Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
Compartir
Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim v.25 n.1 La Paz dic. 2008
IN VITRO PRODUCTION OF LEISHMANICIDAL ALKALOID 2-PHENYL-QUINOLINE BY ANGOSTURA LONGIFLORA KRAUSE A
Marina Guarachia, Magaly Paza, Rogelio Chuquib, Michel Sauvainc, Ninoska Floresa, Ma Teresa Alvareza, Alberto Giméneza,*
aInstituto de Investigaciones Fármaco Bioquímicas-IIFB, Facultad de Ciencias Farmaceuticas y Bioquímicas, Universidad Mayor de San Andrés, P. O. Box 3239 La Paz, Bolivia,
bComunidad Tacana Santa Rosa de Maravilla, Provincia Abel Iturralde, La Paz, Bolivia
cMisión IRD, Institut de Recherche pour le Développement, P. O. Box 9214 La Paz, Bolivia
Key Words: Angostura longiflora; callus; leishmanicidal alkaloids; 2-phenyl-quinoline; Bolivia
ABSTRACT
Angostura longiflora (K.) Kallunki (Rutaceae) is a medicinal wood tree used by Amazonian tribes for the treatment of stomach parasites and leishmaniasis. Cell cultures from leaves of this species were obtained on Murashige and Skoog (MS) medium supplemented with kinetine (kin) and 2,4-dichlorophenoxyacetic acid (2,4-D) based on a factorial design, where concentrations of sucrose and phosphate were changed. The increasing of sucrose and phosphate enhanced the specific growth rate from 0.16 to 0.33d-1. The leishmanicidal alkaloid, 2-phenyl-quinoline was isolated from calli and identified by means of nuclear magnetic resonance (NMR) and mass (EI) spectra. This is the first report concerning the production, isolation and chemical characterization by NMR and mass spectra of the main alkaloid, in Evanta leaves, 2-phenyl-quinoline from cell cultures of A. longiflora.
Corresponding author: agimenez@megalink.com
INTRODUCTION
Angostura longiflora (Krause) Kallunki (Rutaceae) is a tree 10-15 m tall that thrives along areas where the Andes meet the humid forest in the Amazon planes[1,2]. The bark of this tree, known locally as Evanta, is traditionally used as a beverage for the treatment of stomach parasites, diarrhea with blood, as a fortifying to strengthen physical conditions of children and adults and is also used as a cataplasm and infusion for the treatment of leishmaniasis[3]. Chemical[4] and biological[5] studies on this species have reported a total of 13 leishmanicidal quinoline alkaloids from bark, leaves and roots. Among these alkaloids, 2-phenyl-quinoline is the main component and it is found together with minor amounts of other 4-methoxy, 2-aryl and 2-alkyl quinolines[6]. The total alkaloid-extracts from the bark of this tree are currently undergoing clinical studies to evaluate the efficacy of the plant in the treatment of cutaneous leishmaniasis[7]. Hence, the use of biotechnological tools to establish cell cultures of Evanta as potential productive source of the active metabolites, might avoid depredation of the environment[8]. Cell cultures could also facilitate the production, under controlled conditions, of homogenous mixtures of active compounds[9,10]. Several strategies, such as modifying the nutrient concentrations in the culture medium, are being followed in order to favor specific physiological behavior when cells are cultivated in vitro. Nutrients are important to obtain the adequate productive conditions, and vary for each plant species[11]. Thus the aim of this study was to enhance, through a factorial design based on sucrose and phosphate, both the cell biomass concentration and the specific growth rate, in order to have a continuously available cell source for the production of the leishmanicidal quinolinic alkaloids.
RESULTS AND DISCUSSION
Among the nutritional factors, sucrose is the common carbon source for the plant tissue and cell culture, serving as a principal energy source and a component for biosynthesis[12]. Phosphate has been reported to regulate synthesis of secondary metabolites in plant cell cultures[13]. Here, it was demonstrated that the variation of sucrose and phosphate had a positive effect over biomas growth. When, KH2PO4 and C12H22O11 sources were increased, the production of biomas was also increased compared to the basal MS medium composition. The highest value of cell growth rate was 0.33 day-1 (Figure 1), two-fold the initial growth rate.
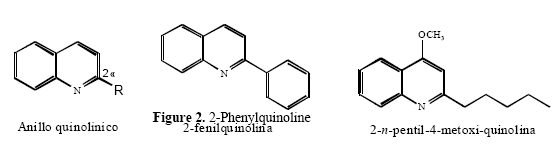
According to the results the increasing in the amounts of nutrients enhances the biomas production and might increase the secondary metabolites synthesis[14,15]. The main alkaloid, 2-phenyl-quinolinee was detected by TLC analysis of the organic extract from cell culture (Rf 0.57). It was isolated fromm the raw organic extract using preparative TLC. The 1H NMR spectrum exhibited signals only in the aromatic region, integrating for a total of 11 protons. Att downfield, two doublets, each integrating to one proton, being easily distinguished at δ 8.23 (J=8.6Hzz) and 8.18 (J==7.9Hz), characteristic of the protons in para and peri positions respect to the nitrogen in the quinoline skeleton. A doublet at δ 8.20, (J=7.0HHz), integrating to 2 protons together with two doublet signals, integrating to 3 protons, at δ 7.48 ((J=7.0Hz) and 7.55 (J=7.0Hz) are consistent with a mono substituted aromatic ring, confirming the presence of the phenyl ring mmoiety in carbon 2 in the quinoliine substructurre of 2-phenyll-quinoline (Figure 2).
Mass spectrum, under electron impact mode of the substance showed a molecular ion peak at m/z 2055 (80%) which was in agreement with the molecular formula C15H11N (Calc. 205.08911, observed 2005.0890), compatible with the 2-phenyl-quinoline the main alkaloid reported from leaves and bark from A. longiflora, it is found in 0.15% (w/w) aproximately from dry leaves[5]. The isolation of this component was about 0.06% (w/w) from dried cells.
Studies performed on leaves extracts from the tree growing in nature showed that is found together with at least other 5 minor quinoline alkaloids [5].
In vitro optimization of alkaloids production in cell cultures using elicitors, and simultaneous analysis of the minor chemical components are currently being done.
EXPERIMENTAL SECTION
Materials and Methods
Plant material and cell culture
Specimens of A. longiflora K. kallunki were collected in Santa Rosa de Maravilla community, Bolivia (S13°56’587, W0.68°00’308) and they were deposited at the Bolivia National Herbarium (Voucher specimen SD17).
Leaves of young plants were surface-sterilized and dissected in order to inoculate on Murashige and Skoog medium. MS medium consist on (mg/L): NH4NO3 1650, H3BO3 6.2, CaCl2 332.2, CoCl2 0.025, CuSO4·6H2O 0.025, Na2EDTA 37.26, FeSO4·7H2O 27.8, MgSO4 180.7, MnSO4·H2O 16.9, Na2MoO4·2H2O 0.25, KI 0.83, KNO3 1900, KH2PO4 170, ZnSO4·H2O 8.6, myo-inositol 100, nicotine acid 0.5, pyridoxine·HCl 0.5, thiamine·HCl 0.1, glycine 2, agar 8000, sucrose 30000, supplemented with kinetine (0.1 ppm) and 2,4-dichlorophenoxyacetic acid (5 ppm). All the chemicals used were of analytical grade (Sigma, Germany) except sucrose. The pH was adjusted to 5.8 and then the culture medium was autoclaved at 121 ºC for 20 min.
The cell lines were maintained at 28±2 ºC into an acclimated darkness-chamber (Paz et al., 2007) and the callus were re-inoculated in fresh medium every 25 to 30 days.
22 Factorial design for biomass optimization
Three experiments were performed in triplicate inoculating approximately 0.5 g of 4-5 weeks-old callus on the middle of petri dishes with MS medium. The composition in g/L of sucrose and mg/L of phosphate was modified as follow: 1) 30 and 170, 2) 40 and 170, 3) 30 and 227, and finally 4) 40 and 227, respectively. The other constituents were kept constant. The maximum growth rate was analyzed by using response surface methodology. This allowed eficient fitting and checking of the second-degree polynomial model which was used to estimate the predicted response. STATISTICA v.6 (StatSoft Inc., USA) was applied for the analysis of statistical data. All results were analyzed with factorial ANOVA (p ≥ 0.05).
Specific cell growth rate
Cellular growth was determined by fresh weight every 7 days for a period of 35 days. The growth indexes were calculated according to the formula: GI = mi/mo, where m0 and mi (g) are the initial and final callus fresh weight, respectively.
The specific cell growth rate (µ, day–1) was calculated from the growth indexes data, according to the formula: µ = (mi/mo)t, where t is the cultivation time (days).
Extraction and characterization of 2-phenyl-quinoline from cell culture.
The calluses were dried at 60 ºC until constant weight. The dried cells were homogenized in a mortar (8.77 g) and then, extraction was made with dichloromethane (10:1 v/w) for 48 h in dark conditions at room temperature. The extract obtained was filtered and concentrated under reduced pressure at 25 ºC. The residue (57.23 mg, 0.65%) was analyzed by TLC (Thin Layer Chromatography) using silica gel aluminum coated plates (250 µm, Whatman, Germany) eluted with the system n-Hexane:CH2Cl2(8:2), the visualization of spots was done by UV (254 and 366nm). The main alkaloid, 2-phenyl-quinoline was detected as dark spot comparing with a reference substance (Sigma-Aldrich 299650). The crude extract was analyzed by PTLC (1mm, glass plates Merck) and eluted with the system n-Hexane:Diethyl ether (8:2, three elutions). The upper band was isolated using CH2Cl2and the residue obtained upon evaporation (4.89 mg, 0.06%, from dry callus) was analyzed at 400 MHz NMR (Bruker, CDCl3, δ 7.24ppm) δ 7.85 (1H, d, J=8.6Hz, H3); 8.23 (1H, brd, J=8.6Hz, H4); 7.81(1H, brd, J=8.1Hz, H6); 7.51 (1H, m, H7); 7.76 (1H, t, J=7.9Hz, H8); 8.18 (1H, brd, J=7.9Hz, H9); 8.20 (2H, d, J=7.0Hz, H2’ and 6’); 7.55 (2H, t, J=7.0Hz, H3’ and 5’); 7.48 (1H, t, J=7.0Hz, H4’). EI-MS and HR-EI-MS were recorded on a Micromass Autospec spectrometer.
ACKNOWLEDGEMENTS
The authors gratefully acknowledge the “Jeunes Equipe” (IRD-BIOLEISH), for financial support. The programs UMSA-SIDA/SAREC and AECID-PCI (A/010794/07) project for chromatographic and spectroscopic support are also grateful.
REFERENCES
- KILLEEN, T., GARCIA, E., STEPHAN, B. Guía de árboles de Bolivia. 1993, Quipus s.r.l., La Paz, 709.
- PIRANI, J. Botanical Journal of the Linnean Society, 2004, 144, 365
- BOURDY, G., QUENEVO, C., GIMÉNEZ, A. Tacana, conozcan nuestros árboles, nuestras hierbas, 1999, Plural, La Paz: 414-416.
- FOURNET, A., HOCQUEMILLER, R., ROBLOT, F., CAVÉ, A., RICHOMME, P., BRUNETON, J. Journal of Natural Products, 1993, 56, 1547.
- FOURNET, A., FERREIRA, M., ROJAS, A., TORRES, S., FUENTES, S., NAKAYAMA, H., SCHININI, A., HOCQUEMILLER, R. Antimicrobial Agents and Chemotherapy, 1996, 40, 2447.
- GIMÉNEZ, A., AVILA, A., RUIZ, G., PAZ, M., UDAETA, E., TICONA, J., SALAMANCA, E., PAREDES, C., RODRÍGUEZ, N., QUINTS, K., FERAUDY, C., GUTIÉRREZ, I., CHUQUI, R., QUENEVO, C., DALENCE, M., BASCOPE, M. Revista Boliviana de Química, 2005, 22, 94.
- GIMÉNEZ, A., MACHICADO, S., MAGARIÑOS, W., SOSA, F., CHOQUE, O. Ethics aval from the National Committee of Bioethics project UMSA-ASDI-SAREC “Infectious diseases new alternative therapies: Evanta in the treatment of leishmania cutanea”, 2006, Bolivia.
- PAZ, M., VÁZQUEZ, F., CHUQUI, R., PAREDES, C., SAUVAIN, M., GIMÉNEZ, A. Latin American Journal of Pharmacy, 2007, 26, 15.
- YEOMAN, M., YEOMAN, C. New Phytologist, 1996, 134, 553.
- VERPOORTE, R., CONTIN, A., MEMELINK, J. Phytochemistry Reviews, 2002, 1, 13.
- COLLIN, H. Plant Growth Regulation, 2001, 34, 119.
- HEA, J., JUNG, E., OH, H.M. Journal of Molecular Biology and Biotechnology, 2005,13, 87.
- LIU, S., ZHONG, J. Process Biochemistry, 1998, 33, 69.
- CHATTOPADHYAY, S., MEHRA, R., SRIVASTAVA, A., BHOJWANI, S., BISARIA, V. Applied Microbiology and Biotechnology, 2003, 60, 541-546.
- NOWAK, B., MICZYNSKI, K., HUDY, L. Acta Physiology Plant, 2007, 29, 479.












 uBio
uBio