Servicios Personalizados
Revista
Articulo
Indicadores
Links relacionados
Compartir
Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim v.22 n.1 La Paz 2005
ARTÍCULO ORIGINAL
THE ORGANIC CHEMISTRY NOTEBOOK SERIES, A DIDACTICAL APPROACH,® (I) A THEORETICAL MECHANISTIC APPROACH TO DIASTEROSELECTIVE SYNTHESIS OF CIS-1,2-DIALKENYLCYCLOPROPANOLS AND SUBSEQUENT OXY-COPE REARRANGEMENT BY JIN KUN CHA ET AL.
José Bravo
Department of Chemistry, Universidad Mayor de San Andrés, P.O. Box 303, La Paz Bolivia jbravo@umsa.bo
ABSTRACT
The Organic Chemistry Notebook Series, A Didactical Approach,® has a reviewing character and it is intended to meet the needs of students debuting in studies of synthesis, in order to increase their capabilities to understand in an explicit manner, synthesis works already published. This discussion analyzes the article published by Prof. Jin Kun Cha (former Professor at the University of Alabama at Tuscaloosa, currently Professor at Wayne University, Detroit) entitled: Diasteroselective Synthesis of cis-1,2-Dialkenylcyclopropanols and Subsequent Oxy-Cope Rearrangement.1
Key Words: Kulinkovich hydroxycyclopropanation, cis-1,2-dialkenylcyclopropanols, oxy-Cope rearrangement, hydroazulenic sesquiterpenes, theoretical reaction mechanism
ANALYSIS AND MECHANISTIC PROPOSALS
Prof. Cha and his collaborators became interested in this subject due to the wide occurrence of hydroazulenic sesquiterpenes and related bicyclic natural products.1 The synthesis method chosen by Cha and col., used the Cope rearrangement of cis-divinylcyclopropanes. On such basis they reported the diasteroselective synthesis of cis-dialkenylcyclopropanols (2: R=H and tert-butyldimethylsilyl) and subsequent oxy-Cope rearrangement (2 → 3). Cha and col. adopted a convenient assembly of cis-1,2-dialkenylcyclopropanols according to the already published Kulinkovich hydroxycyclopropanation. The global process can be envisaged according to the sequence in the following scheme.
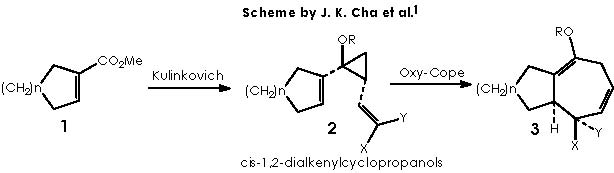
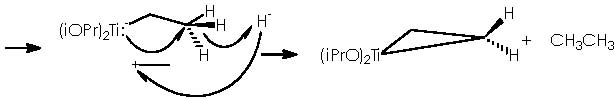
An approach to such reaction scheme will take us to first depict the oxy-Cope rearrangement of cis-1,2-dialkenylcyclopropanols, leaving the first part of the scheme (the Kulinkovich reaction) for afterwards. In this sense the oxy-Cope rearrangement of the cis-1,2-dialkenylcyclopropanols can be explained as follows:
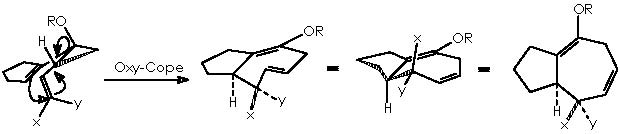
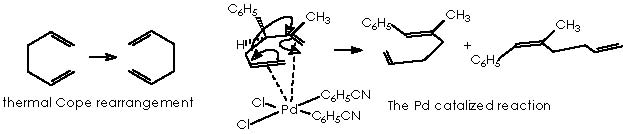
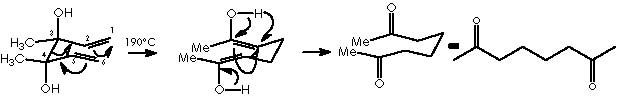
To better understand this oxy-Cope rearrangement we will review some related examples extracted from the literature. The preceding process of the oxy-Cope rearrangement is the well-known Cope [3,3]-sigmatropic rearrangement of 1,5-hexadienes. 2 A revised version uses a catalytic amount of palladium chloride bis(benzonitrile) complex, 3 which avoids the high temperature often required, being thence performed at room temperature. An example of this is the treatment of 2-methyl-3-phenyl-1,5-hexadiene that under palladium catalyst conditions affords the cis and trans dienes shown below.
The oxy-Cope rearrangement of 1,5-hexadienes with a hydroxyl substituent at C-3 provides, under thermal conditions, a useful route to δε-unsaturated aldehydes and ketones.
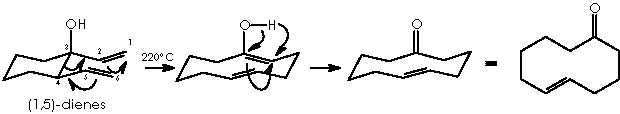
If there is an extra hydroxyl group at C-4 of the diene, 1,6-dicarbonyl compounds are produced.
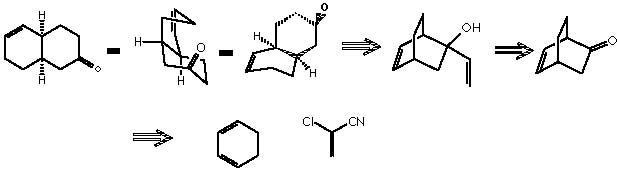
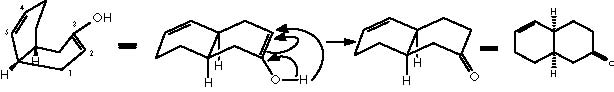
E. J. Corey and X. M. Cheng present an illustration of the oxy-Cope rearrangement in the retrosynthetic sense thus giving rise to the corresponding oxy-Cope transform.4 Although this transform does not belong to the most crucial structurally simplifying transforms, it can facilitate molecular disconnection through the rearrangement of the skeleton. This type of transforms, effect an essentially unchanged molecular complexity being however useful because they modify a target structure to allow the subsequent application of simplifying transforms. A frequent application of such transforms is to generate the retron for some other transform that can thus operate to simplify the structure.4 The oxy-Cope is a disconnecting but not a removing transform. The decalyn-ene-one target structure can be disconnected by applying first an oxy-Cope transform, followed by a functional group removing transform. A final disconnecting and simplifying Diels-Alder transform finishes the retroanalysis.
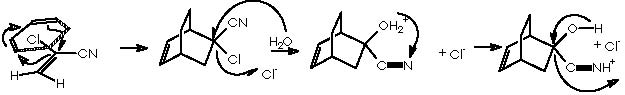
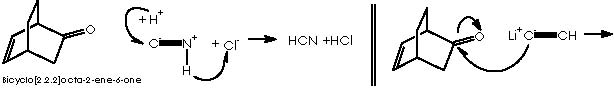
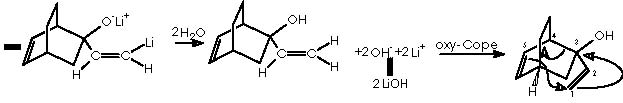
The oxy-Cope rearrangement is the last step of the synthesis which starts with the formation of the Diels-Alder adduct from the diene 1,3-ciclohexene and the dienophile chlorocianoethylene, followed by a nucleophilic substitution of the better departing group Cl- at expense of a water residue. Protonation of the avid nitrogen of the ciano group generates the departing of the :CNH+ group and the formation of the ketone group. This carbonyl group undergoes a nucleophilic attack by the basic lithium acetylene. Reduction of the resulting alkyne group by means of lithium aluminum tetrahydride and subsequent hydrolysis affords the gem-hydroxyethylene substituted bicyclo[2.2.2]octa-2-ene. This intermediate undergoes the oxy-Cope rearrangement to afford the final monoterpene.
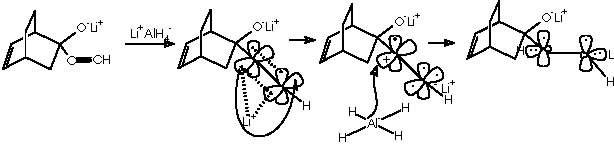
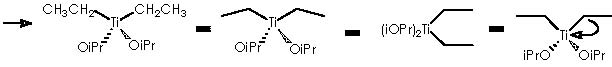
Let us examine now the procedure to effect the Kulinkovich hydroxycyclopropanation which involves treatment of a carboxylic ester with an excess (3 equiv) of Grignard reagent at –78° to 0°C in the presence of Ti(O-iPr)4 (1 equiv), to afford 1-alkylcyclopropan-1-ols.5 Among the various experiments of hydroxycyclopropanation by Kulinkovich and col., the example followed by Cha and col. consists of a catalytic process of 2 equiv of Grignard reagent, 5-10 mol % of Ti(O-iPr)4, ether, at 20 °C. This experiment shows the attractive mechanistic loop which involves the “double alkylation” of titanocyclopropane intermediate 5 as shown below.
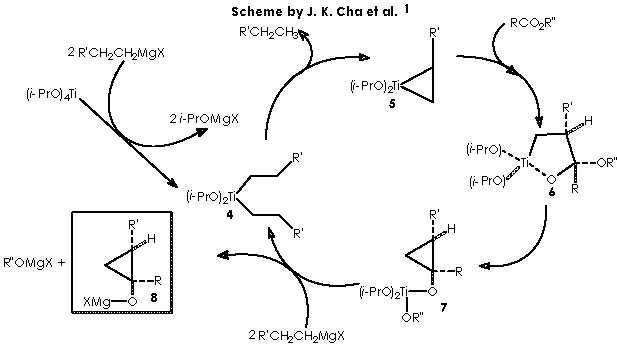
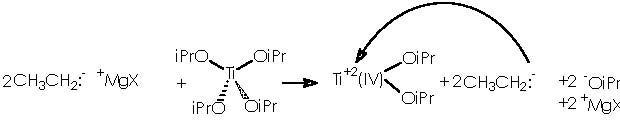
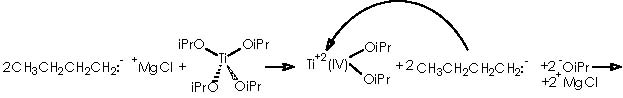
To depict this Kulinkovich hydroxycyclopropanation scheme we start with our 2 equiv of Grignard reagent or 2 R’CH2CH2MgX or 2 R’CH2CH2-+MgX. A first interaction occurs between Grignard reagent and the titanium tetraisopropoxy derivative. Titanium is linked to the four alkoxy substituents. The reaction provokes the expulsion of two alkoxy residues to leave a Ti 2+(IV) (OiPr)2 species. The metal di-cationic charge is used by two carbanions each from 1 equiv of the Grignard reagent, to obtain a dialkoxy-dialkyl titanium derivative (4).
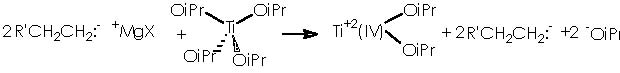
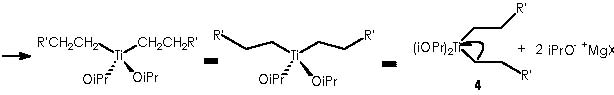
Titanium receives temporarily an extra electron from an alkyl methylene to afford the Ti-1(IV) (iPrO)2(CH2CH2R’) species and the carbonium ion R’CH2CH2+. A reductive nucleophilic attack by the anionic titanium leads to the apparition of the titanocyclopropane intermediate (5).
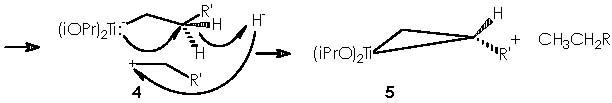
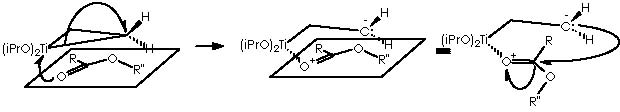
The next step in this pathway consists of the condensation of intermediate 5 with a carboxylic ester RCO2R”. The carbonyl group of this ester drives the reaction by interacting with the titanocyclopropane in a transition state that implies a co-planarity of both species. A nucleophilic attack by the nucleophile carbonylic oxygen over titanium dispatches the two electrons of the bond Ti-CH(R’)(CH2) over that carbon. Hence the titanocyclopropane splits therefore to afford a carbanion and a cationic oxygen over the same molecule. A nucleophilic attack over the carbonylic carbon (with an exacerbated electrophilic character) leads to the formation of a titanium furanoid ring.
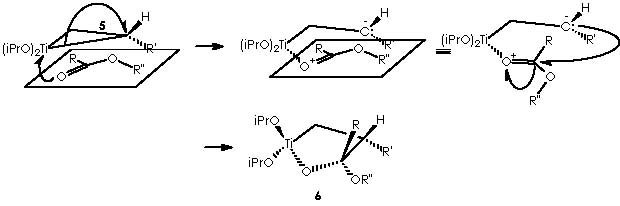
An intramolecular nucleophilic attack by the ethereal oxygen of the α -O-R” substituent group over the electrophilic titanium dispatches the two electrons of the Ti-CH2- bond on intermediate 6 to afford the corresponding split species bearing a carbanion and a cationic oxygen, which in turn recovers neutrality by splitting the four-membered, di-oxygenated (oxirane-type) ring. The result is the dipolar (a carbanion and carbonium ion) species that neutralizes the charged extremes into a substituted cyclopropane or intermediate 7.
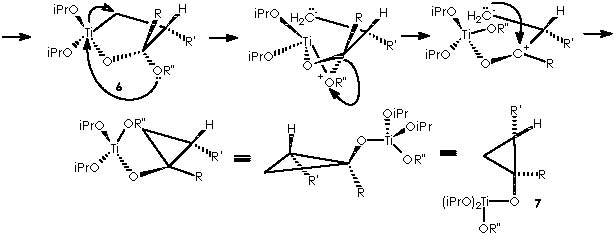
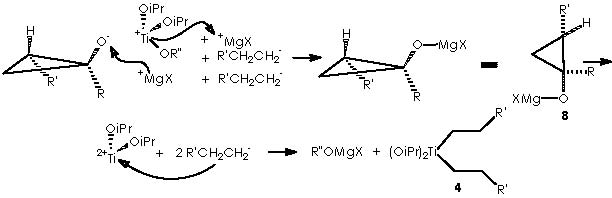
It is now time for two more equiv of Grignard reagent and for intermediate 7 to interact to afford species 8, closing thus our looping cycle. This meeting takes place by means of cationic interchange, or the cationic moiety of Grignard MgX+ that replaces cationic Ti+(OiPr)2(OR”) through the temporary apparition of an anionic oxygen. The remaining Ti2+(OiPr)2 species neutralizes the two anionic moieties liberated by the 2 equiv of Grignard reagent to form two new equiv of the species 4 to continue the cycle.
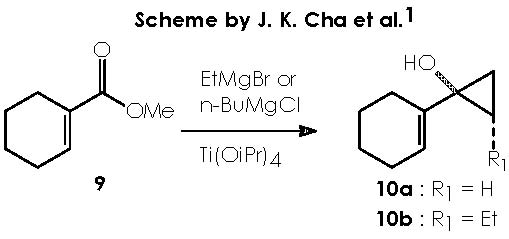
Cha and col. oriented their initial efforts to the application of this Kulinkovich reaction to the cyclopropanation of commercially available methyl-1-cyclohexene-1-carboxylate (9). The addition of EtMgBr or n-BuMgCl in the presence of Ti(OiPr)4 under catalytic conditions afforded the expected cyclopropanols 10a and 10b respectively.
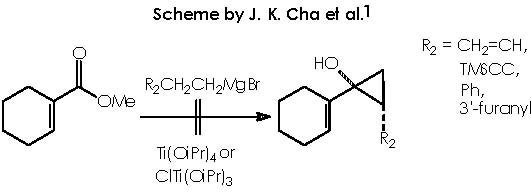
However these products did not accomplish the requirements to effect an ulterior oxy-Cope rearrangement. Hence, the use of CH2=CHCH2CH2MgBr was assayed with no success (a mixture of unidentified products was obtained). Also, no cyclopropanols could be obtained by the use of (2-(3’furanyl)ethyl))magnesium bromide or 2-phenethylmagnesium bromide. If the reactions with these substrates had been successful, this procedure would have provide a one-step synthesis of the required substrates for the subsequent oxy-Cope rearrangement.
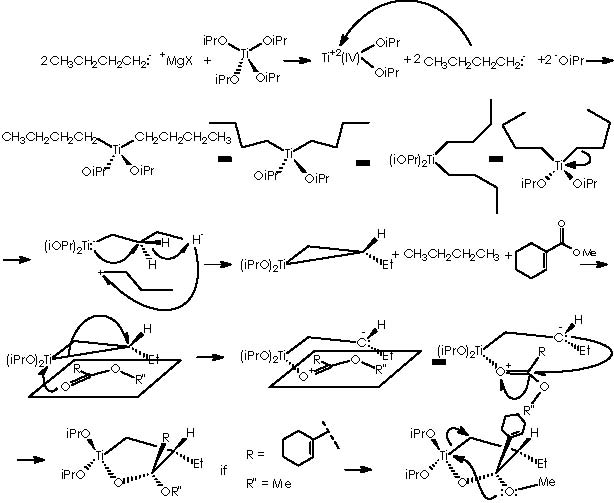
Let us now focus on a more detailed scope on the mechanistic procedure to obtain cyclopropanols 10a and 10b by Kulinkovich’s hydroxycyclopropanation. Following a parallel route to the already examined for the cyclopropanation loop cycle, we confront the tetraoxyisopropyl titanium derivative with the currently used Grignard reagent or ethyl magnesium bromide to obtain a dialkyl-dialkoxy titanium derivative.
Again, the titanium intermediate undergoes an excision to afford a carbonium ethyl and the Ti–1(IV) species which at its turn experiments an electronic re-accommodation through an intramolecular oxidative (dispatching a hydride) nucleophilic attack by Ti-1 to the methyl extreme carbon. The hydride and the ethyl carbonium neutralize mutually.
Let us consider now the condensation of the titanocyclopropane derivative with a carboxylic ester, which our case corresponds to compound 9. The result is the furane-type cycle titanium derivative.
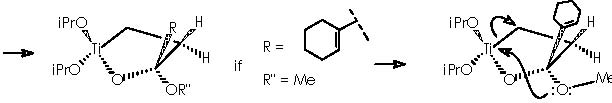
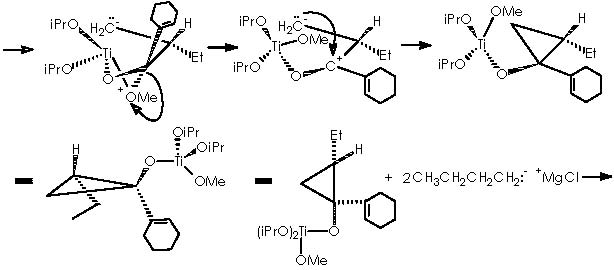
As described above, cyclopropanation comes out as a result of the intra-molecular nucleophilic attack of the methylether residue oxygen over titanium to form an α-methylene carbanion. The 4-membered diether cycle formed is broken by the methylated cationic oxygen which recovers neutrality by means of capturing one extra electron from a cyclic bond. The two emerging opposing charges neutralize each other affording the expected cyclopropane.
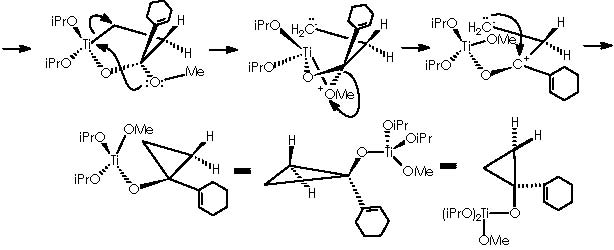
The substituted titanocyclopropane interacts now with two additional equiv of Grignard reagents to afford a cyclopropane oxymagnesium bromide.
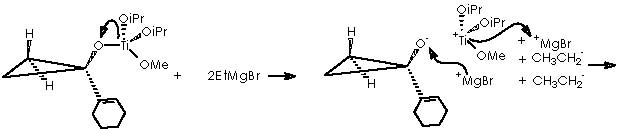
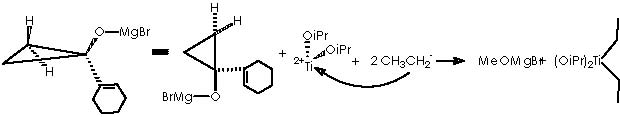
A protic medium as pure water is sufficient to destroy this magnesium bromide derivative (a kind of Grignard r.) to afford cyclopropanol 10a.
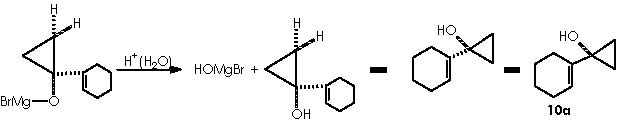
The cyclopropanol 10b can be obtained by the use of n-BuMgCl containing Ti(OiPr)4. The mechanistic proposal remains identical to the one already proposed for 10a as follows.
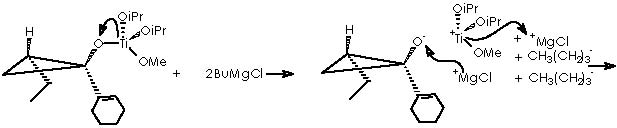
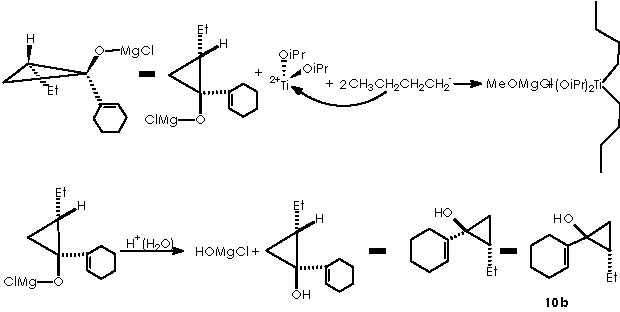
Disappointed by these results Cha and col. employed a 4-alkoxybutyl Grignard reagent.
Reagents 11a, b (see scheme by Cha and col. below) originated the desired cyclopropanols 12a,b in 77% and 46% respectively. The mechanistic approach is identical to that already extensively explained to obtain of 10a,b out of compound 9.
Removal of the protective group provided alcohol 13 as a single isomer. The hydroxyl function of the 4-alkoxy Grignard reagent can be set as protected or unprotected for both protecting groups (TIPS and THP).
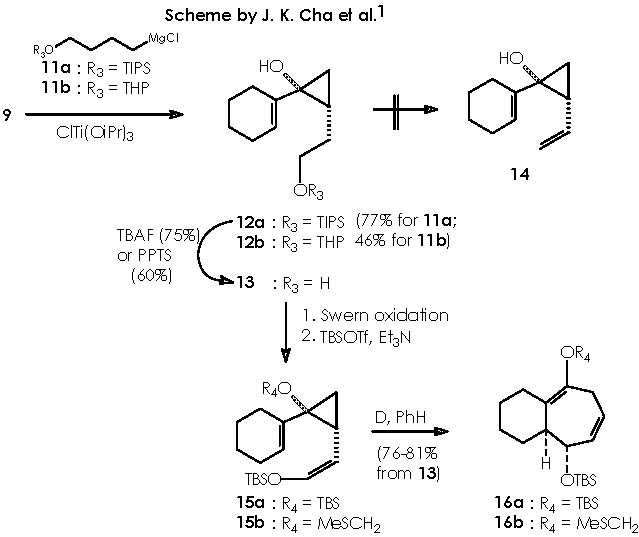
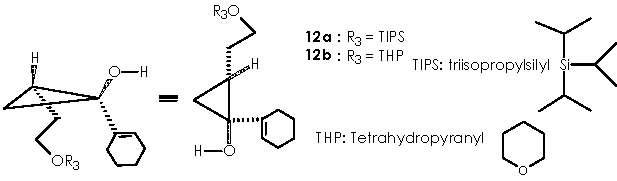
All mechanisms for protection or removal of the installed protective group, are nucleophilic substitutions taking place over the electrophilic oxygenated carbon bearing the hydroxyl group (if protecting) or the silyl or tetrahydropyranyl group (if removing the protective group). Let us examine the protection mechanism by TIPS first. TIPS is bonded to a leaving group (LG) besides the three isopropyl groups. A nucleophilic attack by the alkoxy group over silicon provokes the departure of the leaving group. The conditions of the protective reaction must be taken into account in order to prevent the environment from protic species to avoid damaging the magnesium chloride moiety of the Grignard reagent.
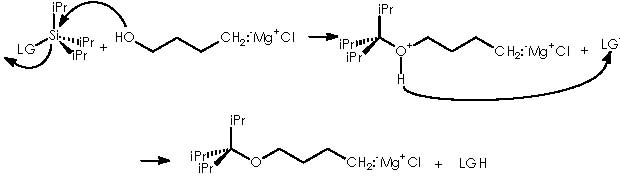
The reaction for uninstalling the protecting group to regenerate the alcohol function, in the protected product (12a) or the cyclopropane derivative that evolves (by expelling the protecting group) to cyclopropanol, implies the use of TBAF (tetra-n-butylamine fluoride) or PPTS.
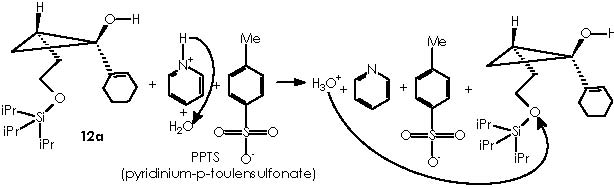
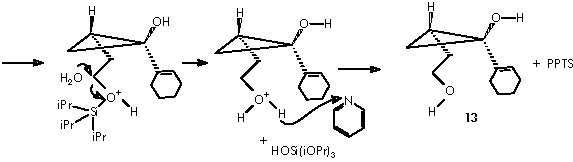
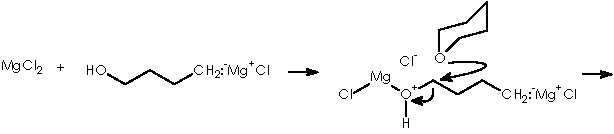
With respect to the protecting by using THP, the use of a Lewis acid catalyst can be envisaged to promote a nucleophilic attack by the protective group tetrahydropyrane (here the protecting procedure implies attacking the electrophilic oxygenated carbon of the alcohol function under catalysis by Mg). Mg2+ from Mg Cl2 can be mixed with the magnesian Grignard reagent thus avoiding a destructive protic catalyst. The resulting protected 4-alkoxy-n-butyl magnesium bromide, acquires a double cationic character that can be neutralized by the two chloride anions. This, however, can hinder its solubility. Tetrahydropyrane thus displaces the better leaving MgClOH species.
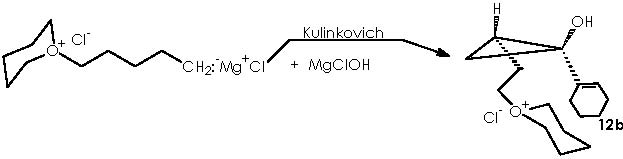
Removal of the tetrahydropyrane moiety from the cyclopropane derivative (12b) can be achieved by adding TBAF to the solution containing 12b to generate the alcohol 13.
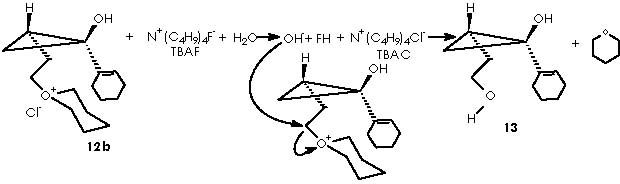
Unfortunately, several attempts by Cha and col. to convert 13 into olefin 14 were unsuccessful, a difficulty being finally circumvented by the application of a Swern oxidation of alcohol 13 or the formation of the silyl enol ether. Subsequent treatment with TBSOTf and triethylamine afforded almost exclusively the (Z)-silyl enol ether 15a (J = 5.9 Hz) along with a small amount of 15b. 1 Swern oxidation consists of oxidizing primary and secondary alcohols to aldehydes an ketones by action of a base on the derived alkoxysulphonium salt. 6,7 The conditions of reaction are mild and high yields of carbonyl compounds are generally obtained. One of these procedures involved reaction of the alcohol with dimethyl sulphoxide and dicyclohexylcarbodiimide in the presence of a proton source. 7, 8 The mechanism of the reaction involves initial formation of a sulphoxide-carbodiimide adduct which reacts with the alcohol to give the alkoxysulphonium ion. This then undergoes proton abstraction to form an ylid which collapses into the aldehyde and dimethyl sulphide by an intramolecular concerted process. This process can be visualized by the oxidation of the sensitive natural product 3’-O-acetylthymidine.
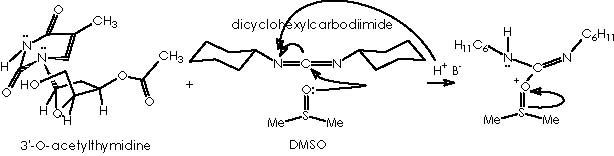
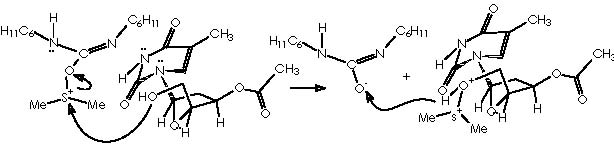
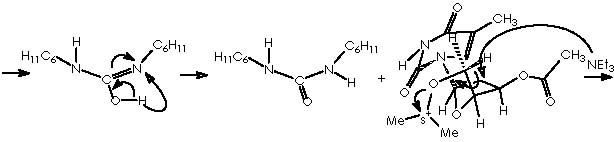
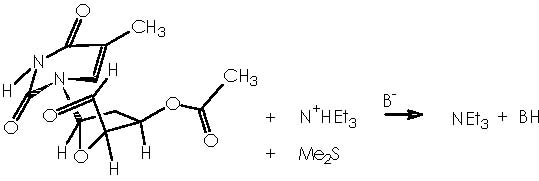
Applying this reaction mechanistic scheme to our Kulinkovich product (alcohol 13) to oxidize it according to Swern, we obtain the following pathway.
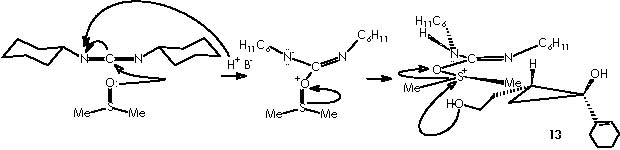
The sylil enol ether is obtained through a nucleophilic attack by the quaternary ammonium salt from which the enol nucleophile interacts over tri-ter-butylsilyl-oxy-triflate (TBSOTf) replacing the -OTf group.
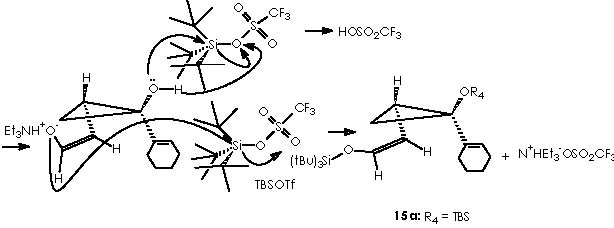
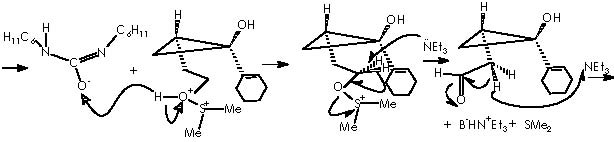
Simultaneously, the other hydroxyl moiety is replaced by a tri-ter-butylsilyl ether extremity to afford derivative 15a. The Z stereochemistry of the enol ether double bond could be established by the protons’ coupling constant of 5.9 Hz. At this stage the apparition of a synthetic companion (15b) could be explained by the formation of the species methylmethylene carbonium sulphide. In a competitive process to the protection of the hydroxyl moiety, this could be distracted by a electrophilic species other than the protecting group TBSOTf namely a methylmethylene carbonium sulphide.
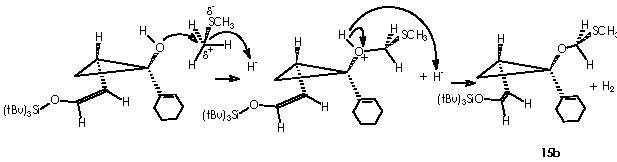
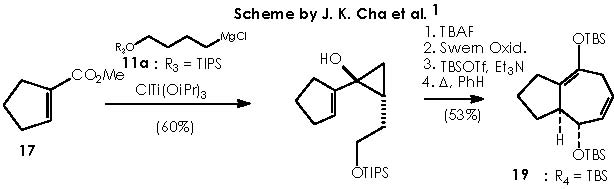
A nucleophilic attack over a carbon of dimethyl sulphide is conceivable due to the positive polar attraction of this carbon itself developed by the electronegative sulfur. The strongly basic hydride dispatched is immediately compensated by the acid H+ escaped from the acidic oxygen of the now DMSO+H moiety. Next in the reaction is the advent of the oxy-Cope rearrangement of the adducts 15a and 15b when heated at reflux in benzene. The enol ethers underwent the oxy-Cope rearrangement to provide bicyclic cycloheptadiene 16a (72% overall yield from 13) and 16b (4%).1 The mechanistic view for the oxy-Cope rearrangement was exposed at the beginning of this paper (first page). Cha and col. tried the identical synthetic sequence on methyl 1-cyclopentene-1-carboxylate (17).
The obtained compound was azulene 19 in 53 % overall yield by means of cyclopropanol 181 (a similar mechanistic approach to the one already developed until now can be applied to 17 by the reader).
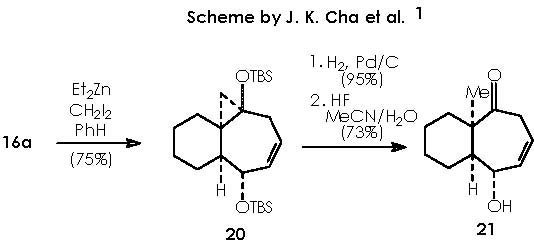
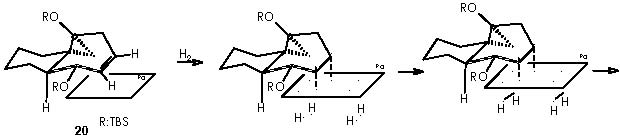
Once developed a convenient synthetic method for fused bicyclic systems 16a and 19, Cha and col. examined subsequent elaborations of the cycloheptadiene functionality. Simmons-Smith cyclopropanation (CH2I2, Et2Zn) of 16a gave cyclopropane 20 as a single diastereomer (75%).9
This reaction (S-S) is widely used for the synthesis of cyclopropane derivatives from alkenes by reaction with methylene iodide and zinc-copper or, better, zinc-silver couple. This valuable method permits to not to affect many functional groups when forming the cyclopropane moiety. 10, 11 Stereo-specifically, cis-addition of methylene to the less hindered side of the double bond occurs. The reactive intermediate is believed to be an iodomethylenezinc iodide complex which reacts with the alkene in a bimolecular process to give a cyclopropane and zinc iodide. When a neighboring hydroxyl group is present (allylic or homoallylic alcohol or ether) in 5- or 6-membered rings, the product with a cis-cyclopropane to the hydroxyl group is formed stereo-specifically. The transition state involves a co-ordination of oxygen to the zinc with subsequent transfer of the methylene group to the nearer face of the adjacent double bond.10
As an example of the S-S reaction let us evoke the following reaction.
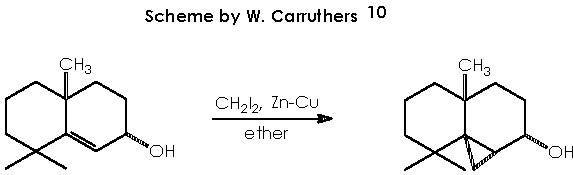
The intermediate iodomethylenezinc iodide complex that reacts with the alkene can be described as follows.
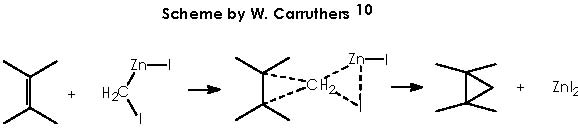
As a surface catalyst, the Zn offers to the medium avilable unpaired electrons which will incite homolytic excision of covalent bonds in CH2I2.
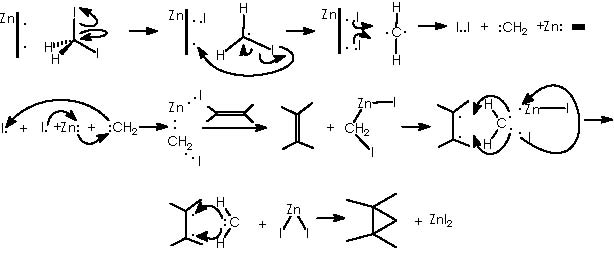
For the 6-membered cyclic system we can picture the corresponding transition state as the one developed by coordination of Zn with the allylic alcohol hydroxyl function thus defining the formation of the cyclopropane moiety also in a β position (just as the OH group position).
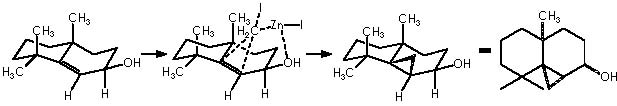
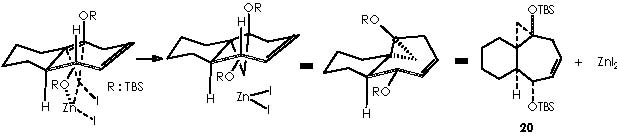
Applying these mechanisms to 16a, we obtain its cyclopropanation by Simmons-Smith or compound 20.
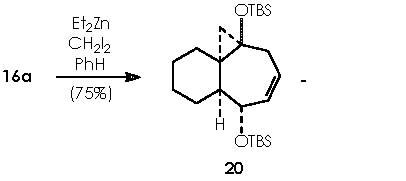
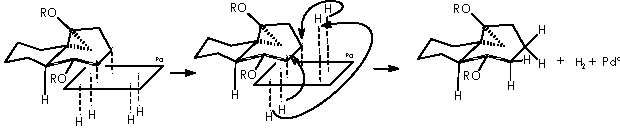
In this reaction, Zn appears as Zn2+, thus we must reduce this oxidized state by means of procuring 2 electrons. Iodomethylene affords carbene and two iodine atoms which can oxidize to afford two electrons. The two cationic iodine atoms neutralize with carbanion species from diethyl zinc (II). The Zn° species thus obtained can act catalytically when adding carbene to the corresponding double bond following the already exposed mechanism. The coordinating agent (Zn), acts in this case with a TBSO group situated in an α position (equatorial), thus giving rise to a transition state that involves a homoallylic alkoxy group. As expected, the cyclopropane moiety will be formed also in an α position. The stereochemical assignment was firmly established by Cha and col. on the basis of the NOE difference experiment.
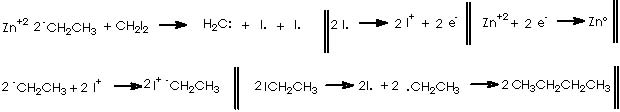
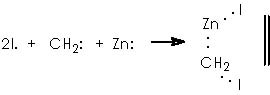
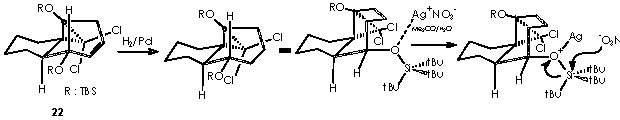
The derivative 21 was obtained by treatment with H2/Pd, to saturate the only remaining double bond, and subsequent oxidation by HF (MeCN-H2O) to afford the ketone function. Removal of the OTBS protecting group by an acid agent followed this partial stepwise procedure.
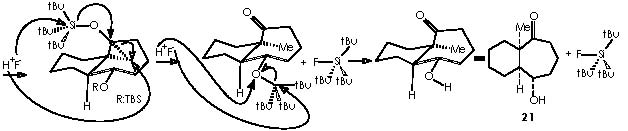
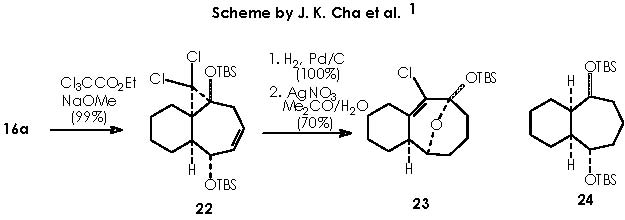
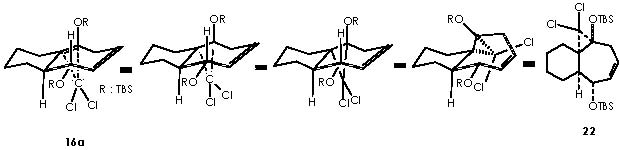
Next in this structural elaboration by Cha and col., the treatment of 16a with dichlorocarbene to afford dichlorocyclopropane 22 was reported. Hydrogenation on Pd of 22 and subsequent submission to silver nitrate (in aqueous acetone) gave rise to a 1 C ring expansion or ketal 23. In a parallel reaction , they obtained a full reduction of unsaturated bonds or compound 24. A variety of methods is available for the generation of carbenes.12 Accordingly, dihalogenocarbenes can be obtained by action of a base on a haloforme or also by the halophenylmercury compounds PhHgCXYZ (X, Y, Z = halogens). Such compounds can be prepared from phenylmercury (II) halide interacting with haloform and potassium t-butoxide as follows.
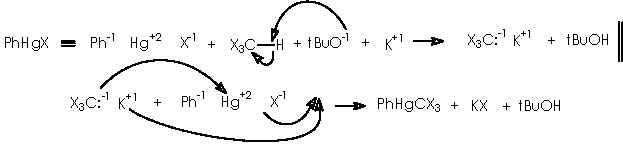
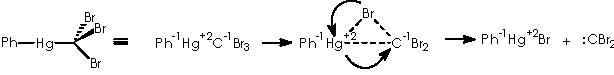
This type of compounds are source of halogenocarbenes, according to a mechanism that involves the moving of a bromide from carbon of the haloform moiety to mercury, passing by a transitory mercurobromocarbon tricycle. The transitory state collapses into a phenylbromomercury adduct and the expected dibromo carbene.12
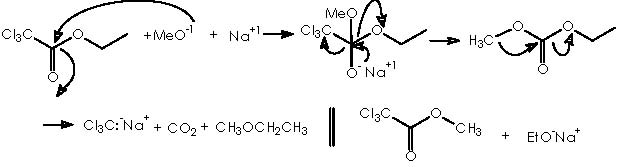
Hypothetically, by using this method, dichlorocarbene could be obtained from the chlorine source trichloroacetic ethyl ester. This may take the place of the haloform. The alkaline alkoxide or sodium methoxide in this case, acts more as a nucleophile than as a base in contrast with the role attributed to potassium ter-butoxide as shown above. The competitiveness to go out of the two potential leaving groups after insertion of the methoxy group over the carbonyl, namely a trichloromethane carbanion and the ethoxy group, is assumed to be comparable. The collapse of the negatively charged oxygen of the sodium salt intermediate into a carbonyl group could generate on one hand a trichloroacetic methyl ester, and/or a trichloromethane carbanion and a very stressed di-oxygenated carbonyl compound on the other, which must forcefully decarboxylate to give CO2 and ethylmethyl ether.
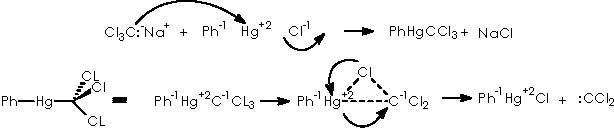
Thence to go ahead on the generation of the dichlorocarbene, the use of Hg 2+ must be supposed. The species to be added to generate a halophenyl mercury (PhHgCXYZ) is a phenylmercury halide, PhHgCl. The already existing trichloromethane carbanion as already mentioned, came out not from the haloform/potassium ter-butoxide couple, but in our case from the trichloroacetic ethyl ester residue as shown above. Following the mechanism described above for the interaction between the carbanion and the phenylmercury halide we obtain the following.
The addition of dichlorocarbene to the double bond occurs from an α position to afford the corresponding α-gem-dichlorocyclopropane moiety. The α position of this is obliged by the very hindering β-oriented OTBS group.
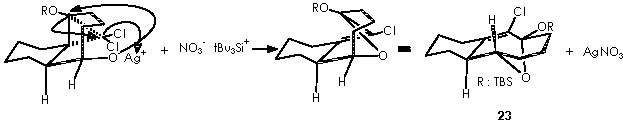
The next transformation is a hydrogenation of compound 22 by means of Pd catalyst and a ring expansion of it by means of application of silver nitrate in aqueous acetone to afford compound 23. The cationic silver replaces the tri-ter-butylsiliconium ion thus generating a silver salt. The anionic oxygen thus generated provokes an intramolecular nucleophilic attack over the electrophilic oxygenated carbon supporting the protecting group or C-OTBS. As a result, the ether-bridge-containing bicycle is formed. The chloride anion is neutralized by the silver cation.
ACKNOWLEDGEMENT
The author expresses his gratitude to Prof. Eduardo Palenque from the Physics Department, Universidad Mayor de San Andrés, for his bibliographic support.
REFERENCES
1 J. Lee; H. Kim; J. K. Cha, J. Am. Chem. Soc., 1995, 117, 9919.
2 W. Carruthers, Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 175-176. [ Links ]
3 L.E. Overman, A. F. Renaldo, Tetrahedron Lett. 1980, 24, 3757.
4 E .J. Corey, X-M Cheng, The Logic of Chemical Synthesis, John Wiley and Sons, Inc., 1989, New York, U.S., pp 9-13 [ Links ]
5 O. G. Kulinkovich, S. V. Sviridov, D. A. Vaasilevskii, T. S. Pritytskaya, Zh. Org. Khim., 1989, 25, 2244.
6 A. J. Mancuso, D. Swern, Synthesis, 1981, 165.
7 W. Carruthers, Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 357-358. [ Links ]
8 W. W. Epstein, F. W. Sweat, C hem. Rev., 1967, 67, 247.
9 J. Furukawa, N. Kawabata, J. Nishimura, Tetrahedron, 1968, 24, 53.
10 W. Carruthers, Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 95-96. [ Links ]
11 H. E. Simmons, T. L. Cairns, S. A. Vladuchick, C. M. Hoiness, 1973, Organic Reactions, 20, 1.
12 W. Carruthers, Some Modern Methods of Organic Synthesis, Cambridge University Press, 3rd ed., 1987, Worcester, U.K., pp. 87-97. [ Links ]












 uBio
uBio 

