Revista Boliviana de Química
versión On-line ISSN 0250-5460
Rev. Bol. Quim v.23 n.1 La Paz 2006
ARTÍCULO ORIGINAL
SIMULTANEOUS DETERMINATION OF GLUCOSE, MALTOSE AND SUCROSE IN STARCH OF SIX VARIETIES OF CASSAVA
C. Carola C. Rojas1,3, Baboo Nair2, Adelina Herbas3, Björn Bergenståhl1
1Food Technology, Lund University, P.O Box 124, SE-221 00 Lund, Sweden
2Applied Nutrition, Lund University, P.O Box 124, SE-221 00 Lund, Sweden
3Centro de Alimentos y Productos Naturales, Universidad Mayor de San Simón, Cochabamba, Bolivia
ABSTRACT
An enzymatic method has been developed to simultaneously determine glucose, maltose and sucrose in tubers The principle of the method is based on an enzymatic cleavage of these disaccharides and specific measurement the resulting glucose using an enzymatic procedure described by Holm (3).
The study comprises two parts; the first one includes modifications of the pretreatment method to minimize errors due to ethanol used to precipitate polymeric material interfering with the activity of α-glucosidase and invertase. In the second part the usability of the method has been probed by employing it on six samples of cassava that come from Chapare-Bolivia.
Key words: Enzymatic hydrolysis, analysis, glucose, maltose, sucrose, Cassava. sugars determination is by using enzymatic
INTRODUCTION
Cassava (Mannihot esculenta Crantz) is a tropical root that has mainly carbohydrates in its composition such as starch, glucose, sucrose and fructose Huang (13). Lewthwaite (8) and Takata (11) have reported important changes in the composition of potato saccharides during its processing by showing variations in the sugars concentration which is an essential variable to determine the quality of the food. Reducing sugars has been identified as critical in potato for the tendency for acrylamide formation during heat processing of potato (14).
To determine the sugars content in plants and food, different techniques have been developed such as High Performance Liquid Cromatography (HPLC) (4,7); Gasses Cromatography (GC) (10). However, these techniques require considerable investments for the equipments and also normally demand a high cost of operation including larges periods of time for the analysis. An alternative procedure for methods, in this case the procedure is relatively simple and requires and UV-Vis spectrophotometer. The enzymatic kits are available for many sugars (1, 2, 9, 11, 5, 6) and also some other assays set have been developed for the determination of sugars (i.e.maltose, saccharose) by Dörner (5). Therefore, the main objective of this study was to develop a simple enzymatic method to determine the content of sugars (glucose, maltose and sucrose) in order to achieve a method with good performance in terms of accuracy-precision, easy-handling and simultaneous quick determination. The methodology is applied on a determination of the content of sugars in cassava of six varieties was characterized.
RESULTS AND DISCUSSION
To evaluate the ethanol interference, a screening with different concentrations of ethanol was performed to determine glucose as well as maltose and sucrose response obtained
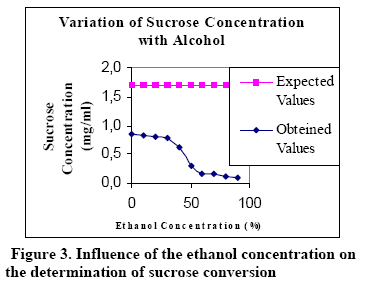
Figures 1, 2 and 3 show the relationship between the ethanol concentration (in the final samples) and conversion (end concentration of the sugar).
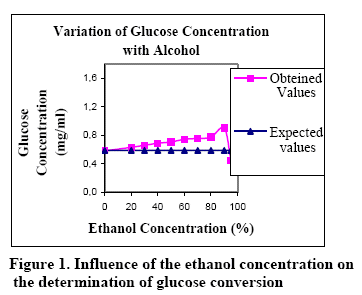
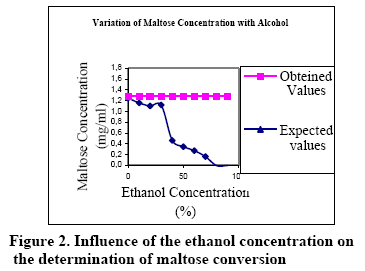
As shown in the Figure 1, the obtained glucose response varies as a function of the ethanol concentration. If compared the true values of glucose concentration (expected values) vs the obtained response (obtained values), it can be seen that the difference is not significant (less than 2%) up to 48% of ethanol in the final sample. As shown in the Figure 2, the obtained maltose response is more sensitive against the ethanol concentration. If compared the true values of maltose concentration (expected values) versus the obtained response (obtained values), it can be seen that the difference is not significant (less than 2%) up to 30% of ethanol in the final sample.
Figure 3 shows that there is a significant variation between the true values of sucrose (expected values) and the ones obtained by the analysis. This implies that there is a marked influence due to the presence of ethanol even when working at low concentrations.
Simultaneous analysis of glucose, maltose and sucrose in samples of cassava.
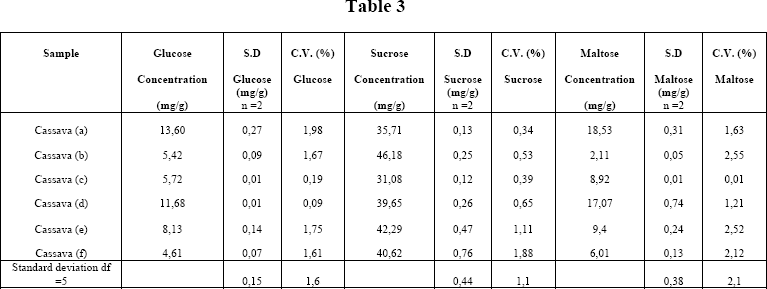
Once the technique was standardized, six varieties of Bolivian cassavas were analyzed; the results are shown in Table 3
Therefore, an experimental design (See Table 1) was performed to improve the sucrose conversion by modifying the conditions based on changes in incubation temperature, incubation time and initial concentration of the enzyme.
According to table 1, the appropriated conditions for the process to achieve best conversions were: incubation time = 60 minutes, incubation Temperature = 24 °C,V enzyme = 640 μL (150 U/ml), C ethanol = 30%)
Determination of maltose and sucrose concentration
Based on the experiments and corrections, the sugar concentrations were calculated with the empiric deduced equations of Table 2.

The results show a high variability in terms of glucose for the six varieties, from 5 to 14 mg/g DM of sample. The content of sucrose varies between 36 and 46 mg/g DM and the content of maltose varies between 2 to 19 mg/g DM
Precision of the method
The precision of the method is analyzed from the data in Table 3. with 2 replicates, standard deviations and coefficients of variance were calculated for the six varieties of cassava. The standard deviation for glucose is 0.15 mg/g and about twice times higher for sucrose and maltose wich is determined in two steps. The coefficient of error depends very much of the concentration (varies between 1 and 2%) and it is lowest for sucrose, present in a higher amount.
Method SpecificityThe α-Glucosidase hydrolyzes the terminal, nonreducing 1,4-linked α-D-glucose residues with release of α-D-glucose (15)
EXPERIMENTAL
a) Substrates
The varieties of Cassava were: Bobore (a), Noveton (b), Valencia (c), Amarilla (d) and Cambayuca (e).Those come from “Estación Experimental la Jota”, located in Chapare -Cochabamba -Bolivia The Bobore (f) sample comes from “Chimore. Experimental Plantations” IBTA-Chapare It is located in the Municipio de Chimoré of Chapare to 200 Km of the Cochabamba city). As a reference system potato starch p.a. was used. (Sigma-Aldrich)
b) EquipmentsAn analytical balance (Metler Toledo AE160), a water bath (Heto Birkeröd, Type 02 PG623, N° 7912, spectrophotometer (Perkin Elmer UV/VIS Lambda2).
c) ReagentsReagents: anhydrous D-Glucose, Maltose Sucrose p.a. Buffer Solutions: Acetate Buffer 0,1 M, pH 6,6, Acetate Buffer 0.1 M, pH 4.6 and Buffer Tris 0.5 M. pH 7.
d) Enzymesα-amylase (Fungamyl 800L) (Novo Nordisk, Copenhagen, Denmark), α-Glycosidase 240 U/ml (Sigma-Aldrich), Invertase 150 U/mg (Sigma-Aldrich) and Glucose Oxidase/peroxidase (3), and Dihydrochloride like stain
f) Analytical Procedure.The method basically consists of determinations of the glucose concentration after different steps of the hydrolysis. Glucose concentration was evaluated from samples that contain maltose by using α-glucosidase and sucrose by using invertase. The background concentration of glucose was determined by an initial glucose analyses.
Standard Curve. Initially, 50 mg of anhydrous Dglucose was dissolved in 1000 mL of ethanol (48%). Dilutions were prepared ranging form 0.01-0.05 mg/mL to obtain the standard curve. 4 mL of glucose oxidase/peroxidase enzyme solution was added then they were incubated for one hour at room temperature (20°C). The wavelength used for the determination was 450 nm.
Sample Preparation: 1 g of milled vacuum dried sample (particle size< 0.8 mm) ) was suspended in ethanol (46%) to make up 100 ml of suspension, the aim of the alcohol treatment is to precipitate all starch and proteins. The system was extracted in the shaker at room temperature during 40 min. Afterwards the suspension was filtrated by paper media filter 0.22 µm.
Glucose Analysis: 1 mL of the filtered solution was mixed with 1 mL of buffert (pH 6) and 4 mL of glucose oxidase/peroxidase (5,6 g/100 ml) as described by Holm (3). The mixture was incubated for one hour at room temperature and then the absorbance was read at 450 nm.
Maltose Analysis: 1 mL of the filtered solution was mixed with 1 mL of acetate buffer pH 6.6 and 0.2 mL of α-glucosidase (240 U/ml), the mix was incubated for one hour at room temperature according to the procedure by Gutmann (6). Afterwards, 0.1 mL of the obtained solution was mixed with 1.9 mL of water and 4 mL of glucose oxidase/peroxidase (5.6 g/100 ml) as described. Holm (3).The incubation process was carried out for one hour at room temperature. The absorbance was read at 450nm.
Sucrose Analysis: 0.3 mL of the filtered solution was mixed with 1 mL of acetate buffer pH 4.6 and 0.640 mL of invertase (150U/ml), the mixture was incubated for one hour at room temperature; then, 0.5 mL of 0.5 M Tris-buffer pH 9 was added as described by Dömer (5). Afterwards, 0.6 mL of the obtained solution mixed with 1.4 ml of water and 4 mL of glucose oxidase/peroxidase (5,6 g/100 ml) as described. Holm (3). The incubation process was carried out for one hour at room temperature. The absorbance was read at 450 nm.
Interference corrections of the ethanol in the determinations:The samples contain 46% of etanol during the extraction of sugars. It can be assumed that the etanol may inhipit the enzymatic reactions. Thus, the sensitivity of α-glucosidase, invertase and glucose oxidase against ethanol was tested.
Improvement on the sucrose conversion:In order to improve the invertase hydrolysis an experimental design was carried to evaluate the conversion by taking into account the temperature. Incubation time and enzyme concentration are considered as the variables with more influence in the process.
CONCLUSION
The presented method allows for measurements of the content of free glucose, maltose and sucrose in starch rich foods such as cassava. The method is quick, easy and reliable. However, in the analyses, the starch and proteins needs to be precipitated using ethanol. The study showed that the maximum concentration of ethanol to avoid interferences for determining free glucose, maltose and sucrose were 48%, 30%, 30% respectively. The conditions to perform the sucrose analysis need to be adjusted relative to previously published standard conditions (2).
ACKNOWLEDGEMENTS
The study was supported by Swedish International Development Agency (SIDA/SAREC) in the project “Desarrollo de nuevos productos y mejoramiento de las tecnologías tradicionales de procesamiento de raices tropicales”, between San Simon University (Bolivia) and Lund University (Sweden).
REFERENCES
(3) HOLM, J., BJÖRCK, I., DREWS, A., & ASP, N.-G. A rapid method for analysis of starch. Starch/Stärke, 1986, 38 , 224-226. [ Links ]
(4) JACK N. DAVIES, RONALD J. KEMPTON “Changes in the individual sugars of tomato fruit during ripening.” J. Sci. Food Agric, 1975, 26, 1103-1110.
(5) Klaus M. Dörner “Quantitative Determination of lactose, Maltose, and Sucrose in Urine” European Journal of Pediatrics 1971, 26 , 45-52.
(6) GUTMANN, I. Methods of Enzymatic Analysis. Ed. HU Bergmeyer - New York: Academic Press, 1974. [ Links ]
(7) HAILA K, K. J., HÄKKINEN U, TAHVONEN R.. “Sugar and organic acid contents of vegetables consumed in Finland during 1988-1989.” J Food Comp Anal 1992, 5, 100-107.
(8) LEWTHWAITE, S. L. S., K. H.; TRIGGS, C. M. “Free sugars composition of sweet potato cultivars after storage.” Crop Hortc. Sci. 1997 25, 33-41.
(9) MOELLERING H, G. W. “Determination of citrate with citrate lyase”. Anal Bichem, 1996, 17, 369-376.
(10) MOLNÁR-PERL I, M. M. “Rapid method for the silultaneous GC quantitation of acids and sugars in fruits and vegetables.” Food Add Contamin 1992, 9, 505-514.
(11) TAKAHATA, Y. N., T. SATO, T. “Changes in carbohydrates and enzyme activities of sweet potato lines during storage.” J. Agric. Food Chem. 1995, 43, 1923-1928.
(12) JOYCE S. VELTEROP, FEMKE VOS. “A Rapid and Inexpensive Microplate Assay for the Enzymatic Determination of Glucose, Fructose, Sucrose, L - Malate and Citrate in Tomato (Lycopersicon esculentum) Extracts and in Orange Juice.” Phytochemical Analysis, 2001, 12, 299-304.
(13) Y. H. HUANG, D. H. P., A. W. KILILI, AND C. E. JOHNSON. “Changes in invertase Activities and Reducing Sugar Content in Sweetpotato Stored at Different Temperatures” Journal of Agricultural and Food Chemistry, 1999, 47, 4927-4931.
(14) BECALSKI A, LAU BPY , LEWIS D , SEAMAN SW , Acrylamide in Foods: Occurrence, [ Links ]
(15) MATSUSAKA, K.; CHIBA, S.; Sources, and Modeling. J. Agricultural Food SHIMOMURA, T.;Purification and substrate Chem, 2003, 51: 802-808 specificity of brewer's yeast alpha-glucosidase, Agric. Biol. Chem., 1977, 41, 1917-1923 [ Links ]












 uBio
uBio 

