substituent at C-2 is obviously para-substituted, and this was indicated by the second order pattern of protons ![]() respectively. HMBC correlations from 2-H to C-1' and C-2/6', from 2/6'-H to C-2, C-2/6' and C-4', as well as from 3/5'-H to C-1', C-3/5' and C-4' confirm that the C-2 substituent is 4-hydroxyphenyl and that 1 in fact is a flavanone. The remaining benzene ring has two protons at 5 5.90 (1H, d, J=2.1; 8-H) and 5 5.88 (1H, d, J=2.1; 6-H) and is consequently 1,2,3,5-tetrasubtituted. HMBC correlations from 6-H to C-5, C-7, C-8 and C-10, as well as from 8-H to C-6, C-7, C-9 and C-10 establish that 1 is 5,7,4' -trihydroxyflavanone, known as Naringenin and previously reported from Euphorbia tuckeyana [11]. The NMR data of compounds 2 and 3 (recorded in CDCl3) are similar to those of 1, except that 2 has one and 3 two methoxy groups. The ESI-HRMS data show that the elemental composition of 2 and 3 are C16H14O5 and C17H16O5, respectively. The same NMR experiments as discussed above were carried out with 2 and 3, showing that both are flavanones oxygenated in positions 5, 7 and 4'. As a confirmation, the chelated 5-OH is visible in the 1H NMR spectra (recorded CDCl3) of both compounds at just above 12 ppm. HMBC correlations from the methoxy protons to C-4' in 2 and to C-7 as well as C-4' in 3 demonstrate that 2 is 5,7-dihydroxy-4'-methoxyflavanone while 3 is 5-hydroxy-4',7-dimethoxyflavanone. 2 was isolated from Artemisa campestris subsp. maritima [12], while 3 has previously been reported from an Aniba species [13].
respectively. HMBC correlations from 2-H to C-1' and C-2/6', from 2/6'-H to C-2, C-2/6' and C-4', as well as from 3/5'-H to C-1', C-3/5' and C-4' confirm that the C-2 substituent is 4-hydroxyphenyl and that 1 in fact is a flavanone. The remaining benzene ring has two protons at 5 5.90 (1H, d, J=2.1; 8-H) and 5 5.88 (1H, d, J=2.1; 6-H) and is consequently 1,2,3,5-tetrasubtituted. HMBC correlations from 6-H to C-5, C-7, C-8 and C-10, as well as from 8-H to C-6, C-7, C-9 and C-10 establish that 1 is 5,7,4' -trihydroxyflavanone, known as Naringenin and previously reported from Euphorbia tuckeyana [11]. The NMR data of compounds 2 and 3 (recorded in CDCl3) are similar to those of 1, except that 2 has one and 3 two methoxy groups. The ESI-HRMS data show that the elemental composition of 2 and 3 are C16H14O5 and C17H16O5, respectively. The same NMR experiments as discussed above were carried out with 2 and 3, showing that both are flavanones oxygenated in positions 5, 7 and 4'. As a confirmation, the chelated 5-OH is visible in the 1H NMR spectra (recorded CDCl3) of both compounds at just above 12 ppm. HMBC correlations from the methoxy protons to C-4' in 2 and to C-7 as well as C-4' in 3 demonstrate that 2 is 5,7-dihydroxy-4'-methoxyflavanone while 3 is 5-hydroxy-4',7-dimethoxyflavanone. 2 was isolated from Artemisa campestris subsp. maritima [12], while 3 has previously been reported from an Aniba species [13].
Compound 4 contains only unsaturated carbons according to the NMR data (recorded in CD3OD), which are typical for a flavonol. According to ESI-HRMS experiments, 4 has the elemental composition C15H10O6, which is in agreement with a trihydroxylated flavonol. The 1H NMR spectrum showed two signals for meta-coupled protons at 5 6.37 (1H, d, J=2.1; 8-H) and 5 6.16 (1H, d, J=2.1; 6-H) with the corresponding HMBC correlations discussed for 1 above, indicating that ring A has hydroxyl groups in positions 5 and 7. Two doublets at 5 8.07 (2H, d, J=9; 2/6'-H) and 5 6.89 (2H, d, J=9; 3/5'-H) suggest the presence of a typical AA'BB' coupling system, in this case a para-hydroxy substituted ring B. Compound 4 was therefore found to be 5,7,4'-trihydroxyflavonol, identical to Kaempferol previously isolated from Polygonatum polyanthemum [14]. The NMR data of compound 5 (recorded in CD3OD) is similar to those of 4, except for the presence of a methoxy group in 5. Consequently, the elemental composition of 5 should be C16H12O6 and this was confirmed by ESI-HRMS experiments. The position of the methoxy group was determined by the HMBC correlation from the methoxy protons to C-4', and 5 is 5,7-dihydroxy-4'-methoxyflavonol. 5 has previously been reported from a Hippophae species [15].The 1D NMR data of compound 6 do not suggest that it is a flavanone or flavonol, although it appears to be a flavonoid. The elemental composition of 6 was determined to be C16H12O5, from ESI-HRMS experiments and the 1D NMR spectra recorded in DMSO-d6.
The 1H NMR spectrum showed a signal at![]() characteristic for a proton at C-3 in flavones. The presence of a doublet at
characteristic for a proton at C-3 in flavones. The presence of a doublet at![]() (both with J=2.0) was attributed to the protons at 6-H and 8-H in ring A, while doublets at 5 7.11 (2H; 3/5'-H) and 5 8.04 (2H; 2/6'-H) (both with J=8.9) were attributed to the AA'BB' spin system in ring B. HMBC correlations from 3-H to C-4, C-10 and C-1', from 2/6'-H to C-2 and C-4', as well as from 3/5'-H to C-1' and C-4' determine the structure of 6, which consequently is 5,7-dihydroxy-4'-methoxyflavone. 6 has previously been reported from Cirsium japonicum [16]. All data reported for the six compounds are in agreement with those in this investigation.
(both with J=2.0) was attributed to the protons at 6-H and 8-H in ring A, while doublets at 5 7.11 (2H; 3/5'-H) and 5 8.04 (2H; 2/6'-H) (both with J=8.9) were attributed to the AA'BB' spin system in ring B. HMBC correlations from 3-H to C-4, C-10 and C-1', from 2/6'-H to C-2 and C-4', as well as from 3/5'-H to C-1' and C-4' determine the structure of 6, which consequently is 5,7-dihydroxy-4'-methoxyflavone. 6 has previously been reported from Cirsium japonicum [16]. All data reported for the six compounds are in agreement with those in this investigation.
EXPERIMENTAL
General experimental procedures
The optical rotations were measured with a Perkin-Elmer 341 polarimeter at 20°C. Infrared spectral data were collected on a Bruker-Alpha-P ATR-IR spectrometer. ESI-HRMS spectra were recorded using Waters Q-TOF Micro system spectrometer, using H3PO4 for calibration and as internal standard. 1H NMR (400 MHz) and 13C NMR (100 MHz) were measured with a Bruker DRX400 spectrometer; the spectra were recorded in chloroform-d (solvent residual signals at 5H 7.26 and 5C 77.16), methanol-d4 (solvent residual signals at 5H 3.31 and 5C 49.00) and DMSO-d6 (solvent residual signals at 5H 2.50 and 5C 39.52). The chemical shift (5) are given in ppm, and coupling constants (J) in Hz. Vacuum liquid chromatography (VLC) and centrifugal preparative TLC (CTLC) separations were carried out using TLC grade silica gel (Merck), while column chromatography (CC) were run on silica gel 60 (230-400 mesh, Merck). TLC analyses were carried out on silica gel GF254 precoated plates (Merck); chromatograms were visualized under a UV lamp (254 nm) and by spraying with vanillin (6%)-sulfuric acid (1.5%)-ethanol solution, followed by heating.
Plant material
The whole aerial parts of Baccharis polycephala Weddell were collected from south mountainous region of Cochabamba-Bolivia at 3500 m of altitude in April 2008. A voucher specimen (MZ-1860) was deposited at National Herbarium "Herbario Nacional Martin Cardenas".
Extraction and isolation
The plant material was air-dried at room temperature, finely grounded (300 g) and successively extracted, with solvents of ascending polarity at room temperature for 24 hours each solvent (hexane, chloroform and ethanol). From all crude extracts, only the chloroformic extract was further subjected to isolation. The chloroformic extract was concentrated under reduced pressure to give a dark brown mass (5 g). This extract was suspended in dichloromethane, a precipitate (fraction A) was observed. After filtering, the supernatant was evaporated in vacuum to give fraction B. Fraction A (1 g) was subjected to VLC eluted with toluene-acetone (8:2) to give three fractions (A1-3). A yellow precipitate was observed in fraction A1. After filtering, A1 (107 mg) was subjected to CTLC (Chromatotron) using toluene-acetone (9:1) to afford 4 (6 mg). Separation on fraction B (4 g) was performed by VLC, eluted with heptane-EtOAc (9:1) to give six main fractions (B1-6). Then, the yellow mixture in fraction B4 (200 mg) was further separated into eight fractions (B4A-H) by CC eluted with CHCl3-MeOH (1:1) to give 2 (1.5 mg) (fraction B4D). Compound 3 (2 mg) was obtained from fraction B4B by CC with toluene-heptane (8:2) as eluent. Compound 6 (3 mg) was precipitated from fraction B6. The mother liquid of fraction B6 (2 g) was separated by VLC using CHCl3-MeOH (30:1) to give five sub-fractions (B6A-E). Compounds 5 (5 mg) and 1 (7 mg) were obtained from sub-fraction B6C (100 mg) by CC with CHCl3-MeOH (1:1) as eluent.

ACKNOWLEDGMENTS
The authors gratefully acknowledge financial support from Swedish International Development Cooperation Agency (SIDA). We thank Lic. Modesto Zárate from the National Herbarium "Herbario Nacional Martin Cardenas, for the taxonomic determination of the plant material.
REFERENCES
[I] CUSHNIE, T. P. T., LAMB, A. J. International Journal of Antimicrobial Agents, 2005, 26, 343.
[2] GUARDIA, T., ROTELLI, A. E., JUAREZ, A. O., PELZER, L. E. Il Farmaco, 2001,56, 683.
[3] SEYOUM, A., ASRES, K., EL-FIKY, F. K. Phytochemistry, 2006, 67, 2058. [ Links ]
[4] HARBORNE, J. B., WILLIAMS, C. A. Phytochemistry, 2000, 55, 481. [ Links ]
[5] KANADASWAMI, C., LEE, L. T., LEE, P. P. H., HWANG, J. J., KE, F. C., HUANG, Y. T., LEE, M. T. In Vivo, 2005,19, 895. [ Links ]
[6] ABAD, M. J., BERMEJO, P. ARKIVOC, 2007, 76. [ Links ]
[7] ABAD, M. J., BESSA, A. L., BALLARIN, B., ARAGON, O., GONZALES, E., BERMEJO, P. J. Ethnopharmacol., 2006,103, 338. [ Links ]
[8] MORALES, G., PAREDES, A., SIERRA, P., LOYOLA, L. A. Molecules, 2008,13, 790. [ Links ]
[9] http://www.tropicos.org TROPICOS.ORG. Tropicos.org. Missouri Botanical Garden, (20 Sep 2012). [ Links ]
[10] KOTESWARA RAO, Y., VIMALAMMA, G., VENKATA RAO, C., TZENG, Y.-M. Phytochemistry, 2004, 65, 2317. [ Links ]
[11] DUARTE, N., LAGE, H., FERREIRA, M.-J. U. Planta Med, 2008, 74, 61. [ Links ]
[12] VASCONCELOS, J. M. J., SILVA, A. M. S., CAVALEIRO, J. A. S. Phytochemistry, 1998, 49, 1421. [ Links ]
[13] ROSSI, M. H., YOSHIDA, M., SOARES MAIA, J. G. Phytochemistry, 1997, 45, 1263. [ Links ]
[14] GVAZAVA, L., KIKOLADZE, V. Chem. Nat. Compd., 2011, 47, 818. [ Links ]
[15] PANDURANGAN, N., BOSE, C., BANERJI, A. Biorg. Med. Chem. Lett., 2011, 21, 5328. [ Links ]
[16] PENG,W. Chem. Nat. Compd., 2011, 47, 279. [ Links ]












 uBio
uBio